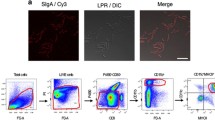
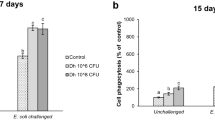
Abstract
Recombinant Saccharomyces cerevisiae strains expressing HIV antigens have shown promising pre-clinical results. Probiotic S. cerevisiae strains naturally induce gut immunity; thus, genetically engineered probiotic strains could be used to stimulate immune responses against HIV in the mucosa. Probiotic strains have a higher rate of heterologous protein production, meaning higher antigen’s epitope expression levels per yeast cell. We expressed HIV-1 Gag protein in the probiotic yeasts’ surface, which was eagerly phagocytosed by and induced type 1 polarization of human monocyte-derived dendritic cells (DCs) from healthy donors in vitro. We further matured DCs derived from HIV-1+ donors with transformed yeasts and incubated them with autologous T cells. Only DCs matured with Gag-expressing probiotic strains were able to efficiently present antigen to CD8+ T cells and induced their clonal expansion. Our results show that genetically engineered probiotic S. cerevisiae strains are a promising vaccination strategy against HIV.





Similar content being viewed by others
Change history
20 May 2019
In the Funding section, the following statement is missing: The MACS cohort study was supported by the NIH, National Institute of Allergy and Infectious Diseases grant U01-AI35041.
References
Ardiani A, Higgins JP, Hodge JW (2010) Vaccines based on whole recombinant Saccharomyces cerevisiae cells. FEMS Yeast Res 10:1060–1069
Barron MA, Blyveis N, Pan SC, Wilson CC (2006) Human dendritic cell interactions with whole recombinant yeast: implications for HIV-1 vaccine development. J Clin Immunol 26:251–264. https://doi.org/10.1007/s10875-006-9020-8
Bax M, Kuijf ML, Heikema AP, van Rijs W, Bruijns SCM, García-Vallejo JJ, Crocker PR, Jacobs BC, van Vliet SJ, van Kooyk Y (2011) Campylobacter jejuni lipooligosaccharides modulate dendritic cell-mediated T cell polarization in a sialic acid linkage-dependent manner. Infect Immun 79:2681–2689. https://doi.org/10.1128/IAI.00009-11
Berk E, Muthuswamy R, Kalinski P (2012) Lymphocyte-polarized dendritic cells are highly effective in inducing tumor-specific CTLs. Vaccine 30:6216–6224. https://doi.org/10.1016/j.pestbp.2011.02.012.Investigations
Boder ET, Wittrup KD (1997) Yeast surface display for screening combinatorial polypeptide libraries. Nat Biotechnol 15:553–557. https://doi.org/10.1038/nbt0697-553
Braga JM, Oliveira RR, de Castro Martins R, Vieira LQ, Sobrinho APR (2015) Assessment of the cytotoxicity of a mineral trioxide aggregate-based sealer with respect to macrophage activity. Dent Traumatol 31:390–395. https://doi.org/10.1111/edt.12190
Cereda V, Vergati M, Huen NY, di Bari MG, Jochems C, Intrivici C, Gulley JL, Apelian D, Schlom J, Tsang KY (2011) Maturation of human dendritic cells with Saccharomyces cerevisiae (yeast) reduces the number and function of regulatory T cells and enhances the ratio of antigen-specific effectors to regulatory T cells. Vaccine 29:4992–4999. https://doi.org/10.1016/j.vaccine.2011.04.101
Colleton BA, Huang XL, Melhem NM, Fan Z, Borowski L, Rappocciolo G, Rinaldo CR (2009) Primary human immunodeficiency virus type 1-specific CD8+ T-cell responses induced by myeloid dendritic cells. J Virol 83:6288–6299. https://doi.org/10.1128/JVI.02611-08
Dementhon K, El-Kirat-Chatel S, Noël T (2012) Development of an in vitro model for the multi-parametric quantification of the cellular interactions between Candida yeasts and phagocytes. PLoS One 7. https://doi.org/10.1371/journal.pone.0032621
Detels R, Jacobson L, Margolick J, Martinez-Maza O, Muñoz A, Phair J, Rinaldo C, Wolinsky S (2012) The multicenter AIDS cohort study, 1983 to …. Public Health 126:196–198. https://doi.org/10.1016/j.puhe.2011.11.013
Douradinha B, Reis VCB, Rogers MB, Torres FAG, Evans JD, Marques ETA (2014) Novel insights in genetic transformation of the probiotic yeast Saccharomyces boulardii. Bioengineered 5:1–9
