Abstract
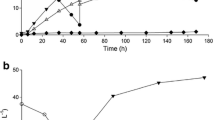
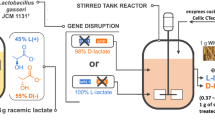
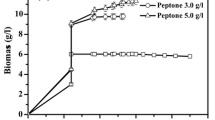
Simultaneous saccharification and fermentation (SSF) of d-lactic acid was performed using brown rice as both a substrate and a nutrient source. An engineered Lactobacillus plantarum NCIMB 8826 strain, in which the ʟ-lactate dehydrogenase gene was disrupted, produced 97.7 g/L d-lactic acid from 20% (w/v) brown rice without any nutrient supplementation. However, a significant amount of glucose remained unconsumed and the yield of lactic acid was as low as 0.75 (g/g-glucose contained in brown rice). Interestingly, the glucose consumption was significantly improved by adapting L. plantarum cells to the low-pH condition during the early stage of SSF (8–17 h). As a result, 117.1 g/L d-lactic acid was produced with a high yield of 0.93 and an optical purity of 99.6% after 144 h of fermentation. SSF experiments were repeatedly performed for ten times and d-lactic acid was stably produced using recycled cells (118.4–129.8 g/L). On average, d-lactic acid was produced with a volumetric productivity of 2.18 g/L/h over 48 h.





Similar content being viewed by others
References
Baldwin K, Childs N, Dyck J, Hansen J (2012) Southeast Asia’s rice surplus. A report from the economic research service. United States Department of Agriculture
Bruinenberg P, Vos P, de Vos WM (1992) Proteinase overproduction in Lactococcus lactis strains: regulation and effect on growth and acidification in milk. Appl Environ Microbiol 58:78–84
Council for Science and Technology (2015) Standard tables of food composition in Japan 7th ed. Ministry of Education, Culture, Sports, Science and Technology, Japan. National Printing Bureau, Tokyo, Japan
Dumbrepatil A, Adsul M, Chaudhari S, Khire J, Gokhale D (2008) Utilization of molasses sugar for lactic acid production by Lactobacillus delbrueckii subsp. delbrueckii mutant Uc-3 in batch fermentation. Appl Environ Microbiol 74:333–335
Fukushima K, Sogo K, Miura S, Kimura Y (2004) Production of d-lactic acid by bacterial fermentation of rice starch. Macromol Biosci 4:1021–1027
Gao MT, Kaneko M, Hirata M, Toorisaka E, Hano T (2008) Utilization of rice bran as nutrient source for fermentative lactic acid production. Bioresour Technol 99:3659–3664
Hama S, Mizuno S, Kihara M, Tanaka T, Ogino C, Noda H, Kondo A (2015) Production of d-lactic acid from hardwood pulp by mechanical milling followed by simultaneous saccharification and fermentation using metabolically engineered Lactobacillus plantarum. Bioresour Technol 187:167–172
Ikada Y, Jamshidi K, Tsuji H, Hyon SH (1987) Stereocomplex formation between enantiomeric poly (lactides). Macromolecules 20:904–906
Linko YY, Javanainen P (1996) Simultaneous liquefaction, saccharification, and lactic acid fermentation on barley starch. Enzym Microb Technol 19:118–123
Lunt J (1998) Large-scale production, properties and commercial applications of polylactic acid polymers. Polym Degrad Stabil 59:145–152
Oh H, Wee YJ, Yun JS, Han SH, Jung S, Ryu HW (2005) Lactic acid production from agricultural resources as cheap raw materials. Bioresour Technol 96:1492–1498
Ohara H, Owaki M, Sonomoto K (2007) Calculation of metabolic flow of xylose in Lactococcus lactis. J Biosci Bioeng 103:92–94
Okano K, Tanaka T, Ogino C, Fukuda H, Kondo A (2010) Biotechnological production of enantiomeric pure lactic acid from renewable resources: recent achievements, perspectives, and limit. Appl Microbiol Biotechnol 85:413–423
