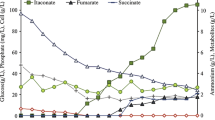
Abstract
To investigate the molecular mechanisms that interplay between oxygen metabolism and secondary metabolism in Streptomyces natalensis, we compared the transcriptomes of the strains CAM.02 (ΔsodF), pimaricin under-producer phenotype, and CAM.04 (ΔahpCD), pimaricin over-producer phenotype, with that of the wild type at late exponential and stationary growth phases. Microarray data interpretation was supported by characterization of the mutant strains regarding enzymatic activities, phosphate uptake, oxygen consumption and pimaricin production.
Both mutant strains presented a delay in the transcription activation of the PhoRP system and pimaricin biosynthetic gene cluster that correlated with the delayed inorganic phosphate (Pi) depletion in the medium and late onset of pimaricin production, respectively. The carbon flux of both mutants was also altered: a re-direction from glycolysis to the pentose phosphate pathway (PPP) in early exponential phase followed by a transcriptional activation of both pathways in subsequent growth phases was observed. Mutant behavior diverged at the respiratory chain/tricarboxylic acid cycle (TCA) and the branched chain amino acid (BCAA) metabolism. CAM.02 (ΔsodF) presented an impaired TCA cycle and an inhibition of the BCAA biosynthesis and degradation pathways. Conversely, CAM.04 (ΔahpCD) presented a global activation of BCAA metabolism.
The results highlight the cellular NADPH/NADH ratio and the availability of biosynthetic precursors via the BCAA metabolism as the main pimaricin biosynthetic bottlenecks under oxidative stress conditions. Furthermore, new evidences are provided regarding a crosstalk between phosphate metabolism and oxidative stress in Streptomyces.





Similar content being viewed by others
References
Ahn B-E, Cha J, Lee E-J, Han A-R, Thompson CJ, Roe J-H (2006) Nur, a nickel-responsive regulator of the Fur family, regulates superoxide dismutases and nickel transport in Streptomyces coelicolor. Mol Microbiol 59(6):1848–1858. doi:10.1111/j.1365-2958.2006.05065.x
Antón N, Mendes MV, Martín JF, Aparicio JF (2004) Identification of PimR as a positive regulator of pimaricin biosynthesis in Streptomyces natalensis. J Bacteriol 186(9):2567–2575. doi:10.1128/JB.186.9.2567-2575.2004
Antón N, Santos-Aberturas J, Mendes MV, Guerra SM, Martín JF, Aparicio JF (2007) PimM, a PAS domain positive regulator of pimaricin biosynthesis in Streptomyces natalensis. Microbiology 153(Pt 9):3174–3183. doi:10.1099/mic.0.2007/009126-0
Aparicio JF, Fouces R, Mendes MV, Olivera N, Martín JF (2000) A complex multienzyme system encoded by five polyketide synthase genes is involved in the biosynthesis of the 26-membered polyene macrolide pimaricin in Streptomyces natalensis. Chem Biol 7(11):895–905. doi:10.1016/S1074-5521(00)00038-7
Aparicio JF, Mendes MV, Antón N, Recio E, Martín JF (2004) Polyene macrolide antibiotic biosynthesis. Curr Med Chem 11(12):1645–1656
Armstrong DA, Buchanan JD (1978) Reactions of O-2, H2O2 and other oxidants with sulfhydryl enzymes. Photochem Photobiol 28(4–5):743–754. doi:10.1111/j.1751-1097.1978.tb07011.x
Beites T, Pires SD, Santos CL, Osorio H, Moradas-Ferreira P, Mendes MV (2011) Crosstalk between ROS homeostasis and secondary metabolism in S. natalensis ATCC 27448: modulation of pimaricin production by intracellular ROS. PLoS One 6(11):e27472
Berdy J (2005) Bioactive microbial metabolites. J Antibiot (Tokyo) 58(1):1–26. doi:10.1038/ja.2005.1
Bibb MJ (2005) Regulation of secondary metabolism in streptomycetes. Curr Opin Microbiol 8:208–215. doi:10.1016/j.mib.2005.02.016
Borodina I, Siebring J, Zhang J, Smith CP, van Keulen G, Dijkhuizen L, Nielsen J (2008) Antibiotic overproduction in Streptomyces coelicolor A3(2) mediated by phosphofructokinase deletion. J Biol Chem 283(37):25186–25199. doi:10.1074/jbc.M803105200
Brekasis D, Paget MSB (2003) A novel sensor of NADH/NAD(+) redox poise in Streptomyces coelicolor A3(2). EMBO J 22(18):4856–4865. doi:10.1093/emboj/cdg453
