Abstract
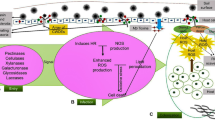
On the north regions of Portugal and Spain, the Castanea sativa Mill. culture is extremely important. The biggest productivity and yield break occurs due to the ink disease, the causal agent being the oomycete Phytophthora cinnamomi. This oomycete is also responsible for the decline of many other plant species in Europe and worldwide. P. cinnamomi and Phytophthora cambivora are considered, by the generality of the authors, as the C. sativa ink disease causal agents. Most Phytophthora species secrete large amounts of elicitins, a group of unique highly conserved proteins that are able to induce hypersensitive response (HR) and enhances plant defense responses in a systemic acquired resistance (SAR) manner against infection by different pathogens. Some other proteins involved in mechanisms of infection by P. cinnamomi were identified by our group: endo-1,3-beta-glucanase (complete cds); exo-glucanase (partial cds) responsible by adhesion, penetration, and colonization of host tissues; glucanase inhibitor protein (GIP) (complete cds) responsible by the suppression of host defense responses; necrosis-inducing Phytophthora protein 1 (NPP1) (partial cds); and transglutaminase (partial cds) which inducts defense responses and disease-like symptoms. In this mini-review, we present some scientifically advanced solutions that can contribute to the resolution of ink disease.

Similar content being viewed by others
References
Aryantha IP, Guest DI (2006) Mycoparasitic and antagonistic inhibition on Phytophthora cinnamomi rands by microbial agents isolated from manure composts. Plant Pathol J 5:291–298
Aryantha IP, Cross R, Guest DI (2000) Suppression of Phytophthora cinnamomi in potting mixes amended with uncomposted and composted animal manures. Phytopathology 90:775–782
Bishop JG, Ripoll DR, Bashir S, Damasceno CM, Seeds JD, Rose JK (2005) Selection on glycine beta-1,3-endoglucanase genes differentially inhibited by a Phytophthora glucanase inhibitor protein. Genetics 169:1009–1019
Damasceno CM, Bishop JG, Ripoll DR, Win J, Kamoun S, Rose JK (2008) Structure of the glucanase inhibitor protein (GIP) family from Phytophthora species suggests coevolution with plant endo-beta-1,3-glucanases. Mol Plant Microbe Interact 21:820–830
Day B, Graham T (2007) The plant host pathogen interface: cell wall and membrane dynamics of pathogen-induced responses. Ann N Y Acad Sci 1113:123–134
de Wit PJ (2007) How plants recognize pathogens and defend themselves. Cell Mol Life Sci 64:2726–2732
Duan CG, Wang CH, Guo HS (2012) Application of RNA silencing to plant disease resistance. Silence 3:5
Duclos J, Fauconnier A, Coelho AC, Bollen A, Cravador A, Godfroid E (1998) Identification of an elicitin gene cluster in Phytophthora cinnamomi. DNA Seq 9:231–237
Erwin DC, Ribeiro OK (1996) Phytophthora diseases worldwide. American Phytopathological Society, St. Paul
Furones-Pérez P, Fernández-López J (2009) Morphological and phenological description of 38 sweet chestnut cultivars (Castanea sativa Miller) in a contemporary collection. Span J Agric Res 7:829–843
Gijzen M, Nurnberger T (2006) Nep1-like proteins from plant pathogens: recruitment and diversification of the NPP1 domain across taxa. Phytochemistry 67:1800–1807
Hardham AR (2005) Phytophthora cinnamomi. Mol Plant Pathol 6:589–604
Hoopen GM, Rees R, Aisa P, Stirrup T, Krauss U (2003) Population dynamics of epiphytic mycoparasites of the genera Clonostachys and Fusarium for the biocontrol of black pod (Phytophthora palmivora) and moniliasis (Moniliophthora roreri) on cocoa (Theobroma cacao). Mycol Res 107:587–596
Horta M, Sousa N, Coelho AC, Neves D, Cravador A (2009) In vitro and in vivo quantification of elicitin expression in Phytophthora cinnamomi. Physiol Mol Plant Pathol 73:48–57
Judelson HS, Blanco FA (2005) The spores of Phytophthora: weapons of the plant destroyer. Nat Rev Microbiol 3:47–58
