Abstract
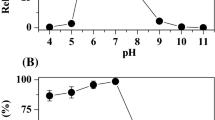
Quinone reductase activity of azoreductase AZR from Rhodobacter sphaeroides was reported. High homologies were found in the cofactor/substrate-binding regions of quinone reductases from different domains. 3D structure comparison revealed that AZR shared a common overall topology with mammal NAD(P)H/quinone oxidoreductase NQO1. With menadione as substrate, the optimal pH value and temperature were pH 8–9 and 50°C, respectively. Following the ping-pong kinetics, AZR transferred two electrons from NADPH to quinone substrate. It could reduce naphthoquinones and anthraquinones, such as menadione, lawsone, anthraquinone-2-sulfonate, and anthraquinone-2,6-disulfonate. However, no activity was detected with 1,4-benzoquinone. Dicoumarol competitively inhibited AZR’s quinone reductase activity with respect to NADPH, with an obtained K i value of 87.6 μM. Significantly higher survival rates were obtained in Escherichia coli YB overexpressing AZR than in the control strain when treated by heat shock and oxidative stressors such as H2O2 and menadione.




Similar content being viewed by others
References
Andrade SLA, Patridge EV, Ferry JG, Einsle O (2007) Crystal structure of the NADH:quinone oxidoreductase WrbA from Escherichia coli. J Bacteriol 189:9101–9107
Asher G, Dym O, Tsvetkov P, Adler J, Shaul Y (2006) The crystal structure of NAD(P)H quinone oxidoreductase 1 in complex with its potent inhibitor dicoumarol. Biochemistry 45:6372–6378
Baker CJ, O’Neill NR, Keppler LD, Orlandi EW (1991) Early responses during plant-bacteria interactions in tobacco cell suspensions. Phytopathology 81:1504–1507
Beyer RE (1994) The relative essentiality of the antioxidative function of coenzyme Q-the interactive role of DT-diaphorase. Mol Aspects Med 15(suppl.):117–129
Bianchet MA, Faig M, Amzel LM (2004) Structure and mechanism of NAD(P)H:quinone acceptor oxidoreductases (NQO). Methods Enzymol 382:144–174
Blümel S, Knackmuss HJ, Stolz A (2002) Molecular cloning and characterization of the gene coding for the aerobic azoreductase from Xenophilus azovorans KF46F. Appl Environ Microbiol 68:3948–3955
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cases I, de Lorenze V (2005) Genetically modified organisms for the environment: stories of success and failure and what we have learned from them. Int Microbiol 8:213–222
Cenas N, Nemeikaite-Ceniene A, Sergediene E, Nivinskas H, Anusevicius Z, Sarlauskas J (2001) Quantitative structure-activity relationships in enzymatic single-electron reduction of nitroaromatic explosives: implications for their cytotoxicity. Biochim Biophys Acta 1528:31–38
Chen H (2006) Recent advances in azo dye degrading enzyme research. Curr Protein Pept Sci 7:101–111
Chen S, Wu K, Zhang D, Sherman M, Knox R, Yang CS (1999) Molecular characterization of binding of substrates and inhibitors to DT-diaphorase: combined approach involving site-directed mutagenesis, inhibitor-binding analysis, and computer modeling. Mol Pharmacol 56:272–278
Deller S, Sollner S, Trenker-El-Toukhy R, Jelesarov I, Gubitz GM, Macheroux P (2006) Characterization of a thermostable NADPH:FMN oxidoreductase from the mesophilic bacterium Bacillus subtilis. Biochemistry 45:7083–7091
Deller S, Macheroux P, Sollner S (2008) Flavin-dependent quinone reductases. Cell Mol Life Sci 65:141–160
Dos Santos AB, Cervantes FJ, Van Lier JB (2007) Review paper on current technologies for decolourisation of textile wastewaters: pespectives for anaerobic biotechnology. Biores Technol 98:2369–2385
Ercal N, Gurer-Orhan H, Aykin-Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Garbisu C, Alkorta I (1999) Utilization of genetically engineered microorganisms (GEMs) for bioremediation. J Chem Technol Biotechnol 74:599–606
Gonzalez CF, Ackerley DF, Lynch SV, Matin A (2005) ChrR, a soluble quinone reductase of Pseudomonas putida that defends against H2O2. J Biol Chem 280:22590–22595
