Abstract
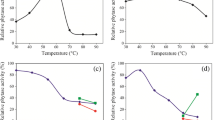
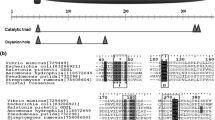
Phytases are enzymes degrading phytic acid and thereby releasing inorganic phosphate. While the phytases reported to date are majorly from culturable microorganisms, the fast-growing quantity of publicly available metagenomic data generated in the last decade has enabled bioinformatic mining of phytases in numerous data mines derived from a variety of ecosystems throughout the world. In this study, we are interested in the histidine acid phosphatase (HAP) family phytases present in insect-cultivated fungus gardens. Using bioinformatic approaches, 11 putative HAP phytase genes were initially screened from 18 publicly available metagenomes of fungus gardens and were further overexpressed in Escherichia coli. One phytase from a south pine beetle fungus garden showed the highest activity and was then chosen for further study. Biochemical characterization showed that the phytase is mesophilic but possesses strong ability to withstand high temperatures. To our knowledge, it has the longest half-life time at 100 °C (27 min) and at 80 °C (2.1 h) as compared to all the thermostable phytases publicly reported to date. After 100 °C incubation for 15 min, more than 93 % of the activity was retained. The activity was 3102 μmol P/min/mg at 37 °C and 4135 μmol P/min/mg at 52.5 °C, which is higher than all the known thermostable phytases. For the high activity level demonstrated at mesophilic temperatures as well as the high resilience to high temperatures, the phytase might be promising for potential application as an additive enzyme in animal feed.



Similar content being viewed by others
References
Adedokun SA, Owusu-Asiedu A, Ragland D, Plumstead P, Adeola O (2015) The efficacy of a new 6-phytase obtained from Buttiauxella spp. expressed in Trichoderma reesei on digestibility of amino acids, energy, and nutrients in pigs fed a diet based on corn, soybean meal, wheat middlings, and corn distillers’ dried grains with solubles. J Anim Sci 93:168–175. doi:10.2527/jas.2014-7912
Ariza A, Moroz OV, Blagova EV, Turkenburg JP, Waterman J, Roberts SM, Vind J, Sjøholm C, Lassen SF, De Maria L, Glitsoe V, Skov LK, Wilson KS (2013) Degradation of phytate by the 6-phytase from Hafnia alvei: a combined structural and solution study. PLoS One 8:e65062. doi:10.1371/journal.pone.0065062
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL Workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201. doi:10.1093/bioinformatics/bti770
Azeem M, Riaz A, Chaudhary AN, Hayat R, Hussain Q, Tahir MI, Imran M (2015) Microbial phytase activity and their role in organic P mineralization. Arch Agron Soil Sci 61:751–766. doi:10.1080/03650340.2014.963796
Böhm K, Herter T, Müller JJ, Borriss R, Heinemann U (2010) Crystal structure of Klebsiella sp. ASR1 phytase suggests substrate binding to a preformed active site that meets the requirements of a plant rhizosphere enzyme. FEBS J 277:1284–1296. doi:10.1111/j.1742-4658.2010.07559.x
Bharti RK, Srivastava S, Thakur IS (2014) Extraction of extracellular lipids from chemoautotrophic bacteria Serratia sp. ISTD04 for production of biodiesel. Bioresour Technol 165:201–204. doi:10.1016/j.biortech.2014.02.075
Biasini M, Bienert S, Waterhouse A, Arnold K, Studer G, Schmidt T, Kiefer F, Cassarino TG, Bertoni M, Bordoli L, Schwede T (2014) SWISS-MODEL: modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res 42:W252–W258. doi:10.1093/nar/gku340
