Abstract
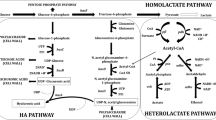
Hyaluronic acid (HA) is a biopolymer with wide biomedical and cosmetic applications, wherein the molecular weight of HA (MWHA) is an important quality parameter that determines its suitability for the targeted application. To produce HA with desired molecular weight, it is important to identify parameters that offer tunability and control of MWHA at a desired value during fermentation. In this work, two tunable parameters, viz. glucose concentration and combination of HA biosynthetic genes expressed, were used to produce HA of different molecular weights. Three recombinant strains of Lactococcus lactis were constructed, using a combination of the has-operon genes from Streptococcus zooepidemicus (hasA, hasB, hasE) and the α-phosphoglucomutase gene (pgmA) from L. lactis. Batch fermentations of these recombinant strains at different initial glucose concentrations enabled production of HA with different molecular weights. Co-expression of hasABE was observed to be particularly effective in improving the MWHA. It was observed during batch fermentations of all these recombinant L. lactis cultures that the MWHA decreases steadily during the later part of the fermentation and the final value is 19–43% lower than the peak MWHA produced. Analysis of the fermentation data showed that the decrease in MWHA correlated strongly with the decrease in specific productivity of the culture. To overcome this decrease in MWHA, a glucostat strategy was successfully devised which could maintain a high value of specific productivity throughout the glucostat phase and result in constant-MW HA production. Glucostat processes were designed with the three recombinant L. lactis strains at two different glucose concentrations to produce constant molecular weight HA ranging from 0.4 to 1.4 MDa. This is the first report of its kind in literature that demonstrates production of controlled MW HA over a wide range by using a combination of tunable parameters and suitable process control strategies.








Similar content being viewed by others
References
Armstrong DC, Johns MR (1997) Culture conditions affect the molecular weight properties of hyaluronic acid produced by Streptococcus zooepidemicus. Appl Environ Microbiol 63:2759–2764
Badle SS, Jayaraman G, Ramachandran KB (2014) Ratio of intracellular precursors concentration and their flux influences hyaluronic acid molecular weight in Streptococcus zooepidemicus and recombinant Lactococcus lactis. Bioresour Technol 163C:222–227. https://doi.org/10.1016/j.biortech.2014.04.027
Chauhan AS, Badle SS, Ramachandran KB, Jayaraman G (2014) The P170 expression system enhances hyaluronan molecular weight and production in metabolically-engineered Lactococcus lactis. Biochem Eng J 90:73–78. https://doi.org/10.1016/j.bej.2014.05.012
Chen WY, Marcellin E, Hung J, Nielsen LK (2009) Hyaluronan molecular weight is controlled by UDP-N-acetylglucosamine concentration in Streptococcus zooepidemicus. J Biol Chem 284:18007–18014. https://doi.org/10.1074/jbc.M109.011999
Chen WY, Marcellin E, Steen JA, Nielsen LK (2014) The role of hyaluronic acid precursor concentrations in molecular weight control in Streptococcus zooepidemicus. Mol Biotechnol 56:147–156. https://doi.org/10.1007/s12033-013-9690-4
Duan X-J, Yang L, Zhang X, Tan W-S (2008) Effect of oxygen and shear stress on molecular weight of hyaluronic acid. J Microbiol Biotechnol 18:718–724
Ercan O, Wels M, Smid EJ, Kleerebezem M (2015) Genome-Wide Transcriptional Responses to Carbon Starvation in Nongrowing Lactococcus lactis. Appl Environ Microbiol 81:2554–2561. https://doi.org/10.1128/AEM.03748-14
Hmar RV, Prasad SB, Jayaraman G, Ramachandran KB (2014) Chromosomal integration of hyaluronic acid synthesis (has) genes enhances the molecular weight of hyaluronan produced in Lactococcus lactis. Biotechnol J 9:1554–1564. https://doi.org/10.1002/biot.201400215
Im JH, Song JM, Kang JH, Kang DJ (2009) Optimization of medium components for high-molecular-weight hyaluronic acid production by Streptococcus sp. ID9102 via a statistical approach. J Ind Microbiol Biotechnol 36:1337–1344. https://doi.org/10.1007/s10295-009-0618-8
Jagannath S, Ramachandran KB (2010) Influence of competing metabolic processes on the molecular weight of hyaluronic acid synthesized by Streptococcus zooepidemicus. Biochem Eng J 48:148–158. https://doi.org/10.1016/j.bej.2009.09.003
Jin P, Kang Z, Yuan P, Du G, Chen J (2016) Production of specific-molecular-weight hyaluronan by metabolically engineered Bacillus subtilis 168. Metab Eng 35:21–30. https://doi.org/10.1016/j.ymben.2016.01.008