Darbon E, Martel C, Nowacka A, Pegot S, Moreau P, Virolle M-J (2012) Transcriptional and preliminary functional analysis of the six genes located in divergence of phoR phoP in Streptomyces lividans. Appl Microbiol Biotechnol 95(6):1553–1566. doi:10.1007/s00253-012-3995-2
Denoya CD, Fedechko RW, Hafner EW, McArthur HA, Morgenstern MR, Skinner DD, Stutzman-Engwall K, Wax RG, Wernau WC (1995) A second branched-chain alpha-keto acid dehydrogenase gene cluster (bkdFGH) from Streptomyces avermitilis: its relationship to avermectin biosynthesis and the construction of a bkdF mutant suitable for the production of novel antiparasitic avermectins. J Bacteriol 177(12):3504–3511
Edgar R, Domrachev M, Lash AE (2002) Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30(1):207–210. doi:10.1093/nar/30.1.207
el-Enshasy HA, Farid MA, el SA E-S (2000) Influence of inoculum type and cultivation conditions on natamycin production by Streptomyces natalensis. J Basic Microbiol 40(5–6):333–342. doi:10.1002/1521-4028(200012)40:5/6<333::AID-JOBM333>3.0.CO;2-Q
Flint DH, Tuminello JF, Emptage MH (1993) The inactivation of Fe–S cluster containing hydro-lyases by superoxide. J Biol Chem 268(30):22369–22376
Gadgil M, Lian W, Gadgil C, Kapur V, Hu WS (2005) An analysis of the use of genomic DNA as a universal reference in two channel DNA microarrays. BMC Genomics 6:66. doi:10.1186/1471-2164-6-66
Ghorbel S, Kormanec J, Artus A, Virolle M-J (2006) Transcriptional studies and regulatory interactions between the phoR–phoP operon and the phoU, mtpA, and ppk genes of Streptomyces lividans TK24. J Bacteriol 188(2):677–686. doi:10.1128/jb.188.2.677-686.2006
Gort AS, Imlay JA (1998) Balance between endogenous superoxide stress and antioxidant defenses. J Bacteriol 180(6):1402–1410
Hahn JS, Oh SY, Chater KF, Cho YH, Roe JH (2000) H2O2-sensitive fur-like repressor CatR regulating the major catalase gene in Streptomyces coelicolor. J Biol Chem 275(49):38254–38260. doi:10.1074/jbc.M006079200
Hahn JS, Oh SY, Roe JH (2002) Role of OxyR as a peroxide-sensing positive regulator in Streptomyces coelicolor A3(2). J Bacteriol 184(19):5214–5222. doi:10.1128/JB.184.19.5214-5222.2002
Higgens CE, Hamill RL, Sands TH, Hoehn MM, Davis NE (1974) Letter: the occurrence of deacetoxycephalosporin C in fungi and streptomycetes. J Antibiot 27(4):298–300
Holland MJ, Westhead EW (1973) Chemical reactivity at the catalytic sites of aspartic β-semialdehyde dehydrogenase and glyceraldehyde 3-phosphate dehydrogenase. Biochemistry 12(12):2276–2281. doi:10.1021/bi00736a015
Imlay JA (2008) Cellular defenses against superoxide and hydrogen peroxide. Annu Rev Biochem 77(1):755–776. doi:10.1146/annurev.biochem.77.061606.161055
Kallifidas D, Thomas D, Doughty P, Paget MSB (2010) The σR regulon of Streptomyces coelicolor A3(2) reveals a key role in protein quality control during disulphide stress. Microbiology 156(6):1661–1672. doi:10.1099/mic.0.037804-0
Kieser T, Bibb M, Buttner M, Chater K, Hopwood DA (2000) Practical Streptomyces genetics. John Innes Centre, Norwich, United Kingdom
Kwon HJ, Kim SU (1998) Enhanced biosynthesis of clavulanic acid in Streptomyces clavuligerus due to oxidative challenge by redox-cycling agents. Appl Microbiol Biotechnol 49(1):77–83
Lanzetta PA, Alvarez LJ, Reinach PS, Candia OA (1979) An improved assay for nanomole amounts of inorganic phosphate. Anal Biochem 100(1):95–97. doi:10.1016/0003-2697(79)90115-5
Li Q, Harvey LM, McNeil B (2009) Oxidative stress in industrial fungi. Crit Rev Biotechnol 29(3):199–213. doi:10.1080/07388550903004795
Lounès A, Lebrihi A, Benslimane C, Lefebvre G, Germain P (1995) Glycerol effect on spiramycin production and valine catabolism in Streptomyces ambofaciens. Curr Microbiol 31(5):304–311. doi:10.1007/bf00314585