Borgi M, Boudebbouze S, Aghajari N, Szukala F, Pons N, Maguin E, Rhimi M (2014) The attractive recombinant phytase from Bacillus licheniformis: biochemical and molecular characterization. Appl Microbiol Biotechnol 98:5937–5947. doi:10.1007/s00253-013-5421-9
Bradford MM (1976) A rapid and sensitive method for the quantization of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Brinch-Pedersen H, Madsen CK, Holme IB, Dionisio G (2014) Increased understanding of the cereal phytase complement for better mineral bio-availability and resource management. J Cereal Sci 59:373–381. doi:10.1016/j.jcs.2013.10.003
Choi JH, Lee SY (2004) Secretory and extracellular production of recombinant proteins using Escherichia coli. Appl Microbiol Biotechnol 64:625–635. doi:10.1007/s00253-004-1559-9
Cleto S, Van der Auwera G, Almeida C, Vieira MJ, Vlamakis H, Kolter R (2014) Genome sequence of Serratia plymuthica V4. Genome Announc 2:e00340–00314. doi:10.1128/genomeA.00340-14
Dono ND, Sparks NH, Olukosi OA (2014) Association between digesta pH, body weight, and nutrient utilization in chickens of different body weights and at different ages. J Poultry Sci 51:180–184. doi:10.2141/jpsa.0120151
Emanuelsson O, Brunak S, von Heijne G, Nielsen H (2007) Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc 2:953–971. doi:10.1038/nprot.2007.131
Fu D, Huang H, Luo H, Wang Y, Yang P, Meng K, Bai Y, Wu N, Yao B (2008) A highly pH-stable phytase from Yersinia kristeensenii: cloning, expression, and characterization. Enzyme Microb Technol 42:499–505. doi:10.1016/j.enzmictec.2008.01.014
Garrett JB, Kretz KA, O’Donoghue E, Kerovuo J, Kim W, Barton NR, Hazlewood GP, Short JM, Robertson DE, Gray KA (2004) Enhancing the thermal tolerance and gastric performance of a microbial phytase for use as a phosphate-mobilizing monogastric-feed supplement. Appl Environ Microbiol 70:3041–3046. doi:10.1128/aem.70.5.3041-3046.2004
Georgiou G, Segatori L (2005) Preparative expression of secreted proteins in bacteria: status report and future prospects. Curr Opin Biotechnol 16:538–545. doi:10.1016/j.copbio.2005.07.008
Graminho ER, Takaya N, Nakamura A, Hoshino T (2015) Purification, biochemical characterization, and genetic cloning of the phytase produced by Burkholderia sp. strain a13. J Gen Appl Microbiol 61:15–23. doi:10.2323/jgam.61.15
Greiner R, da Silva LG, Couri S (2009) Purification and characterisation of an extracellular phytase from Aspergillus niger 11T53A9. Braz J Microbiol 40:795–807. doi:10.1590/S1517-83822009000400010
Gu W-N, Huang H-Q, Yang P-L, Luo H-Y, Meng K, Wang Y-R, Yao B (2007) Gene cloning, expression and characterization of a novel phytase from Hafnia alvei. Chin J Biotechnol 23:1017–1021
Gu W, Huang H, Meng K, Yang P, Fu D, Luo H, Wang Y, Yao B, Zhan Z (2009) Gene cloning, expression, and characterization of a novel phytase from Dickeya paradisiaca. Appl Biochem Biotechnol 157:113–123. doi:10.1007/s12010-008-8329-6
Gulati HK, Chadha BS, Saini HS (2007a) Production and characterization of thermostable alkaline phytase from Bacillus laevolacticus isolated from rhizosphere soil. J Ind Microbiol Biotechnol 34:91–98. doi:10.1007/s10295-006-0171-7
Gulati HK, Chadha BS, Saini HS (2007b) Production, purification and characterization of thermostable phytase from thermophilic fungus Thermomyces lanuginosus TL-7. Acta Microbiol Immunol Hung 54:121–138. doi:10.1556/AMicr.54.2007.2.3
