Abstracts
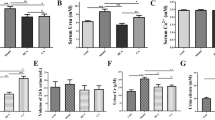
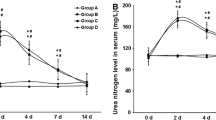
Our aim was to develop a stone-forming animal model involving renal tubular injury using a cyclooxygenase 2 selective inhibitor. Male Sprague-Dawley rats fed chow containing 3% sodium oxalate with or without 100 mg/kg celecoxib were compared to animals fed normal chow. Rats were killed after 2 or 4 weeks and the kidneys were harvested for morphological examination. Collections of 24-h urine were made before kidney harvest. After 2 weeks only a few crystals were observed in rats that received oxalate and celecoxib, but after 4 weeks more crystals were observed at the renal papilla than in rats that received only oxalate. Few crystals were found in rats fed normal chow with or without celecoxib. The urinary activities of gamma-glutamyl transpeptidase (GGT) were increased by celecoxib administration whereas creatinine clearance rates were unchanged. In rats fed oxalate, urinary oxalate excretion increased, but calcium excretion decreased. This model using a cyclooxygenase 2 selective inhibitor is a useful stone forming animal model involving mild renal tubular injury together with mild hyperoxaluria.



Similar content being viewed by others
References
Kahn SR, Hackett RL (1985) Calcium oxalate urolithiasis in the rat: is it a model for human stone disease? A review of recent literature. Scanning Elect Microsc 1985: 759
Kumar S, Sigmon D, Carpenter B, Khan S, Scheid C, Menon M et al. (1991) A new model of nephrolithiasis involving tubular dysfunction/injury. J Urol 146: 1384
Kwak C, Kim HK, Kim EC, Choi MS, Kim HH (2003) Urinary oxalate levels and the enteric bacterium Oxalobacter formigenes in patients with calcium oxalate urolithiasis. Eur Urol 44: 475
Kleinschmidt K, Mahlmann A, Hautmann R (1993) Anaerobic oxalate-degrading bacteria in the gut decrease faecal and urinary oxalate concentrations in stone formers. In: Ryall R, Basis R, Marshall VR, Rofe AM, Smith LH, Walker VR (eds) Urolithiasis 2. Plenum, New York, p 439
Campieri C, Campieri M, Bertuzzi V, Swennen E, Matteuzzi D, De Simone C et al. (2001) Reduction of oxaluria after an oral course of lactic acid bacteria at high concentration. Kidney Int 60: 1097
Kajander EO, Ciftcioglu N (1998) Nanobacteria: an alternative mechanism for pathogenic intra- and extracellular calcification and stone formation. Proc Natl Acad Sci U S A 95: 8274
Szasz G (1969) A kinetic photometric method for serum gamma-glutamyl transpeptidase. Clin Chem Acta 15: 124
Stirling JL (1969) Beta galactosidase, beta-glucosidase and N-acetyl-beta-glucosaminidase in human kidney. Clin Chim Acta 24: 189
Chiriboga J (1966) Purification and properties of oxalic acid oxidase. Arch Biochem Biophys 116: 516
Toftegoarrd NT (1976) A method for enzymatic determination of citrate in serum and urine. Scand J Clin Lab Invest 36: 513
Cohen SA, Sideman L (1979) Modification of the o-cresolphthalein complexone method for determining calcium. Clin Chem 25: 1519
Jaffe M (1886) Uber den Niederschlag, welchen Picrinsaure in normalen Harn erzeugt und über eine neue Reaction des Keratinins. Hoppe Zeylersz Physiol Chem 10: 391
Lyon ES, Borden TA, Vermeulen CW (1966) Experimental oxalate lithiasis produced with ethylene glycol. Invest Urol 4: 143
McIntosh GH, Belling GB, Bulman FH (1979) Experimental oxalate urolith formation in rats. Aust J Exp Biol Med Sci 57: 251
Randall A (1940) The etiology of primary renal calculus. Int Abs Surg 71: 209
Gill WB, Ruggiero K, Straus FH (1979) Crystallization studies in an urothelial-lined living test tube (the catheterized female rat bladder). I. Calcium oxalate crystal adhesion to the chemically injured rat bladder. Invest Urol 17: 257
Khan SR, