Abstract
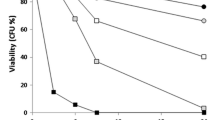
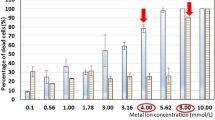
The effect of Pb on Saccharomyces cerevisiae cell structure and function was examined. Membrane integrity was assessed by the release of UV-absorbing compounds and by the intracellular K+ efflux. No leakage of UV260-absorbing compounds or loss of K+ were observed in Pb (until 1,000 μmol/l) treated cells up to 30 min; these results suggest that plasma membrane seems not to be the immediate and primary target of Pb toxicity. The effect of Pb on yeast metabolism was examined using the fluorescent probe FUN-1 and compared with the ability to reproduce, evaluated by colony-forming units counting. The exposition of yeast cells, during 60 min to 1,000 μmol/l Pb, induces a decrease in the ability to process FUN-1 although the cells retain its proliferation capacity. A more prolonged contact time (120 min) of yeast cells with Pb induces a marked (> 50%) loss of yeast cells metabolic activity and replication competence through a mechanism which most likely requires protein synthesis.





Similar content being viewed by others
References
ATSDR (2007) Toxicological profile for lead. Agency for Toxic Substances and Disease Registry (ATSDR). U.S. Department of Health and Human Services—Public Health Service
Borst-Pauwels GWFH (1981) Ion transport in yeast. Biochim Biophys Acta 650:88–127
Brady D, Duncan JR (1994) Cation loss during accumulation of heavy metal cations by Saccharomyces cerevisiae. Biotechnol Lett 16:543–548
Chen C, Wang JL (2007) Response of Saccharomyces cerevisiae to lead ion stress. Appl Microbiol Biotechnol 74:683–687
De Rome L, Gadd GM (1987) Measurement of copper uptake in Saccharomyces cerevisiae using a Cu2+-selective electrode. FEMS Microbiol Lett 43:283–287
Delisle AL, Phaff HJ (1961) The release of nitrogenous substances by brewer’s yeast. Proc Am Soc Brew Chem 103–118
Gadd GM (1993) Interaction of fungi with toxic metals. New Phytologist 124:25–60
Gadd GM, Mowll JL (1983) The relationship between cadmium uptake, potassium release and viability in Saccharomyces cerevisiae. FEMS Microbiol Lett 16:45–48
Gharieb MM, Gadd GM (2004) Role of glutathione in detoxification of metal(loid)s by Saccharomyces cerevisiae. Biometals 17:183–188
Gilfillan SC (1965) Lead poisoning and the fall of Rome. J Occup Med 7:53–60
Goffeau A, Barrell BG, Bussey H, Davis RW, Dujon B, Feldmann H, Galibert F, Hoheisel JD, Jacq C, Johnston M, Louis EJ, Mewes HW, Murakami Y, Philippsen P, Tettelin H, Oliver SG (1996) Life with 6000 genes. Science 274:546–567
Joho M, Ishibe A, Murayama T (1984) The injurious effect of heavy metal ions on the cell membrane in Saccharomyces cerevisiae. Trans Mycol Soc Japan 25:485–488
Klis FM, Mol P, Hellingwerf K, Brul S (2002) Dynamics of cell wall structure in Saccharomyces cerevisiae. FEMS Microbiol Rev 26:239–256
Kuo SC, Cano FR, Lampen JO (1973) Lomofungin, an inhibitor of ribonucleic acid synthesis in yeast protoplasts: its effect on enzyme formation. Antimicrob Agents Chemother 3:716–722
Kuypers GAJ, Roomans GM (1979) Mercury induced loss of K+ from yeast cells investigated by electron-probe X-ray-microanalysis. J Gen Microbiol 115:13–18
Millard PJ, Roth BL, Thi HPT, Yue ST, Haugland RP (1997) Development of the FUN-1 family of fluorescent probes for vacuole labeling and viability testing of yeasts. Appl Environ Microbiol 63:2897–2905
Mowll JL, Gadd GM (1983) Zinc uptake and toxicity in the yeasts Sporobolomyces roseus and Saccharomyces cerevisiae. J Gen Microbiol 129:3421–3425
Norris PR, Kelly DP (1977) Accumulation of cadmium and cobalt by Saccharomyces cerevisiae. J Gen Microbiol 99:317–324
Ohsumi Y, Kitamoto K, Anraku Y (1988) Changes induced in the permeability barrier of the yeast plasma membrane by cupric ion. J Bacteriol 170:2676–2682
Ramsay LM, Gadd GM (1997) Mutants of Saccharomyces cerevisiae defective in vacuolar function confirm a role for the vacuole in toxic metal ion detoxification. FEMS Microbiol Lett 152:293–298
Soares HMVM, Conde PCFL, Almeida AAN, Vasconcelos MTSD (1999a) Evaluation of n-substituted aminosulfonic acid pH buffers with a morpholinic ring for cadmium and lead speciation studies by electroanalytical techniques. Anal Chim Acta 394:325–335
Soares HMVM, Pinho SC, Barros MGRTM (1999b) Influence of N-substituted aminosulfonic acids with a morpholinic ring pH buffers on the redox processes of copper or zinc ions: a contribution to speciation studies. Electroanalysis 11:1312–1317
Soares EV, Duarte A, Soares H (2000) Study of the suitability of 2-(N-morpholino) ethanesulfonic acid pH buffer for heavy metals accumulation studies using Saccharomyces cerevisiae. Chem Speciation Bioavail 12:59–65
Soares EV, Duarte APSR, Boaventura RA, Soares HMVM (2002) Viability and release of complexing compounds during accumulation of heavy metals by a brewer’s yeast. Appl Microbiol Biotechnol 58:836–841
Soares EV, Hebbelinck K, Soares HMVM (2003) Toxic effects caused by heavy metals in the yeast Saccharomyces cerevisiae: a comparative study. Can J Microbiol 49:336–343
Suh JH, Yun JW, Kim DS (1999) Cation (K+, Mg2+, Ca2+) exchange in Pb2+ accumulation by Saccharomyces cerevisiae. Bioprocess Eng 21:383–387
US-EPA (2006) National recommended water quality criteria. United States Environmental Protection Agency EPA. Office of Water, Science and Technology (4304T), Washington, DC
Wang J, Chen C (2006) Biosorption of heavy metals by Saccharomyces cerevisiae: a review. Biotechnol Adv 24:427–451
Acknowledgment
Maarten Van der Heggen (KaHo St.-Lieven, Ghent, Belgium) wishes to thank Dr. Luc De Cooman for the opportunity to participate in the ERASMUS bilateral agreement program between his school and ISEP (Portugal).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Van der Heggen, M., Martins, S., Flores, G. et al. Lead toxicity in Saccharomyces cerevisiae . Appl Microbiol Biotechnol 88, 1355–1361 (2010). https://doi.org/10.1007/s00253-010-2799-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2799-5