Guo B, Chen X-L, Sun C-Y, Zhou B-C, Zhang Y-Z (2009) Gene cloning, expression and characterization of a new cold-active and salt-tolerant endo-β-1,4-xylanase from marine Glaciecola mesophila KMM 241. Appl Microbiol Biotechnol 84:1107–1115. doi:10.1007/s00253-009-2056-y
Heinonen JK, Lahti RJ (1981) A new and convenient colorimetric determination of inorganic orthophosphate and its application to the assay of inorganic pyrophosphatase. Anal Biochem 113:313–317. doi:10.1016/0003-2697(81)90082-8
Hesampour A, Siadat SER, Malboobi MA, Mohandesi N, Arab SS, Ghahremanpour MM (2015) Enhancement of thermostability and kinetic efficiency of Aspergillus niger PhyA phytase by site-directed mutagenesis. Appl Biochem Biotechnol 175:2528–2541. doi:10.1007/s12010-014-1440-y
Hmida-Sayari A, Elgharbi F, Farhat A, Rekik H, Blondeau K, Bejar S (2014) Overexpression and biochemical characterization of a thermostable phytase from Bacillus subtilis US417 in Pichia pastoris. Mol Biotechnol 56:839–848. doi:10.1007/s12033-014-9764-y
Huang H, Luo H, Yang P, Meng K, Wang Y, Yuan T, Bai Y, Yao B (2006) A novel phytase with preferable characteristics from Yersinia intermedia. Biochem Biophys Res Commun 350:884–889. doi:10.1016/j.bbrc.2006.09.118
Huang H, Shao N, Wang Y, Luo H, Yang P, Zhou Z, Zhan Z, Yao B (2009a) A novel beta-propeller phytase from Pedobacter nyackensis MJ11 CGMCC 2503 with potential as an aquatic feed additive. Appl Microbiol Biotechnol 83:249–259. doi:10.1007/s00253-008-1835-1
Huang H, Shi P, Wang Y, Luo H, Shao N, Wang G, Yang P, Yao B (2009b) Diversity of beta-propeller phytase genes in the intestinal contents of grass carp provides insight into the release of major phosphorus from phytate in nature. Appl Environ Microbiol 75:1508–1516. doi:10.1128/AEM.02188-08
Huang H, Zhang R, Fu D, Luo J, Li Z, Luo H, Shi P, Yang P, Diao Q, Yao B (2011) Diversity, abundance and characterization of ruminal cysteine phytases suggest their important role in phytate degradation. Environ Microbiol 13:747–757. doi:10.1111/j.1462-2920.2010.02379.x
Huttenhower C, Hofmann O (2010) A quick guide to large-scale genomic data mining. PLoS Comp Biol 6:e1000779. doi:10.1371/journal.pcbi.1000779
Inoue H, Fujii T, Yoshimi M, Taylor LE II, Decker SR, Kishishita S, Nakabayashi M, Ishikawa K (2013) Construction of a starch-inducible homologous expression system to produce cellulolytic enzymes from Acremonium cellulolyticus. J Ind Microbiol Biotechnol 40:823–830. doi:10.1007/s10295-013-1286-2
Irving GCJ, Cosgrove DJ (1972) Inositol phosphate phosphatases of microbiological origin: the inositol pentaphosphate products of Aspergillus ficuum phytases. J Bacteriol 112:434–438
Jaenicke R, Bohm G (1998) The stability of proteins in extreme environments. Curr Opin Struct Biol 8:738–748. doi:10.1016/s0959-440x(98)80094-8
Jaenicke R, Schurig H, Beaucamp N, Ostendorp R (1996) Structure and stability of hyperstable proteins: glycolytic enzymes from hyperthermophilic bacterium Thermotoga maritima. Adv Protein Chem 48:181–269. doi:10.1016/S0065-3233(08)60363-0
Jermutus L, Tessier M, Pasamontes L, van Loon A, Lehmann M (2001) Structure-based chimeric enzymes as an alternative to directed enzyme evolution: phytase as a test case. J Biotechnol 85:15–24. doi:10.1016/s0168-1656(00)00373-4
Joyner J, Wanless D, Sinigalliano CD, Lipp EK (2014) Use of quantitative real-time PCR for direct detection of Serratia marcescens in marine and other aquatic environments. Appl Environ Microbiol 80:1679–1683. doi:10.1128/aem.02755-13