Martín J, Santos-Beneit F, Rodríguez-García A, Sola-Landa A, Smith M, Ellingsen T, Nieselt K, Burroughs N, Wellington E (2012) Transcriptomic studies of phosphate control of primary and secondary metabolism in Streptomyces coelicolor. Appl Microbiol Biotechnol 95(1):61–75. doi:10.1007/s00253-012-4129-6
Mendes MV, Recio E, Anón N, Guerra SM, Santos-Aberturas J, Martín JF, Aparicio JF (2007a) Cholesterol oxidases act as signaling proteins for the biosynthesis of the polyene macrolide pimaricin. Chem Biol 14(3):279–90. doi:10.1016/j.chembiol.2007.01.010
Mendes MV, Recio E, Fouces R, Luiten R, Martín JF, Aparicio JF (2001) Engineered biosynthesis of novel polyenes: a pimaricin derivative produced by targeted gene disruption in Streptomyces natalensis. Chem Biol 8(7):635–44. doi:10.1016/S1074-5521(01)00033-3
Mendes MV, Tunca S, Antón N, Recio E, Sola-Landa A, Aparicio JF, Martín JF (2007b) The two-component phoR–phoP system of Streptomyces natalensis: Inactivation or deletion of phoP reduces the negative phosphate regulation of pimaricin biosynthesis. Metab Eng 9(2):217–27. doi:10.1016/j.ymben.2006.10.003
Muschko K, Kienzlen G, Fiedler H-P, Wohlleben W, Schwartz D (2002) Tricarboxylic acid cycle aconitase activity during the life cycle of Streptomyces viridochromogenes Tü494. Arch Microbiol 178(6):499–505. doi:10.1007/s00203-002-0483-6
Olmos E, Mehmood N, Haj Husein L, Goergen JL, Fick M, Delaunay S (2013) Effects of bioreactor hydrodynamics on the physiology of Streptomyces. Bioprocess Biosystems Eng 36(3):259–72. doi:10.1007/s00449-012-0794-1
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9). doi:10.1093/nar/29.9.e45
Rodríguez-Garcia A, Barreiro C, Santos-Beneit F, Sola-Landa A, Martín JF (2007) Genome-wide transcriptomic and proteomic analysis of the primary response to phosphate limitation in Streptomyces coelicolor M145 and in a DeltaphoP mutant. Proteomics 7(14):2410–29. doi:10.1002/pmic.200600883
Roubos JA, Krabben P, Luiten RG, Verbruggen HB, Heijnen JJ (2001) A quantitative approach to characterizing cell lysis caused by mechanical agitation of Streptomyces clavuligerus. Biotechnol Prog 17(2):336–47. doi:10.1021/bp0001617
Rudack D, Chisholm EM, Holten D (1971) Rat liver glucose 6-phosphate dehydrogenase. J Biol Chem 246(5):1249–1254
Rui B, Shen T, Zhou H, Liu J, Chen J, Pan X, Liu H, Wu J, Zheng H, Shi Y (2010) A systematic investigation of Escherichia coli central carbon metabolism in response to superoxide stress. BMC Syst Biol 4(1):122. doi:10.1186/1752-0509-4-122
Santos-Aberturas J, Vicente CM, Guerra SM, Payero TD, Martín JF, Aparicio JF (2011) Molecular control of polyene macrolide biosynthesis: direct binding of the regulator PimM to eight promoters of pimaricin genes and identification of binding boxes. J Biol Chem 286(11):9150–61. doi:10.1074/jbc.M110.182428
Santos-Aberturas J, Vicente CM, Payero TD, Martín-Sánchez L, Cañibano C, Martín JF, Aparicio JF (2012) Hierarchical control on polyene macrolide biosynthesis: PimR modulates pimaricin production via the PAS-LuxR transcriptional activator PimM. PLoS One 7(6):e38536. doi:10.1371/journal.pone.0038536
Santos-Beneit F, Rodríguez-Garcia A, Franco-Domínguez E, Martín JF (2008) Phosphate-dependent regulation of the low- and high-affinity transport systems in the model actinomycete Streptomyces coelicolor. Microbiology 154(Pt 8):2356–2370. doi:10.1099/mic.0.2008/019539-0
Schwartz D, Kaspar S, Kienzlen G, Muschko K, Wohlleben W (1999) Inactivation of the tricarboxylic acid cycle aconitase gene from Streptomyces viridochromogenes Tu494 impairs morphological and physiological differentiation. J Bacteriol 181(22):7131–7135
Smyth GK (2004) Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3(1). doi:10.2202/1544-6115.1027
Smyth GK, Michaud J, Scott HS (2005) Use of within-array replicate spots for assessing differential expression in microarray experiments. Bioinformatics 21(9):2067–2075. doi:10.1093/bioinformatics/bti270
Stirrett K, Denoya C, Westpheling J (2009) Branched-chain amino acid catabolism provides precursors for the type II polyketide antibiotic, actinorhodin, via pathways that are nutrient dependent. J Ind Microbiol Biotechnol 36(1):129–137. doi:10.1007/s10295-008-0480-0
