Abstract
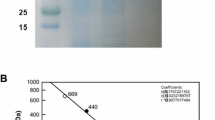
A gene in Bradyrhizobium japonicum USDA 110, annotated as a ribitol dehydrogenase (RDH), had 87 % sequence identity (97 % positives) to the N-terminal 31 amino acids of an l-glucitol dehydrogenase from Stenotrophomonas maltophilia DSMZ 14322. The 729-bp long RDH gene coded for a protein consisting of 242 amino acids with a molecular mass of 26.1 kDa. The heterologously expressed protein not only exhibited the main enantio selective activity with d-glucitol oxidation to d-fructose but also converted l-glucitol to d-sorbose with enzymatic cofactor regeneration and a yield of 90 %. The temperature stability and the apparent K m value for l-glucitol oxidation let the enzyme appear as a promising subject for further improvement by enzyme evolution. We propose to rename the enzyme from the annotated RDH gene (locus tag bll6662) from B. japonicum USDA as a d-sorbitol dehydrogenase (EC 1.1.1.14).







Similar content being viewed by others
References
Baek SH, Park SJ, Lee HG (2010) d-psicose, a sweet monosaccharide, ameliorate hyperglycemia, and dyslipidemia in C57BL/6 J db/db mice. J Food Sci 75:H49–H53
Beerens K, Desmet T, Soetaert W (2012) Enzymes for the biocatalytic production of rare sugars. J Ind Microbiol Biotechnol 39:823–834
Bon Saint Côme Y, Lalo H, Wang Z, Kohring GW, Hempelmann R, Etienne M, Walcarius A, Kuhn A (2013) Interest of the sol–gel approach for multiscale tailoring of porous bioelectrode surfaces. Electroanalysis 25:621–629
Brechtel E, Huwig A, Giffhorn F (2002) l-glucitol catabolism in Stenotrophomonas maltophilia Ac. Appl Environ Microbiol 68:582–587
Chenault HK, Whitesides GM (1989) Lactate dehydrogenase-catalyzed regeneration of NAD from NADH for use in enzyme-catalyzed synthesis. Bioorg Chem 17:400–409
Frush HL, Isbell HS (1956) Sodium borohydride reduction of aldonic lactones to glucitol. J Am Chem Soc 78:2844–2846
Gajdizik J, Szamocki R, Natter H, Kohring GW, Giffhorn F, Hempelmann R (2007) Electroenzymatic reactions with sorbitol dehydrogenase on gold electrodes. J Solid State Electrochem 11:144–149
Granstrom TB, Takata G, Tokuda M, Izumori K (2004) Izumoring: a novel and complete strategy for bioproduction of rare sugars. J Biosci Bioeng 97:89–94
Gumina G, Song GY, Chu CK (2001) l-nucleosides as chemotherapeutic agents. FEMS Microbiol Lett 202:9–15
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Hollmann F, Schmid A (2004) Electrochemical regeneration of oxidoreductases for cell-free biocatalytic redox reactions. Biocatal Biotransform 22:63–88
Hollmann F, Arends IWCE, Buehler K (2010) Biocatalytic redox reactions for organic synthesis: nonconventional regeneration methods. ChemCatChem 2:762–782
Huwig A, Emmel S, Giffhorn F (1996) Preparation of d-sorbose from l-glucitol by bioconversion with Pseudomonas sp Ac. Carbohydr Res 281:183–186
Kaneko T, Nakamura Y, Sato S, Minamisawa K, Uchiumi T, Sasamoto S, Watanabe A, Idesawa K, Iriguchi M, Kawashima K, Kohara M, Matsumoto M, Shimpo S, Tsuruoka H, Wada T, Yamada M, Tabata S (2002) Complete genomic sequence of nitrogen-fixing symbiotic bacterium Bradyrhizobium japonicum USDA110. DNA Res 9:189–197
Kavanagh KL, Jornvall H, Persson B, Oppermann U (2008) The SDR superfamily: functional and structural diversity within a family of metabolic and regulatory enzymes. Cell Mol Life Sci 65:3895–3906
Kornberger P, Gajdzik J, Natter H, Wenz G, Giffhorn F, Kohring GW, Hempelmann R (2009) Modification of galactitol dehydrogenase from Rhodobacter sphaeroides D for immobilization on polycrystalline gold surfaces. Langmuir 25:12380–12386
Kornberger P, Giffhorn F, Kohring GW (2010) Dehydrogenases, electrochemical co-factor regeneration. In: Flickinger MC (ed) Encyclopedia of industrial biotechnology, bioprocess, bioseparation, and cell technology. Wiley, Hoboken, pp 1888–1898