Katoh K, Toh H (2008) Recent developments in the MAFFT multiple sequence alignment program. Brief Bioinform 9:286–298. doi:10.1093/bib/bbn013
Kiefer F, Arnold K, Künzli M, Bordoli L, Schwede T (2009) The SWISS-MODEL Repository and associated resources. Nucleic Acids Res 37:D387–D392. doi:10.1093/nar/gkn750
Kim M-S, Lei X (2008) Enhancing thermostability of Escherichia coli phytase AppA2 by error-prone PCR. Appl Microbiol Biotechnol 79:69–75. doi:10.1007/s00253-008-1412-7
Kurze S, Bahl H, Dahl R, Berg G (2001) Biological control of fungal strawberry diseases by Serratia plymuthica HRO-C48. Plant Dis 85:529–534. doi:10.1094/pdis.2001.85.5.529
Lee GW, Kim S (2008) Genome data mining for everyone. BMB Rep 41:757–764. doi:10.5483/BMBRep.2008.41.11.757
Lee JK, Williams PD, Cheon S (2008) Data Mining in Genomics. Clin Lab Med 28:145–166. doi:10.1016/j.cll.2007.10.010
Lehmann M, Kostrewa D, Wyss M, Brugger R, D’Arcy A, Pasamontes L, van Loon APGM (2000) From DNA sequence to improved functionality: using protein sequence comparisons to rapidly design a thermostable consensus phytase. Protein Eng 13:49–57. doi:10.1093/protein/13.1.49
Lehmann M, Loch C, Middendorf A, Studer D, Lassen SF, Pasamontes L, Van Loon APGM, Wyss M (2002) The consensus concept for thermostability engineering of proteins: further proof of concept. Protein Eng 15:403–411. doi:10.1093/protein/15.5.403
Lei X-G, Porres JM (2003) Phytase enzymology, applications, and biotechnology. Biotechnol Lett 25:1787–1794. doi:10.1023/A:1026224101580
Lei X-G, Weaver JD, Mullaney EJ, Ullah AH, Azain MJ (2013) Phytase, a new life for an “old” enzyme. Annu Rev Anim Biosci 1:283–309. doi:10.1146/annurev-animal-031412-103717
Li X, Liu Z, Chi Z, Li J, Wang X (2009) Molecular cloning, characterization, and expression of the phytase gene from marine yeast Kodamaea ohmeri BG3. Mycol Res 113:24–32. doi:10.1016/j.mycres.2008.07.003
Liao Y, Li C-M, Chen H, Wu Q, Shan Z, Han X-Y (2013) Site-directed mutagenesis improves the thermostability and catalytic efficiency of Aspergillus niger N25 phytase mutated by I44E and T252R. Appl Biochem Biotechnol 171:900–915. doi:10.1007/s12010-013-0380-2
Lim BL, Yeung P, Cheng C, Hill JE (2007) Distribution and diversity of phytate-mineralizing bacteria. ISME J 1:321–330. doi:10.1038/ismej.2007.40
Lim D, Golovan S, Forsberg CW, Jia Z (2000) Crystal structures of Escherichia coli phytase and its complex with phytate. Nat Struct Biol 7:108–113. doi:10.1038/72371
Luo H, Huang H, Yang P, Wang Y, Yuan T, Wu N, Yao B, Fan Y (2007) A novel phytase appA from Citrobacter amalonaticus CGMCC 1696: gene cloning and overexpression in Pichia pastoris. Curr Microbiol 55:185–192. doi:10.1007/s00284-006-0586-4
Markowitz VM, Ivanova NN, Szeto E, Palaniappan K, Chu K, Dalevi D, Chen IMA, Grechkin Y, Dubchak I, Anderson I, Lykidis A, Mavromatis K, Hugenholtz P, Kyrpides NC (2008) IMG/M: a data management and analysis system for metagenomes. Nucleic Acids Res 36:D534–D538. doi:10.1093/nar/gkm869
Merchant HA, McConnell EL, Liu F, Ramaswamy C, Kulkarni RP, Basit AW, Murdan S (2011) Assessment of gastrointestinal pH, fluid and lymphoid tissue in the guinea pig, rabbit and pig, and implications for their use in drug development. Eur J Pharm Sci 42:3–10. doi:10.1016/j.ejps.2010.09.019