Okano K, Zhang Q, Shinkawa S, Yoshida S, Tanaka T, Fukuda H, Kondo A (2009) Efficient production of optically pure d-lactic acid from raw corn starch using genetically modified ʟ-lactate dehydrogenase gene-deficient and α-amylase-secreting Lactobacillus plantarum. Appl Environ Microbiol 75:462–467
Pavan S, Hols P, Delcour J, Geoffroy MC, Grangette C, Kleerebezem M, Mercenier A (2000) Adaptation of the nisin-controlled expression system in Lactobacillus plantarum: a tool to study in vivo biological effects. Appl Environ Microbiol 66:4427–4432
Rud I, Jensen PR, Naterstad K, Axelsson L (2006) A synthetic promoter library for constitutive gene expression in Lactobacillus plantarum. Microbiology 152:1011–1019
Saman P, Fuciños P, Vázquez JA, Pandiella SS (2011) Fermentability of brown rice and rice bran for growth of human Lactobacillus plantarum NCIMB 8826. Food Technol Biotechnol 49:128–132
Silva J, Carvalho AS, Ferreira R, Vitorino R, Amado F, Domingues P, Teixeira P, Gibbs PA (2005) Effect of the pH of growth on the survival of Lactobacillus delbrueckii subsp. bulgaricus to stress conditions during spray-drying. J Appl Microbiol 98:775–782
Siragusa S, De Angelis M, Calasso M, Campanella D, Minervini F, Di Cagno R, Gobbetti M (2014) Fermentation and proteome profiles of Lactobacillus plantarum strains during growth under food-like conditions. J Proteome 96:366–380
Tanaka T, Hoshina M, Tanabe S, Sakai K, Ohtsubo S, Taniguchi M (2006) Production of d-lactic acid from defatted rice bran by simultaneous saccharification and fermentation. Bioresour Technol 97:211–217
Wang Y, Yang Z, Qin P, Tan T (2014) Fermentative ʟ-(+)-lactic acid production from defatted rice bran. RSC Adv 4:8907–8913
Wee YJ, Kim JN, Ryu HW (2006) Biotechnological production of lactic acid and its recent applications. Food Technol Biotechnol 44:163–172
Wegkamp A, Teusink B, de Vos WM, Smid EJ (2010) Development of a minimal growth medium for Lactobacillus plantarum. Lett Appl Microbiol 50:57–64
Yang P, Wang J, Qi Q (2015) Prophage recombinases-mediated genome engineering in Lactobacillus plantarum. Microb Cell Factories 14:154
Yun JS, Wee YJ, Kim JN, Ryu HW (2004) Fermentative production of dʟ-lactic acid from amylase-treated rice and wheat brans hydrolyzate by a novel lactic acid bacterium, Lactobacillus sp. Biotechnol Lett 26:1613–1616
Zhang Y, Vadlani PV, Kumar A, Hardwidge PR, Govind R, Tanaka T, Kondo A (2016) Enhance d-lactic acid production from renewable resources using engineered Lactobacillus plantarum. Appl Microbiol Biotechnol 100:279–288
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was partly financed by the Strategic Foundational Technology Improvement Support Operation of the Kinki Bureau of Economy, Trade and Industry, Japan. We acknowledge Special Coordination Funds for Promoting Science and Technology, Creation of Innovative Centers for Advanced Interdisciplinary Research Areas (Innovative Bioproduction Kobe) from the Ministry of Education, Culture, Sports, Science and Technology, Japan. We also thank the Japan Society for the Promotion of Science (JSPS), KAKENHI Grant (16K18299).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Okano, K., Hama, S., Kihara, M. et al. Production of optically pure d-lactic acid from brown rice using metabolically engineered Lactobacillus plantarum . Appl Microbiol Biotechnol 101, 1869–1875 (2017). https://doi.org/10.1007/s00253-016-7976-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7976-8