Fietto JLR, Araújo RS, Valadão FN, Fietto LG, Brandão RL, Neves MJ, Gomes FCO, Nicoli JR, Castro IM (2004) Molecular and physiological comparisons between Saccharomyces cerevisiae and Saccharomyces boulardii. Can J Microbiol 621:615–621
Fiore-Gartland A, Manso BA, Friedrich DP, Gabriel EE, Finak G, Moodie Z, Hertz T, De Rosa SC, Frahm N, Gilbert PB, McElrath MJ (2016) Pooled-peptide epitope mapping strategies are efficient and highly sensitive: an evaluation of methods for identifying human T cell epitope specificities in large-scale HIV vaccine efficacy trials. PLoS One 11:e0147812. https://doi.org/10.1371/journal.pone.0147812
Freed EO (2015) HIV-1 assembly, release and maturation. Nat Rev Microbiol 13:484–496. https://doi.org/10.1038/nrmicro3490
Generoso SV, Viana ML, Santos RG, Arantes RME, Martins FS, Nicoli JR, MacHado JAN, Correia MITD, Cardoso VN (2011) Protection against increased intestinal permeability and bacterial translocation induced by intestinal obstruction in mice treated with viable and heat-killed Saccharomyces boulardii. Eur J Nutr 50:261–269. https://doi.org/10.1007/s00394-010-0134-7
Generoso SV, Viana M, Santos R, Martins FS, Machado J a N, Arantes RME, Nicoli JR, Correia MITD, Cardoso VN (2010) Saccharomyces cerevisiae strain UFMG 905 protects against bacterial translocation, preserves gut barrier integrity and stimulates the immune system in a murine intestinal obstruction model. Arch Microbiol 192:477–484
Globeimmune (2016) Tarmogen platform. https://www.globeimmune.com/platform/technology/. Accessed 11 May 2016
Hamilton R, Watanabe CK, de Boer HA (1987) Compilation and comparison of the sequence context around the AUG startcodons in Saccharomyces cerevisiae mRNAs. Nucl Acids Res 15:3581–3593. https://doi.org/10.1093/nar/15.8.3581
Huang X-L, Fan Z, Borowski L, Rinaldo CR (2008) Maturation of dendritic cells for enhanced activation of anti-HIV-1 CD8+ T cell immunity. J Leukoc Biol 83:1530–1540. https://doi.org/10.1189/jlb.1107795
Ito T, Amakawa R, Inaba M, Hori T, Ota M, Nakamura K, Takebayashi M, Miyaji M, Yoshimura T, Inaba K, Fukuhara S (2004) Plasmacytoid dendritic cells regulate Th cell responses through OX40 ligand and type I IFNs. J Immunol 172:4253–4259. https://doi.org/10.4049/jimmunol.172.7.4253
Kaslow R, Ostrow D, Detels R, Phair J, Polk B, Rinaldo CJ (1987) The multicenter AIDS cohort study: rationale, organization, and selected characteristics of the participants. Am J Epidemiol 126:310–318
Kim HJ, Kim H-O, Lee K, Baek EJ, Kim H-S (2010) Two-step maturation of immature DCs with proinflammatory cytokine cocktail and poly(I:C) enhances migratory and T cell stimulatory capacity. Vaccine 28:2877–2886. https://doi.org/10.1016/j.vaccine.2010.01.061
Kourelis A, Kotzamanidis C, Litopoulou-Tzanetaki E, Papaconstantinou J, Tzanetakis N, Yiangou M (2010) Immunostimulatory activity of potential probiotic yeast strains in the dorsal air pouch system and the gut mucosa. J Appl Microbiol 109:260–271. https://doi.org/10.1111/j.1365-2672.2009.04651.x
Latorre-García L, Adam AC, Polaina J (2008) Overexpression of the glucoamylase-encoding STA1 gene of Saccharomyces cerevisiae var. diastaticus in laboratory and industrial strains of Saccharomyces. World J Microbiol Biotechnol 24:2957–2963
Michael S, Keubler LM, Smoczek A, Meier M, Gunzer F, Pöhlmann C, Krause-Buchholz U, Hedrich H-J, Bleich A (2013) Quantitative phenotyping of inflammatory bowel disease in the IL-10-deficient mouse by use of noninvasive magnetic resonance imaging. Inflamm Bowel Dis 19:185–193
Motta JM, Rumjanek VM (2016) Sensitivity of dendritic cells to microenvironment signals. J Immunol Res 2016:1–10. https://doi.org/10.1155/2016/4753607
Palma ML, Zamith-Miranda D, Martins FS, Bozza FA, Nimrichter L, Montero-Lomeli M, Marques ETA, Douradinha B (2015) Probiotic Saccharomyces cerevisiae strains as biotherapeutic tools: is there room for improvement? Appl Microbiol Biotechnol 99:6563–6570. https://doi.org/10.1007/s00253-015-6776-x
Pohlmann C, Thomas M, Forster S, Brandt M, Hartmann M, Bleich A, Gunzer F (2013) Use of engineered intestinal microorganisms as in situ cytokine delivery system. Bioengineered 4:1–8