Moesseler A, Wintermann M, Sander SJ, Kamphues J (2012) Effect of diet grinding and pelleting fed either dry or liquid feed on dry matter and pH in the stomach of pigs and the development of gastric ulcers. J Anim Sci 90:343–345. doi:10.2527/jas.53772
Moraes VMB, Oviedo-Rondon EO, Leandro NSM, Wineland MJ, Malheiros RD, Eusebio-Balcazar P (2011) Broiler breeder trace mineral nutrition and feeding practices on embryo progeny development. Avian Biology Research 4:122–132. doi:10.3184/175815511x13147937995611
Mullaney EJ, Ullah AH (2003) The term phytase comprises several different classes of enzymes. Biochem Biophys Res Commun 312:179–184. doi:10.1016/j.bbrc.2003.09.176
Nakashima BA, McAllister TA, Sharma R, Selinger LB (2007) Diversity of phytases in the rumen. Microb Ecol 53:82–88. doi:10.1007/s00248-006-9147-4
Nam S-J, Kim Y-O, Ko T-K, Kang J-K, Chun K-H, Auh J-H, Lee I-K, Park S, Oh BC (2014) Molecular and biochemical characteristics of beta-propeller phytase from marine Pseudomonas sp. BS10-3 and its potential application for animal feed additives. J Microbiol Biotechnol 24:1413–1420. doi:10.4014/jmb.1407.07063
Nedeva R, Knev M (1993) A research of the effect of the combined feeds with different participation of calcium and phosphorus for growing pigs. Zhivotnov"dni Nauk 30:61–66
Pandee P, Summpunn P, Wiyakrutta S, Isarangkul D, Meevootisom V (2011) A thermostable phytase from Neosartorya spinosa BCC 41923 and its expression in Pichia pastoris. J Microbiol 49:257–264. doi:10.1007/s12275-011-0369-x
Patil KR, Haider P, Pope PB, Turnbaugh PJ, Morrison M, Scheffer T, McHardy AC (2011) Taxonomic metagenome sequence assignment with structured output models. Nat Methods 8:191–192. doi:10.1038/nmeth0311-191
Patil KR, Roune L, McHardy AC (2012) The PhyloPythiaS web server for taxonomic assignment of metagenome sequences. PLoS One 7:e38581. doi:10.1371/journal.pone.0038581
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. doi:10.1038/nmeth.1701
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF Chimera--a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612. doi:10.1002/jcc.20084
Reddy CS, Achary VMM, Manna M, Singh J, Kaul T, Reddy MK (2015) Isolation and molecular characterization of thermostable phytase from Bacillus subtilis (BSPhyARRMK33). Appl Biochem Biotechnol 175:3058–3067. doi:10.1007/s12010-015-1487-4
Sajidan A, Farouk A, Greiner R, Jungblut P, Müller E-C, Borriss R (2004) Molecular and physiological characterisation of a 3-phytase from soil bacterium Klebsiella sp. ASR1. Appl Microbiol Biotechnol 65:110–118. doi:10.1007/s00253-003-1530-1
Sanchez-Romero I, Ariza A, Wilson KS, Skjøt M, Vind J, De Maria L, Skov LK, Sanchez-Ruiz JM (2013) Mechanism of protein kinetic stabilization by engineered disulfide crosslinks. PLoS One 8:e70013. doi:10.1371/journal.pone.0070013
Sassi P, Perticaroli S, Comez L, Lupi L, Paolantoni M, Fioretto D, Morresi A (2012) Reversible and irreversible denaturation processes in globular proteins: from collective to molecular spectroscopic analysis. J Raman Spectrosc 43:273–279. doi:10.1002/jrs.3013
Selle PH, Ravindran V (2008) Phytate-degrading enzymes in pig nutrition. Livestock Sci 113:99–122. doi:10.1016/j.livsci.2007.05.014
Shao N, Huang H, Meng K, Luo H, Wang Y, Yang P, Yao B (2008) Cloning, expression, and characterization of a new phytase from the phytopathogenic bacterium Pectobacterium wasabiae DSMZ 18074. J Microbiol Biotechnol 18:1221–1226
Shi P, Huang H, Wang Y, Luo H, Wu B, Meng K, Yang P, Yao B (2008) A novel phytase gene appA from Buttiauxella sp. GC21 isolated from grass carp intestine. Aquaculture 275:70–75. doi:10.1016/j.aquaculture.2008.01.021
Shivange AV, Dennig A, Schwaneberg U (2014) Multi-site saturation by OmniChange yields a pH- and thermally improved phytase. J Biotechnol 170:68–72. doi:10.1016/j.jbiotec.2013.11.014
Shivange AV, Serwe A, Dennig A, Roccatano D, Haefner S, Schwaneberg U (2012) Directed evolution of a highly active Yersinia mollaretii phytase. Appl Microbiol Biotechnol 95:405–418. doi:10.1007/s00253-011-3756-7
Singh B, Satyanarayana T (2009) Characterization of a HAP-phytase from a thermophilic mould Sporotrichum thermophile. Bioresour Technol 100:2046–2051. doi:10.1016/j.biortech.2008.10.025
Slominski BA (2011) Recent advances in research on enzymes for poultry diets. Poultry Sci 90:2013–2023. doi:10.3382/ps.2011-01372
Sone M, Kishigami S, Yoshihisa T, Ito K (1997) Roles of disulfide bonds in bacterial alkaline phosphatase. J Biol Chem 272:6174–6178. doi:10.1074/jbc.272.10.6174
Tan H, Barret M, Mooij MJ, Rice O, Morrissey JP, Dobson AD, Griffiths BS, O’Gara F (2013) Long-term phosphorus fertilisation increased the diversity of the total bacterial community and the phoD phosphorus mineraliser group in pasture soils. Biol Fertil Soils 49:661–672. doi:10.1007/s00374-012-0755-5
Tian YS, Peng RH, Xu J, Zhao W, Gao F, Fu XY, Xiong AS, Yao QH (2011) Semi-rational site-directed mutagenesis of phyI1s from Aspergillus niger 113 at two residue to improve its phytase activity. Mol Biol Rep 38:977–982. doi:10.1007/s11033-010-0192-1
Tigerstrom A, Schwarz F, Karlsson G, Okvist M, Alvarez-Rua C, Maeder D, Robb FT, Sjolin L (2004) Effects of a novel disulfide bond and engineered electrostatic interactions on the thermostability of azurin. Biochemistry 43:12563–12574. doi:10.1021/bi048926x
Tran TT, Mamo G, Mattiasson B, Hatti-Kaul R (2010) A thermostable phytase from Bacillus sp. MD2: cloning, expression and high-level production in Escherichia coli. J Ind Microbiol Biotechnol 37:279–287. doi:10.1007/s10295-009-0671-3
Tye AJ, Siu FKY, Leung TYC, Lim BL (2002) Molecular cloning and the biochemical characterization of two novel phytases from B. subtilis 168 and B. licheniformis. Appl Microbiol Biotechnol 59:190–197. doi:10.1007/s00253-002-1033-5
Van Etten RL, Davidson R, Stevis PE, MacArthur H, Moore DL (1991) Covalent structure, disulfide bonding, and identification of reactive surface and active site residues of human prostatic acid phosphatase. J Biol Chem 266:2313–2319
Van Houdt R, Van der Lelie D, Izquierdo JA, Aertsen A, Masschelein J, Lavigne R, Michiels CW, Taghavi S (2014) Genome sequence of Serratia plymuthica RVH1, isolated from a raw vegetable-processing line. Genome Announc 2. doi:10.1128/genomeA.00021-14
Vats P, Banerjee UC (2005) Biochemical characterisation of extracellular phytase (myo-inositol hexakisphosphate phosphohydrolase) from a hyper-producing strain of Aspergillus niger van Teighem. J Ind Microbiol Biotechnol 32:141–147. doi:10.1007/s10295-005-0214-5
Vlassi M, Cesareni G, Kokkinidis M (1999) A correlation between the loss of hydrophobic core packing interactions and protein stability. J Mol Biol 285:817–827. doi:10.1006/jmbi.1998.2342
Wu T-H, Chen C-C, Cheng Y-S, Ko T-P, Lin C-Y, Lai H-L, Huang T-Y, Liu J-R, Guo R-T (2014) Improving specific activity and thermostability of Escherichia coli phytase by structure-based rational design. J Biotechnol 175:1–6. doi:10.1016/j.jbiotec.2014.01.034
