Abstract
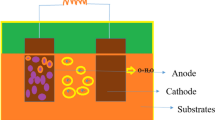
The membraneless bioelectrochemical reactor (Ml-BER) is useful for dark hydrogen fermentation. The effect of the electrochemical reaction on microorganisms in the Ml-BER was investigated using glucose as the substrate and compared with organisms in a membraneless non-bioelectrochemical reactor (Ml-NBER) and bioelectrochemical reactor (BER) with a proton exchange membrane. The potentials on the working electrode of the Ml-BER and BER with membrane were regulated to −0.9 V (versus Ag/AgCl) to avoid water electrolysis with a carbon electrode. The Ml-BER showed suppressed methane production (19.8 ± 9.1 mg-C·L−1·day−1) and increased hydrogen production (12.6 ± 3.1 mg-H·L−1·day−1) at pHout 6.2 ± 0.1, and the major intermediate was butyrate (24.9 ± 2.4 mM), suggesting efficient hydrogen fermentation. In contrast, the Ml-NBER showed high methane production (239.3 ± 17.9 mg-C·L−1·day−1) and low hydrogen production (0.2 ± 0.0 mg-H·L−1·day−1) at pHout 6.3 ± 0.1. In the cathodic chamber of the BER with membrane, methane production was high (276.3 ± 20.4 mg-C·L−1·day−1) (pHout, 7.2 ± 0.1). In the anodic chamber of the BER with membrane (anode-BER), gas production was low because of high lactate production (43.6 ± 1.7 mM) at pHout 5.0 ± 0.1. Methanogenic archaea were not detected in the Ml-BER and anode-BER. However, Methanosarcina sp. and Methanobacterium sp. were found in Ml-NBER. Prokaryotic copy numbers in the Ml-BER and Ml-NBER were similar, as were the bacterial community structures. Thus, the electrochemical reaction in the Ml-BER affected hydrogenotrophic and acetoclastic methanogens, but not the bacterial community.


Similar content being viewed by others
References
Abreu AA, Karakashev D, Angelidaki I, Sousa DZ, Alves MM (2012) Biohydrogen production from arabinose and glucose using extreme thermophilic anaerobic mixed cultures. Biotechnol Biofuels 5:6
Ben Hania W, Godbane R, Postec A, Hamdi M, Ollivier B, Fardeau ML (2012) Isolation and characterization of Defluviitoga tunisiensis gen. nov, sp. nov., a novel thermophilic bacterium pertaining to the order Thermotogales, isolated from a mesothermic anaerobic reactor treating cheese whey in Tunisia. Int J Syst Evol Microbiol 62(Pt 6):1377–1382. doi:10.1099/ijs.0.033720-0
Bryant MP, Boone DR (1987) Isolation and characterization of Methanobacterium formicicum MF. Int J Syst Bacteriol 37:171
Cardinali-Rezende J, Debarry RB, Colturato LFDB, Carneiro EV, Chartone-Souza E, Nascimento AMA (2009) Molecular identification and dynamics of microbial communities in reactor treating organic household waste. Appl Microbiol Biotechnol 84:777–789
Chong ML, Rahman NAA, Yee PL, Aziz SA, Rahim RA, Shirai Y, Hassan MA (2009) Effect of pH, glucose and iron sulfate concentration on the yield of biohydrogen by Clostridium butyricum EB6. Int J Hydrog Energy 34:8859–8865
Cirne DG, Bond P, Pratt S, Lant P, Batstone DJ (2012) Microbial community analysis during continuous fermentation of thermally hydrolysed waste activated sludge. Water Sci Technol 65:7–14
Clauwaert P, Verstraete W (2009) Methanogenesis in membraneless microbial electrolysis cells. Appl Microbiol Biotechnol 82:829–836
Freguia S, Rabaey K, Yuan Z, Kerrer J (2008) Syntrophic processed drive the conversion of glucose in microbial fuel cell anodes. Environ Sci Technol 42:7937–7943
Harold FM, Levin E (1974) Lactic acid translocation: terminal step in glycolysis by Streptococcus faecalis. J Bacteriol 117:1141–1148
Hawkes FR, Hussy I, Kyazze G, Dinsdale R, Hawkes DL (2007) Continuous dark fermentative hydrogen production by mesophilic microflora: principles and progress. Int J Hydrog Energy 32:172–184
Hoover SR, Porges N (1952) Assimilation of dairy wastes by activated sludge. II. The equation of synthesis and oxygen utilization. Sewage Ind Wastes 24:306–312
Jung S, Regan JM (2011) Influence of external resistance on electrogenesis, methanogenesis, and anode prokaryotic communities in microbial fuel cells. Appl Environ Microbiol 77:564–571
Jungermann K, Thauer RK, Leimenstoll G, Decker K (1973) Function of reduced pyridine nucleotide-ferredoxin oxidoreductases in saccharolytic Clostridia. Biochim Biophys Acta 305:268–280
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York, pp 115–175
Lee HS, Vermaas WFJ, Rittmann BE (2010) Biological hydrogen production: prospects and challenges. Trends Biotechnol 28:262–271
Li C, Fang HHP (2007) Fermentative hydrogen production from wastewater and solid wastes by mixed cultures. Crit Rev Environ Sci Technol 37:1–39
