Abstract
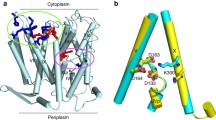
A recent molecular dynamics study questioned the protonation state and physiological role of aspartate 127 (D127) of E. coli porin OmpF. To address that question we isolated two OmpF mutants with D127 either neutralized (D127N) or replaced by a positively charged lysine (D127K). The charge state of the residue at position 127 has clear effects on both conductance and selectivity. The D127K but not the D127N mutant expresses resilient conductance and selectivity fluctuations. These fluctuations reflect, we think, either changes in the ionization state of K127 and/or transitions between unstable subconformations as induced by the electrostatic repulsion between two positively charged residues, K127 and the nearby R167. Our results slightly favor the view that in WT OmpF residue D127 is deprotonated. As for the role of D127 in OmpF functionality, the gating of both mutants shows very similar sensitivity toward voltage as WT OmpF. Moreover, the current fluctuations of the D127K mutant were observed also in the absence of an applied electric field. We therefore dismiss D127 as a key residue in the control mechanism of the voltage-dependent gating of OmpF.







Similar content being viewed by others
References
Alcaraz A, Nestorovich EM, Aguilella-Arzo M, Aguilella VM, Bezrukov SM (2004) Salting out the ionic selectivity of a wide channel: the asymmetry of OmpF. Biophys J 87:943–957
Bainbridge G, Gocke L, Lakey JH (1998) Voltage gating is a fundamental feature of porin and toxin β-barrel membrane channels. FEBS Lett 431:305–308
Baslé A, Qutub R, Mehrazin M, Wibbenmeyer J, Delcour AH (2004) Deletions of single extracellular loops affect pH sensitivity, but not voltage dependence, of the Escherichia coli porin OmpF. Protein Eng 17:665–672
Bredin J, Saint N, Malléa M, Dé E, Molle G, Pagès J-M, Simonet V (2002) Alteration of pore properties of Escherichia coli OmpF induced by mutation of key residues in anti-loop 3 region. Biochem J 363:521–528
Cowan SW, Schirmer T, Rummel G, Steiert M, Ghosh R, Pauptit RA, Jansonius JN, Rosenbusch JP (1992) Crystal structures explain functional properties of two E. Coli porins. Nature 358:727–733
Cymes GD, Ni Y, Grosman C (2005) Probing ion channel pores one proton at a time. Nature 438:975–980
Danelon C, Suenaga A, Winterhalter M, Yamato L (2003) Molecular origin of the cation selectivity in OmpF porin: single channel conductances vs. free energy calculation. Biophys Chem 104:591–603
Delcour AH (2003) Solute uptake through general porins. Front Biosci 8:1055–1071
Eisenberg RS (1996a) Computing the field in proteins and channels. J Membr Biol 150:1–25
Eisenberg RS (1996b) Atomic biology, electrostatics and ionic channels. In: Elber R (eds) New developments and theoretical studies of proteins, World Scientific, Philadelphia, pp 269–357
Im W, Roux B (2002a) Ions and counterions in a biological channel a molecular dynamics simulation of OmpF porin: from Escherichia coli in an explicit membrane with 1 M KCl aqueous salt solution. J Mol Biol 319:1177–1197
Im W, Roux B (2002b) Ion permeation and selectivity of OmpF porin: a theoretical study based on molecular dynamics, Brownian dynamics, and continuum electrodiffusion theory. J Mol Biol 322:851–869
Karshikoff A, Spassov V, Cowan SW, Ladenstein R, Schirmer T (1994) Electrostatic properties of two porin channels from Escherichia coli. J Mol Biol 240:372–384
Klebba PE, Newton SMC (1998) Mechanisms of solute transport through outer membrane porins: burning down the house. Curr Opion Microbiol 1:238–248
Liu N, Delcour AH (1998) The spontaneous gating activity of OmpC porin is affected by mutations of a putative hydrogen bond network or of a salt bridge between the L3 loop and the barrel. Protein Eng 11:797–802