Kamoun S (2003) Molecular genetics of pathogenic oomycetes. Eukaryot Cell 2:191–199
Kamoun S (2007) Groovy times: filamentous pathogen effectors revealed. Curr Opin Plant Biol 10:358–365
Lourenço V, Maffia LA, Romeiro RS, Mizubuti E (2004) Isolation and selection of antagonists to Phytophthora infestans on tomato. Phytopathology 94:No. 6
Martins IM, Martins F, Belo H, Vaz M, Carvalho M, Cravador A, Choupina A (2014) Cloning, characterization and in vitro and in planta expression of a glucanase inhibitor protein (GIP) of Phytophthora cinnamomi. Mol Biol Rep (in press)
McLeod A, Smart CD, Fry WE (2003) Characterization of 1,3-beta-glucanase and 1,3;1,4-beta-glucanase genes from Phytophthora infestans. Fungal Genet Biol 38:250–263
Meirinho S, Carvalho M, Domínguez A, Choupina A (2010) Isolation and characterization by asymmetric PCR of the ENDO1 gene for glucan endo-1,3-B-D-glucosidase in Phytophthora cinnamomi associated with the ink disease of Castanea sativa Mill. Braz Arch Biol Technol 53:513–518
Moreira AC, Martins JM (2005) Influence of site factors on the impact of Phytophthora cinnamomi in cork oak stands in Portugal. For Pathol 35:145–162
Nuñez-Zofío M, Larregla del Palacio S, Garbisu C (2012) Repeated biodisinfection controls the incidence of Phytophthora root and crown rot of pepper while improving soil quality. Span J Agric Res 10:794–805
Okamoto H, Sato M, Miyata Y, Yoshikawa M, Isaka M (2000) Biocontrol of Phytophthora root rot of Angelica trees by Enterobacter cloacae and Serratia ficaria strains. J General Plant Pathol 66:86–94
Oliva R, Win J, Raffaele S, Boutemy L, Bozkurt TO, Chaparro-Garcia A, Segretin ME, Stam R, Schornack S, Cano LM, van Damme M, Huitema E, Thines M, Banfield MJ, Kamoun S (2010) Recent developments in effector biology of filamentous plant pathogens. Cell Microbiol 12:705–715
Rajkumar M, Lee WH, Lee KJ (2005) Screening of bacterial antagonists for biological control of Phytophthora blight of pepper. J Basic Microbiol 45:55–63
Rose JK, Ham KS, Darvill AG, Albersheim P (2002) Molecular cloning and characterization of glucanase inhibitor proteins: coevolution of a counterdefense mechanism by plant pathogens. Plant Cell 14:1329–1345
Schornack S, Huitema E, Cano LM, Bozkurt TO, Oliva R, Van Damme M, Schwizer S, Raffaele S, Chaparro-Garcia A, Farrer R, Segretin ME, Bos J, Haas BJ, Zody MC, Nusbaum C, Win J, Thines M, Kamoun S (2009) Ten things to know about oomycete effectors. Mol Plant Pathol 10:795–803
Sid A, Ezziyyani M, Egea-Gilabert C, Candela ME (2003) Selecting bacterial strains for use in the biocontrol of diseases caused by Phytophthora capsid and Alternaria alternata in sweet pepper plants. BioI Plant 47:569–574
Szittya G, Silhavy D, Molnar A, Havelda Z, Lovas A, Lakatos L, Banfalvi Z, Burgyan J (2003) Low temperature inhibits RNA silencing-mediated defence by the control of siRNA generation. EMBO J 22:633–640
Valueva TA, Mosolov VV (2004) Role of inhibitors of proteolytic enzymes in plant defense against phytopathogenic microorganisms. Biochemistry (Mosc) 69:1305–1309
Whisson SC, Avrova AO, Van West P, Jones JT (2005) A method for double-stranded RNA-mediated transient gene silencing in Phytophthora infestans. Mol Plant Pathol 6:153–63
York WS, Qin Q, Rose JK (2004) Proteinaceous inhibitors of endo-beta-glucanases. Biochim Biophys Acta 1696:223–233
Yu YJ, Wu SC, Chan HH, Chen YC, Chen ZY, Yang MT (2008) Overproduction of soluble recombinant transglutaminase from Streptomyces netropsis in Escherichia coli. Appl Microbiol Biotechnol 81:523–532
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choupina, A.B., Estevinho, L. & Martins, I.M. Scientifically advanced solutions for chestnut ink disease. Appl Microbiol Biotechnol 98, 3905–3909 (2014). https://doi.org/10.1007/s00253-014-5654-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5654-2