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lim YR, Oh DK (2011) Microbial metabolism and biotechnological production of d-allose. Appl Microbiol Biotechnol 91:229–235
Lindstad RI, Köll P, McKinley-McNee JS (1998) Substrate specificity of sheep liver sorbitol dehydrogenase. Biochem J 330:479–487
Livesey G (2003) Health potential of polyols as sugar replacers, with emphasis on low glycaemic properties. Nutr Res Rev 16:163–191
Lu Y, Levin GV, Donner TW (2008) Tagatose, a new antidiabetic and obesity control drug. Diabetes Obes Metab 10:109–134
Mayerskuntzer H, Reichert A, Schneider KH, Giffhorn F (1994) Isolation and characterization of a l-glucitol dehydrogenase from the newly isolated bacterium Pseudomonas sp Ac. J Biotechnol 36:157–164
Monedero V, Perez-Martinez G, Yebra MJ (2010) Perspectives of engineering lactic acid bacteria for biotechnological polyol production. Appl Microbiol Biotechnol 86:1003–1015
Perez-Iratxeta C, Andrade-Navarro MA (2008) K2D2: estimation of protein secondary structure from circular dichroism spectra. BMC Struct Biol 8:25
Philippsen A, Schirmer T, Stein MA, Giffhorn F, Stetefeld J (2005) Structure of zinc-independent sorbitol dehydrogenase from Rhodobacter sphaeroides at 2.4 Angstrom resolution. Acta Crystallogr D 61:374–379
Reetz MT (2012) Laboratory evolution of stereoselective enzymes as a means to expand the toolbox of organic chemists. Tetrahedron 68:7530–7548
Schauder S, Schneider KH, Giffhorn F (1995) Polyol metabolism of Rhodobacter sphaeroides: biochemical characterization of a short-chain sorbitol dehydrogenase. Microbiology 141:1857–1863
Schneider KH, Giffhorn F (1991) Sorbitol dehydrogenase from Pseudomonas sp: purification, characterization and application to quantitative-determination of sorbitol. Enzym Microb Technol 13:332–337
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Steckhan E (1994) Electroenzymatic synthesis. Top Curr Chem 170:83–111
van Loveren C (2004) Sugar alcohols: what is the evidence for caries preventive and caries-therapeutic effects? Caries Res 38:286–293
Wang Z, Etienne M, Kohring GW, Walcarius A (2010) Critical effect of polyelectrolytes on the electrochemical response of dehydrogenases entrapped in sol–gel thin films. Electroanalysis 22:2092–2100
Wang Z, Etienne M, Kohring GW, Bon-Saint-Come Y, Kuhn A, Walcarius A (2011) Electrochemically assisted deposition of sol–gel bio-composite with co-immobilized dehydrogenase and diaphorase. Electrochim Acta 56:9032–9040
Wang Z, Etienne M, Quiles F, Kohring GW, Walcarius A (2012) Durable cofactor immobilization in sol–gel bio-composite thin films for reagentless biosensors and bioreactors using dehydrogenases. Biosens Bioelectron 32:111–117
Wichmann D, Vasic-Racki D (2005) Cofactor regeneration at the lab scale. Adv Biochem Eng Biotechnol 92:225–260
Zumbé A, Lee A, Storey D (2001) Polyols in confectionery: the route to sugar-free, reduced sugar and reduced calorie confectionery. Br J Nutr 85:S31–S45
Acknowledgments
Financial support from the European Community through the FP7-NMP-2007-SMALL 1 collaborative project ERUDESP is gratefully acknowledged. We thank Dr. Josef Zapp, Saarland University, for the NMR and IR determinations, and Birgit Hasper for the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gauer, S., Wang, Z., Otten, H. et al. An l-glucitol oxidizing dehydrogenase from Bradyrhizobium japonicum USDA 110 for production of d-sorbose with enzymatic or electrochemical cofactor regeneration. Appl Microbiol Biotechnol 98, 3023–3032 (2014). https://doi.org/10.1007/s00253-013-5180-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5180-7