Hong Y, Wang G, Maier RJ (2008) The NADPH quinone reductase MdaB confers oxidative stress resistance to Helicobacter hepaticus. Microb Pathog 44:169–174
Iyanagi T (1987) On the mechanisms of one- and two-electron transfer by flavin enzymes. Chemica Scripta 27A:31–36
Kim HJ, Kang BS, Park JW (2005) Cellular defense against heat shock-induced oxidative damage by mitochondrial NADP+-dependent isocitrate dehydrogenase. Free Radic Res 39:441–448
Laskowski MJ, Dreher KA, Gehring MA, Abel S, Gensler AL, Sussex IM (2002) FQR1, a novel primary auxin-response gene, encodes a flavin mononucleotide-binding quinone reductase. Plant Physiol 128:578–590
Li R, Bianchet MA, Talalay P, Amzel LM (1995) The three-dimensional structure of NAD(P)H:quinone reductase, a flavoprotein involved in cancer chemoprotection and chemotherapy: mechanism of the two-electron reduction. Proc Natl Acad Sci U S A 92:8846–8850
Liger D, Graille M, Zhou CZ, Leulliot N, Quevillon-Cheruel S, Blondeau K, Janin J, van Tilbeurgh H (2004) Crystal structure and functional characterization of yeast YLR011wp, an enzyme with NAD(P)H-FMN and ferric iron reductase activities. J Biol Chem 279:34890–34897
Lind C, Hochstein P, Ernster L (1982) DT-diaphorase as a quinone reductase: a cellular control device against semiquinone and superoxide radical formation. Arch Biochem Biophys 216:178–185
Liu G, Zhou J, Lv H, Xiang X, Wang J, Zhou M, Qv Y (2007) Azoreductase from Rhodobacter sphaeroides AS1.1737 is a flavodoxin that also functions as nitroreductase and flavin mononucleotide reductase. Appl Microbiol Biotechnol 76:1271–1279
Nicholas KB, Nicholas HB, Jr, Deerfield DW (1997) GeneDoc: analysis and visualization of genetic variation. EMBnet News 4:1–4
Paterson ES, Boucher SE, Lambert IB (2002) Regulation of the nfsA gene in Escherichia coli by SoxS. J Bacteriol 184:51–58
Patridge EV, Ferry JG (2006) WrbA from Escherichia coli and Archaeoglobus fulgidus is an NAD(P)H:quinone oxidoreductase. J Bacteriol 188:3498–3506
Prestera T, Prochaska HJ, Talalay P (1992) Inhibition of NAD(P)H:(quinone-acceptor) oxidoreductase by cibacron blue and related anthraquinone dyes: a structure-activity study. Biochemistry 31:824–833
Ross D, Siegel D (2004) NAD(P)H:quinone oxidoreductase 1 (NQO1, DT-diaphorase), functions and pharmacogenetics. Methods Enzymol 382:115–144
Russ R, Rau J, Stolz A (2000) The function of cytoplasmic flavin reductases in the reduction of azo dyes by bacteria. Appl Environ Microbiol 66:1429–1434
Soballe B, Poole RK (1999) Microbial ubiquinones: multiple roles in respiration, gene regulation and oxidative stress management. Microbiology 145:1817–1830
Soballe B, Poole RK (2000) Ubiquinone limits oxidative stress in Escherichia coli. Microbiology 146:787–796
Sollner S, Nebauer R, Ehammer H, Prem A, Deller S, Palfey BA, Daum G, Macheroux P (2007) Lot6p from Saccharomyces cerevisiae is a FMN-dependent reductase with a potential role in quinone detoxification. FEBS J 274:1328–1339
Stolz A (2001) Basic and applied aspects in the microbial degradation of azo dyes. Appl Microbiol Biotechnol 56:69–80
Wang G, Maier RJ (2004) An NADPH quinone reductase of Helicobacter pylori plays an important role in oxidative stress resistance and host colonization. Infect Immun 72:1391–1396
Yan B, Zhou J, Wang J, Du C, Hou H, Song Z, Bao Y (2004) Expression and characteristics of the gene encoding azoreductase from Rhodobacter sphaeroides AS1.1737. FEMS Microbiol Lett 236:129–136
Zenno S, Koike H, Kumar AN, Jayaraman R, Tanokura M, Saigo K (1996) Biochemical characterization of NfsA, the Escherichia coli major nitroreductase exhibiting a high amino acid sequence homology to Frp, a Vibrio harveyi flavin oxidoreductase. J Bacteriol 178:4508–4514
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, G., Zhou, J., Jin, R. et al. Enhancing survival of Escherichia coli by expression of azoreductase AZR possessing quinone reductase activity. Appl Microbiol Biotechnol 80, 409–416 (2008). https://doi.org/10.1007/s00253-008-1555-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1555-6