Xiang T, Liu Q, Deacon AM, Koshy M, Kriksunov IA, Lei XG, Hao Q, Thiel DJ (2004) Crystal structure of a heat-resilient phytase from Aspergillus fumigatus, carrying a phosphorylated histidine. J Mol Biol 339:437–445. doi:10.1016/s0022-2836(04)00372-9
Yao MZ, Wang X, Wang W, Fu YJ, Liang AH (2013) Improving the thermostability of Escherichia coli phytase, appA, by enhancement of glycosylation. Biotechnol Lett 35:1669–1676. doi:10.1007/s10529-013-1255-x
Yao MZ, Zhang YH, Lu WL, Hu MQ, Wang W, Liang AH (2012) Phytases: crystal structures, protein engineering and potential biotechnological applications. J Appl Microbiol 112:1–14. doi:10.1111/j.1365-2672.2011.05181.x
Yu P, Chen Y (2013) Purification and characterization of a novel neutral and heat-tolerant phytase from a newly isolated strain Bacillus nealsonii ZJ0702. BMC Biotechnol 13:78–84. doi:10.1186/1472-6750-13-78
Zhang GQ, Dong XF, Wang ZH, Zhang Q, Wang HX, Tong JM (2010) Purification, characterization, and cloning of a novel phytase with low pH optimum and strong proteolysis resistance from Aspergillus ficuum NTG-23. Bioresour Technol 101:4125–4131. doi:10.1016/j.biortech.2010.01.001
Zhang LH, An LJ, Gao XR, Wang YJ (2005) Properties of A. ficuum AS3.324 phytase expressed in tobacco. Process Biochem 40:213–216. doi:10.1016/j.procbio.2003.12.005
Zhang R, Yang P, Huang H, Yuan T, Shi P, Meng K, Yao B (2011) Molecular and biochemical characterization of a new alkaline β-propeller phytase from the insect symbiotic bacterium Janthinobacterium sp. TN115. Appl Microbiol Biotechnol 92:317–325. doi:10.1007/s00253-011-3309-0
Zhang WM, Mullaney EJ, Lei XG (2007) Adopting selected hydrogen bonding and ionic interactions from Aspergillus fumigatus phytase structure improves the thermostability of Aspergillus niger PhyA phytase. Appl Environ Microbiol 73:3069–3076. doi:10.1128/aem.02970-06
Zhao QQ, Liu HL, Zhang Y, Zhang YZ (2010) Engineering of protease-resistant phytase from Penicillium sp.: High thermal stability, low optimal temperature and pH. J Biosci Bioeng 110:638–645. doi:10.1016/j.jbiosc.2010.08.003
Zhu WH, Qiao DR, Huang M, Yang G, Xu H, Cao Y (2010) Modifying thermostability of AppA from Escherichia coli. Curr Microbiol 61:267–273. doi:10.1007/s00284-010-9606-5
Acknowledgments
This research was supported by the following grants: the Special Fund for Strategic Emerging Industry Development of Sichuan Province (SC2013510104007) and the SAAS Young Scientists Fund (2015QNJJ-015).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Statement
This article does not contain any studies with human participants or animals performed by any of the authors. This is the original work of the authors. The work described has not been submitted elsewhere for publication, in whole or in part. All authors confirm that ethical principles have been followed in the research as well as in manuscript preparation, and approved this submission.
Conflict of interest
All the authors (Hao Tan, Xiang Wu, Liyuan Xie, Zhongqian Huang, Weihong Peng, Bingcheng Gan) declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 420 kb)
Rights and permissions
About this article
Cite this article
Tan, H., Wu, X., Xie, L. et al. Identification and characterization of a mesophilic phytase highly resilient to high-temperatures from a fungus-garden associated metagenome. Appl Microbiol Biotechnol 100, 2225–2241 (2016). https://doi.org/10.1007/s00253-015-7097-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-7097-9