Liu N, Samartzidou H, Lee KW, Briggs JM, Delcour AH (2000) Effects of pore mutations and permeant ion concentration on the spontaneous gating activity of OmpC porin. Protein Eng 13:491–500
Miedema H, Meter-Arkema A, Wierenga J, Tang J, Eisenberg B, Nonner W, Hektor H, Gillespie D, Meijberg W (2004) Permeation properties of an engineered bacterial OmpF porin containing the EEEE-locus of Ca2+ channels. Biophys J 87:3137–3147
Nestorovich EM, Rostovtseva TK, Bezrukov SM (2003) Residue ionization and ion transport through OmpF channels. Biophys J 85:3718–3729
Phale PS, Schirmer T, Prilipov A, Lou K-L, Hardmeyer A, Rosenbusch JP (1997) Voltage gating of Escherichia coli porin channels: role of the constriction zone. Proc Natl Acad Sci USA 94:6741–6745
Phale PS, Philippsen A, Widmer C, Pahe VP, Rosenbusch JP, Schirmer T (2001) Role of charged residues at the OmpF porin channel constriction probed by mutagenesis and simulation. Biochemistry 40:6319–6325
Philippsen A, Im W, Engel A, Schirmer T, Roux B, Müller DJ (2002) Imaging the electrostatic potential of transmembrane channels: atomic probe microscopy of OmpF porin. Biophys J 82:1667–1676
Robertson KM, Tieleman DP (2002) Molecular basis of voltage gating of OmpF porin. Biochem Cell Biol 80:517–523
Saint N, Lou K-L, Widmer C, Luckey M, Schirmer T, Rosenbusch JP (1996) Structural and functional characterization of OmpF porin mutants selected for larger pore size. J Biol Chem 271:20676–20680
Saxena K, Drosou V, Maier E, Benz R, Ludwig B (1999) Ion selectivity reversal and induction of voltage-gating by site-directed mutations in the Paracoccus denitrificans porin. Biochemistry 38:2206–2212
Schindler H, Rosenbusch JP (1978) Matrix protein from Escherichia coli outer membranes forms voltage-controlled channels in lipid bilayers. Proc Natl Acad Sci USA 75:3751–3755
Schirmer T (1998) General and specific porins from bacterial outer membranes. J Struct Biol 121:101–109
Schirmer T, Phale PS (1999) Brownian dynamics simulation of ion flow through porin channels. J Mol Biol 294:1159–1167
Schulz GE (2002) The structure of bacterial outer membrane proteins. Biochem Biophys Acta 1565:308–317
Tieleman DP, Berendsen HJC (1998) A molecular dynamics study of the pores formed by Escherichia coli OmpF porin in a fully hydrated plamitoyloleoylphosphatidylcholine bilayer. Biophys J 74:2786–2801
Todt JC, McGroarty EJ (1992) Involvement of Histidine−21 in the pH-induced switch in porin channel size. Biochemistry 31:10479–10482
Todt JC, Roqueand WJ, McGroarty EJ (1992) Effects of pH on bacterial porin function. Biochemistry 31:10471–10478
Van Gelder PN, Saint N, Phale PS, Eppens EF, Prilipov A, van Boxtel R, Rosenbusch JP (1997) Voltage sensing in the PhoE and OmpF outer membrane porins of Escherichia coli: role of charged residues. J Mol Biol 269:468–472
Varma S, Jakobsson E (2004) Ionization states of residues in OmpF and mutants: effects of dielectric constant and interactions between residues and ionic strength. Biophys J 86:690–704
Varma S, Chiu S-W, Jakobsson E (2006) The influence of amino acid protonation states on molecular dynamics simulations of the bacterial porin OmpF. Biophys J 90:112–123
Vrouenraets M, Wierenga J, Meijberg W, Miedema H (2006) Chemical modification of the bacterial porin OmpF: gain of selectivity by volume reduction. Biophys J 90:1202–1211
Acknowledgment
This research is supported by NanoNed, a nanotechnology program of the Dutch Ministry of Economic Affairs.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miedema, H., Vrouenraets, M., Wierenga, J. et al. Conductance and selectivity fluctuations in D127 mutants of the bacterial porin OmpF. Eur Biophys J 36, 13–22 (2006). https://doi.org/10.1007/s00249-006-0084-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-006-0084-4