Storz G, Imlay JA (1999) Oxidative stress. Curr Opin Microbiol 2(2):188–194. doi:10.1016/S1369-5274(99)80033-2
Struyk AP, Hoette I, Drost G, Waisvisz JM, Van Eek T, Hoogerheide JC (1957) Pimaricin, a new antifungal antibiotic. Antibiot Annu 5:878–885
Talaat AM, Howard ST, Wt H, Lyons R, Garner H, Johnston SA (2002) Genomic DNA standards for gene expression profiling in Mycobacterium tuberculosis. Nucleic Acids Res 30(20):e104. doi:10.1093/nar/gnf103
Tunca S, Barreiro C, Sola-Landa A, Coque JJ, Martín JF (2007) Transcriptional regulation of the desferrioxamine gene cluster of Streptomyces coelicolor is mediated by binding of DmdR1 to an iron box in the promoter of the desA gene. FEBS J 274(4):1110–1122. doi:10.1111/j.1742-4658.2007.05662.x
van Veen HW, Abee T, Kortstee GJ, Konings WN, Zehnder AJ (1994) Translocation of metal phosphate via the phosphate inorganic transport system of Escherichia coli. Biochemistry 33(7):1766–1770. doi:10.1021/bi00173a020
van Wezel GP, McDowall KJ (2011) The regulation of the secondary metabolism of Streptomyces: new links and experimental advances. Nat Prod Rep 28(7):1311–1333. doi:10.1039/c1np00003a
Vancurová I, Vancura A, Volc J, Neuzil J, Flieger M, Basarová G, Běhal V (1988) Isolation and characterization of valine dehydrogenase from Streptomyces aureofaciens. J Bacteriol 170(11):5192–5196
Vicente CM, Santos-Aberturas J, Guerra SM, Payero TD, Martín JF, Aparicio JF (2009) PimT, an amino acid exporter controls polyene production via secretion of the quorum sensing pimaricin-inducer PI-factor in Streptomyces natalensis. Microbial cell factories 8:33. doi:10.1186/1475-2859-8-33
Viollier PH, Nguyen KT, Minas W, Folcher M, Dale GE, Thompson CJ (2001) Roles of aconitase in growth, metabolism, and morphological differentiation of Streptomyces coelicolor. J Bacteriol 183(10):3193–203. doi:10.1128/JB.183.10.3193-3203.2001
Wei Z-H, Bai L, Deng Z, Zhong J-J (2011) Enhanced production of validamycin A by H2O2-induced reactive oxygen species in fermentation of Streptomyces hygroscopicus 5008. Bioresour Technol 102:1783–1787. doi:10.1016/j.biortech.2010.08.114
Yague P, Rodríguez-Garcia A, Lopez-Garcia MT, Martin JF, Rioseras B, Sanchez J, Manteca A (2013) Transcriptomic analysis of Streptomyces coelicolor differentiation in solid sporulating cultures: first compartmentalized and second multinucleated mycelia have different and distinctive transcriptomes. PLoS One 8(3):e60665. doi:10.1371/journal.pone.0060665
Yegneswaran PK, Gray MR, Thompson BG (1991) Effect of dissolved oxygen control on growth and antibiotic production in Streptomyces clavuligerus fermentations. Biotechnol Prog 7(3):246–50. doi:10.1021/bp00009a008
Yen HW, Hsiao HP (2013) Effects of dissolved oxygen level on rapamycin production by pellet-form of Streptomyces hygroscopicus. J Biosci Bioeng. doi:10.1016/j.jbiosc.2013.03.011
Acknowledgments
This work was funded by FCOMP-01-0124-FEDER-007080 (PTDC/BIO/64682/2006) project co-financed by FEDER funds through the COMPETE program and by National funds through Fundação para a Ciência e Tecnologia (FCT), and by BIO2010-19911 project funded by the Spanish Ministerio de Ciencia e Innovación. MVM was supported by “Programa Ciência 2007” sponsored by POPH QREN program, funded by the European Social Fund and Portuguese National funds from the MCTES. TB was supported by the FCT fellowship SFRH/BD/40100/2007. We thank P. Magalhães and C. Cañibano for excellent technical assistance in RT-qPCR and microarray hybridization, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beites, T., Rodríguez-García, A., Moradas-Ferreira, P. et al. Genome-wide analysis of the regulation of pimaricin production in Streptomyces natalensis by reactive oxygen species. Appl Microbiol Biotechnol 98, 2231–2241 (2014). https://doi.org/10.1007/s00253-013-5455-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5455-z