Jing W, DeAngelis PL (2004) Synchronized chemoenzymatic synthesis of monodisperse hyaluronan polymers. J Biol Chem 279:42345–42349. https://doi.org/10.1074/jbc.M402744200
Kaur M, Jayaraman G (2016) Hyaluronan production and molecular weight is enhanced in pathway-engineered strains of lactate dehydrogenase-deficient Lactococcus lactis. Metab Eng Commun 3:15–23. https://doi.org/10.1016/j.meteno.2016.01.003
Kim J-H, Yoo S-J, Oh D-K, Kweon Y-G, Park D-W, Lee C-H, Gil G-H (1996) Selection of a Streptococcus equi mutant and optimization of culture conditions for the production of high molecular weight hyaluronic acid. Enzym Microb Technol 19:440–445. https://doi.org/10.1016/S0141-0229(96)00019-1
Lai ZW, Rahim RA, Ariff AB, Mohamad R (2011) Medium formulation and impeller design on the biosynthesis of high molecular weight hyaluronic acid by Streptococcus zooepidemicus ATCC 39920. Afr J Microbiol Res 5:2114–2123. https://doi.org/10.5897/AJMR11.305
Marcellin E, Steen JA, Nielsen LK (2014) Insight into hyaluronic acid molecular weight control. Appl Microbiol Biotechnol 98:6947–6956. https://doi.org/10.1007/s00253-014-5853-x
Oueslati N, Leblanc P, Harscoat-schiavo C, Rondags E, Meunier S, Kapel R, Marc I (2014) CTAB turbidimetric method for assaying hyaluronic acid in complex environments and under cross-linked form. Carbohydr Polym 112:102–108. https://doi.org/10.1016/j.carbpol.2014.05.039
Papagianni M, Avramidis N, Filiousis G (2007) Glycolysis and the regulation of glucose transport in Lactococcus lactis spp. lactis in batch and fed-batch culture. Microb Cell Factories 6:16. https://doi.org/10.1186/1475-2859-6-16
Pires AMB, Santana MHA (2010) Metabolic effects of the initial glucose concentration on microbial production of hyaluronic acid. Appl Biochem Biotechnol 162:1751–1761. https://doi.org/10.1007/s12010-010-8956-6
Prasad SB, Jayaraman G, Ramachandran KB (2010) Hyaluronic acid production is enhanced by the additional co-expression of UDP-glucose pyrophosphorylase in Lactococcus lactis. Appl Microbiol Biotechnol 86:273–283. https://doi.org/10.1007/s00253-009-2293-0
Prasad SB, Ramachandran KB, Jayaraman G (2012) Transcription analysis of hyaluronan biosynthesis genes in Streptococcus zooepidemicus and metabolically engineered Lactococcus lactis. Appl Microbiol Biotechnol 94:1593–1607. https://doi.org/10.1007/s00253-012-3944-0
Redon E, Loubiere P, Cocaign-bousquet M (2005) Transcriptome Analysis of the Progressive Adaptation of Lactococcus lactis to Carbon Starvation. J Bacteriol 187:3589–3592. https://doi.org/10.1128/JB.187.10.3589
Shanmuga Doss S, Bhatt NP, Jayaraman G (2017) Improving the accuracy of hyaluronic acid molecular weight estimation by conventional size exclusion chromatography. J Chromatogr B Anal Technol Biomed Life Sci 1060:255–261. https://doi.org/10.1016/j.jchromb.2017.06.006
Sheng JZ, Ling PX, Zhu XQ, Guo XP, Zhang TM, He YL, Wang FS (2009) Use of induction promoters to regulate hyaluronan synthase and UDP-glucose-6-dehydrogenase of Streptococcus zooepidemicus expression in Lactococcus lactis: a case study of the regulation mechanism of hyaluronic acid polymer. J Appl Microbiol 107:136–144. https://doi.org/10.1111/j.1365-2672.2009.04185.x
Westbrook AW, Ren X, Perry MMC (2018) Engineering of cell membrane to enhance heterologous production of hyaluronic acid in Bacillus subtilis. Biotechnol Bioeng 115:216–231. https://doi.org/10.1002/bit.26459
Yu H, Stephanopoulos G (2008) Metabolic engineering of Escherichia coli for biosynthesis of hyaluronic acid. Metab Eng 10:24–32. https://doi.org/10.1016/j.ymben.2007.09.001
Acknowledgements
The authors would like to acknowledge Indian Institute of Technology Madras for providing infrastructure to carry out this work and Ministry of Human Resource Development, Government of India for providing Fellowships to Sreeja Shanmuga Doss and Pandeeswari Jeeva.
Funding
The Department of Biotechnology (Ministry of Science and Technology, Govt. of India) provided funds to execute this project through Project No. BT/IN/Finland/30/GJ/2013.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1188 kb)
Rights and permissions
About this article
Cite this article
Jeeva, P., Shanmuga Doss, S., Sundaram, V. et al. Production of controlled molecular weight hyaluronic acid by glucostat strategy using recombinant Lactococcus lactis cultures. Appl Microbiol Biotechnol 103, 4363–4375 (2019). https://doi.org/10.1007/s00253-019-09769-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09769-0