Logan BE (2010) Scaling up microbial fuel cells and other bioelectrochemical systems. Appl Microbiol Biotechnol 85:1665–1671
Lueders T, Friedrich MW (2002) Effects of amendment with ferrihydrite and gypsum on the structure and activity of methanogenic populations in rice field soil. Appl Environ Microbiol 68:2484–2494
Lueders T, Manefield M, Friedrich MW (2004) Enhanced sensitivity of DNA- and rRNA-based stable isotope probing by fractionation and quantitative analysis of isopycnic centrifugation gradients. Environ Microbiol 6:73–78
Luo G, Karakashev D, Xie L, Zhou Q, Angeridaki I (2011) Long-term effect of inoculum pretreatment on fermentative hydrogen production by repeated batch cultivations: homoacetogenesis and methanogenesis as competitors to hydrogen production. Biotechnol Bioeng 108:1816–1827
Nanqi R, Wanqian G, Bingfeng L, Guangli C, Jie D (2011) Biological hydrogen production by dark fermentation: challenges and prospects towards scaled-up production. Curr Opin Biotechnol 22:365–370
Patel MA, Ou MS, Harbrucker R, Aldrich HC, Buszko ML, Ingram LO, Shanmugam KT (2006) Isolation and characterization of acid-tolerant, thermophilic bacteria for effective fermentation of biomass-derived sugars to lactic acid. Appl Environ Microbiol 72:3228–3235
Rouvière P, Mandelco L, Winker S, Woese CR (1992) A detailed phylogeny for the Methanomicrobiales. Syst Appl Microbiol 15:363–371
Rozendal RA, Jeremiasse AW, Hamelers HVM, Buisman CJN (2008) Hydrogen production with a microbial biocathode. Environ Sci Technol 42:629–634
Sasaki K, Morita M, Hirano S, Ohmura N, Igarashi Y (2009) Effect of adding carbon fiber textiles to methanogenic bioreactors used to treat an artificial garbage slurry. J Biosci Bioeng 108:130–135
Sasaki K, Sasaki D, Morita M, Hirano S, Matsumoto N, Ohmura N, Igarashi Y (2010) Bioelectrochemical system stabilizes methane fermentation from garbage slurry. Bioresour Technol 101:3415–3422
Sasaki D, Hori T, Haruta S, Ueno Y, Ishii M, Igarashi Y (2011) Methanogenic pathway and community structure in a thermophilic anaerobic digestion process of organic solid waste. J Biosci Bioeng 111:41–46
Sasaki K, Morita M, Matsumoto N, Sasaki D, Hirano S, Ohmura N, Igarashi Y (2012a) Construction of hydrogen fermentation from garbage slurry using the membrane free bioelectrochemical system. J Biosci Bioeng 114:64–69
Sasaki K, Morita M, Sasaki D, Matsumoto N, Ohmura N, Igarashi Y (2012b) Single-chamber bioelectrochemical hydrogen fermentation from garbage slurry. Biochem Eng J 68:104–108
Sawayama S, Tsukahara K, Yagishita T (2006) Phylogenetic description of immobilized methanogenic community using real-time PCR in a fixed-bed anaerobic digester. Bioresour Technol 97:69–76
Soh ALA, Ralambotiana H, Olliver B, Prensier G, Tina E, Garcia JL (1991) Clostridium thermopalmarium sp. nov., a moderately thermophilic butyrate-producing bacterium isolated from palm wine in Senegal. Syst Appl Microbiol 14:135–139
Takai K, Horikoshi K (2000) Rapid detection and quantification of members of the archaeal community by quantitative PCR using fluorogenic probes. Appl Environ Microbiol 66:5066–5072
Thrash JG, Coates JD (2008) Review: Direct and indirect electrical stimulation of microbial metabolism. Environ Sci Technol 42:3921–3931
Ueno Y, Sasaki D, Fukui H, Haruta S, Ishii M, Igarashi Y (2006) Changes in bacterial community during fermentative hydrogen and acid production from organic waste by thermophilic anaerobic microflora. J Appl Microbiol 101:331–343
Ueno Y, Tatara M, Fukui H, Makiuchi T, Goto M, Sode K (2007) Production of hydrogen and methane from organic solid wastes by phase-separation of anaerobic process. Bioresour Technol 98:1861–1865
Updegraff DM (1969) Semimicro determination of cellulose in biological materials. Anal Biochem 32:420–424
Zeikus JG (1977) The biology of methanogenic bacteria. Bacteriol Rev 41:514–541
Zinder SH, Sower KR, Ferry JG (1985) Methanosarcina thermophila sp. nov., a thermophilic, acetotrophic, methane-producing bacterium. Int J Syst Bacteriol 35:522–523
Acknowledgments
This research was supported in part by the New Energy and Industrial Technology Development Organization (NEDO), Japan, and a Grant-in-Aid for Young Scientists (B) (24780067). We thank Yumi Kotake for her help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sasaki, K., Morita, M., Sasaki, D. et al. The membraneless bioelectrochemical reactor stimulates hydrogen fermentation by inhibiting methanogenic archaea. Appl Microbiol Biotechnol 97, 7005–7013 (2013). https://doi.org/10.1007/s00253-012-4465-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4465-6