Reis VCB, Nicola AM, de Souza Oliveira Neto O, Batista VDF, de Moraes LMP, Torres FAG (2012) Genetic characterization and construction of an auxotrophic strain of Saccharomyces cerevisiae JP1, a Brazilian industrial yeast strain for bioethanol production. J Ind Microbiol Biotechnol 39:1673–1683
Sakuragi S, Goto T, Sano K, Morikawa Y (2002) HIV type 1 Gag virus-like particle budding from spheroplasts of Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 99:7956–7961. https://doi.org/10.1073/pnas.082281199
Schreuder MP, Deen C, Boersma WJ, Pouwels PH, Klis FM (1996) Yeast expressing hepatitis B virus surface antigen determinants on its surface: implications for a possible oral vaccine. Vaccine 14:383–388
Shen L, Siliciano RF (2008) Viral reservoirs, residual viremia, and the potential of highly active antiretroviral therapy to eradicate HIV infection. J Allergy Clin Immunol 122:22–28. https://doi.org/10.1016/j.jaci.2008.05.033
Smith KN, Mailliard RB, Piazza PA, Fischer W, Korber BT, Fecek RJ, Ratner D, Gupta P, Mullins JI, Rinaldob CR (2016) Effective cytotoxic T lymphocyte targeting of persistent HIV-1 during antiretroviral therapy requires priming of naive CD8+ T cells. MBio 7:1–18. https://doi.org/10.1128/mBio.00473-16
Stephenson KE, D’Couto HT, Barouch DH (2016) New concepts in HIV-1 vaccine development. Curr Opin Immunol 41:39–46. https://doi.org/10.1016/j.coi.2016.05.011
Stubbs AC, Martin KS, Coeshott C, Skaates SV, Kuritzkes DR, Bellgrau D, Franzusoff A, Duke RC, Wilson CC (2001) Whole recombinant yeast vaccine activates dendritic cells and elicits protective cell-mediated immunity. Nat Med 7:625–629
Vickers CE, Bydder SF, Zhou Y, Nielsen LK (2013) Dual gene expression cassette vectors with antibiotic selection markers for engineering in Saccharomyces cerevisiae. Microb Cell Factories 12:96. https://doi.org/10.1186/1475-2859-12-96
Wang T, Sun H, Zhang J, Liu Q, Wang L, Chen P, Wang F, Li H, Xiao Y, Zhao X (2014) The establishment of Saccharomyces boulardii surface display system using a single expression vector. Fungal Genet Biol 64:1–10
World Health Organization (2016) HIV/AIDS fact sheet. http://www.who.int/mediacentre/factsheets/fs360/en/. Accessed 11 Oct 2016
Wyszyńska A, Kobierecka P, Bardowski J, Jagusztyn-Krynicka EK (2015) Lactic acid bacteria—20 years exploring their potential as live vectors for mucosal vaccination. Appl Microbiol Biotechnol 99:2967–2977. https://doi.org/10.1007/s00253-015-6498-0
Zamith-Miranda D, Palma ML, Matos GS, Schiebel J, Maya-Monteiro CM, Aronovich M, Bozza PT, Bozza FA, Nimrichter L, Montero-lomeli M, Marques ETA, Martins FS, Douradinha B (2016) Lipid droplet levels vary heterogeneously in response to simulated gastrointestinal stresses in different probiotic Saccharomyces cerevisiae strains. J Funct Foods 21:193–200
Acknowledgements
We thank Prof. Charles R. Rinaldo and Prof. Robbie B. Mailliard, both from Department of Infectious Diseases and Microbiology, University of Pittsburgh Graduate School of Public Health, for the cells of the HIV+ patients from MACS cohort as well as helpful scientific discussions.
Funding
This work was supported by internal funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human subject research was approved by the University of Pittsburgh Institutional Review Board.
Conflict of interest
Bruno Douradinha is an inventor of the patent WO2018091637, partially based in this work. The other authors declare that they have no conflict of interest.
Ethical statement
All procedures performed in studies involving human participants’ samples were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 737 kb)
Rights and permissions
About this article
Cite this article
Palma, M.L., Garcia-Bates, T.M., Martins, F.S. et al. Genetically engineered probiotic Saccharomyces cerevisiae strains mature human dendritic cells and stimulate Gag-specific memory CD8+ T cells ex vivo. Appl Microbiol Biotechnol 103, 5183–5192 (2019). https://doi.org/10.1007/s00253-019-09842-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09842-8