Abstract
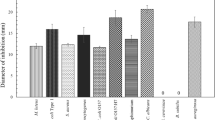
The yeast Kluyveromyces siamensis HN12-1 isolated from mangrove ecosystem was found to be able to produce killer toxin against the pathogenic yeast (Metschnikowia bicuspidata WCY) in crab. When the killer yeast was grown in the medium with pH 4.0 and 0.5% NaCl and at 25 °C, it could produce the highest amount of killer toxin against the pathogenic yeast M. bicuspidata WCY. The killing activity of the purified killer toxin against the pathogenic yeast M. bicuspidata WCY was the highest when it was incubated at 25 °C in the assay medium without added NaCl and pH 4.0. The molecular weight of the purified killer toxin was 66.4 kDa. The killer toxin produced by the yeast strain HN12-1 could kill only the whole cells of M. bicuspidata WCY among all the yeast species tested in this study. This is the first time to report that the killer toxin produced by the yeast K. siamensis HN12-1 isolated from the mangrove ecosystem only killed pathogenic yeast M. bicuspidata WCY.






Similar content being viewed by others
References
Am-In S, Yongmanitchai W, Limtong S (2008) Kluyveromyces siamensis sp. nov., an ascomycetous yeast isolated from water in a mangrove forest in Ranong Province, Thailand. FEMS Yeast Res 8:823–8
Chi ZM, Liu GL, Zhao SF, Li J, Peng Y (2010) Marine yeasts as biocontrol agents and producers of bio-products. Appl Microbiol Biotechnol 86:1227–1241
Comitini F, Di Pietro N, Zacchi L, Mannazzu I, Ciani M (2004) Kluyveromyces phaffii killer toxin active against wine spoilage yeasts: purification and characterization. Microbiol 150:2535–2541
de Araujo EV, Soares CAG, Hagler AN, Mendonqa-Hagler LC (1995) Ascomycetous yeast communities of marine invertebrates in a Southeast Brazilian mangrove ecosystem. Antonie van Leeuwenhoek 68:91–99
Gatesoupe FJ (2007) Live yeasts in the gut: natural occurrence, dietary introduction, and their effects on fish health and development. Aquaculture 267:20–30
Gunge N, Tamaru A, Ozawa F, Sakaguchi K (1981) Isolation and characterization of linear deoxyribonucleic acid plasmids from Kluyveromyces lactis and the plasmid-associated killer character. J Bacteriol 145:382–390
Guyard C, Dehecq E, Tissier JP, Polonelli L, Dei-Cas E, Cailliez JC, Menozzi FD (2002) Involvement of [beta]-glucans in the wide-spectrum antimicrobial activity of Williopsis saturnus var. mrakii MUCL 41968 killer toxin. Mol Med 8:686–694
Hua MX, Chi Z, Liu GL, Buzdar MA, Chi ZM (2010) Production of a novel and cold-active killer toxin by Mrakia frigida 2E00797 isolated from sea sediment in Antarctica. Extremophiles 14:515–521
Izg¨u F, Altinbay D, T¨ureli AE (2007) In vitro susceptibilities of Candida spp. to Panomycocin, a novel exo-beta-1,3-glucanase isolated from Pichia anomala NCYC 434. Microbiol Immunol 51:797–803
Jablonowski D, Schaffrath R (2007) Zymocin, a composite chitinase and tRNase killer toxin from yeast. Biochem Soc Trans 35:1533–1537
Kurtzman CP, Fell JW (1998) The yeast: a taxonomic study, 4th edn. Elsevier, Amsterdam, pp 77–107
Laemmli UK (1970) Cleavage of structural proteins during assembly of head of bacteriophage T4. Nature 227:680–685
Magliani W, Conti S, Gerloni M, Beretolotti D, Luciano PL (1997) Yeast killer systems. Clin Microbiol Rev 10:369–400
Magliani W, Conti S, Travassos LR, Polonelli L (2008) From yeast killer toxins to antibiobodies and beyond. FEMS Microbiol Lett 288:1–8
Marquina D, Barroso J, Santos A, Peinado JM (2001) Production and characteristics of Debaryomyces hansenii killer toxin. Microbiol Res 156:387–391
Meyers SP, Ahearn DG, Alexander SK, Cook WL (1975) Pichia spartinae, a dominant yeast of the Spartina salt marsh. Dev Ind Microbiol 16:262–267
Nagahama T (2006) Yeast biodiversity in freshwater, marine and deep-sea environments. In: Biodiversity and Ecophysiology of Yeasts, The Yeast Handbook, Springer Berlin Heidelberg. pp 241–269
Peng Y, Chi ZM, Wang XH, Li J (2010) β-1,3-Glucanase inhibits activity of the killer Toxin produced by the marine-derived yeast Williopsis saturnus WC91-2. Mar Biotechnol 12:479–485
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Beijing, pp 367–370, Chinese translating ed
Santos A, Marquina D, Leal JA, Peinado JM (2000) (1 → 6)-beta-D-glucan as cell wall receptor for Pichia membranifaciens killer toxin. Appl Environ Microbiol 66:1809–1813
Stark MJR, Boyd A (1986) The killer toxin of Kluyveromyces lactis: characterization of the toxins subunits and identification of the genes which encode them. EMBO J 5:1995–2002
Sugita T, Takashima M, Kodama M, Tsuboi R, Nishikawa A (2003) Description of a new yeast species, Malassezia japonica, and its detection in patients with atopic dermatitis and healthy subjects. J Clin Microbiol 41:4695–4699
Varghese G, Diwan AM (1983) Simultaneous staining of proteins during polyacrylamide gel electrophoresis in acidic gels by countermigration of Coomassie brilliant blue R-250. Anal Biochem 132:481–483
Wang X, Chi ZM, Yue L, Li J, Li M, Wu L (2007a) A marine killer yeast against the pathogenic yeast strain in crab (Portunus trituberculatus) and an optimization of the toxin production. Microbiol Res 162:77–85
Wang X, Chi ZM, Yue L, Li J (2007b) Purification and characterization of killer toxin from a marine yeast Pichia anomala YF07b against the pathogenic yeast in crab. Curr Microbiol 55:396–401
Wang L, Yue L, Chi ZM, Wang X (2008) Marine killer yeasts active against a yeast strain pathogenic to crab Portunus trituberculatus. Dis Aquat Organ 80:211–218
Zhang YB, Lin P, Zhuang TC (2003) Microbial decomposition and its roles in nutrient recycle in mangrave ecosystem. Mar Sci 27:9–12
Acknowledgment
This work was supported by grant 30771645 from National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buzdar, M.A., Chi, Z., Wang, Q. et al. Production, purification, and characterization of a novel killer toxin from Kluyveromyces siamensis against a pathogenic yeast in crab. Appl Microbiol Biotechnol 91, 1571–1579 (2011). https://doi.org/10.1007/s00253-011-3220-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3220-8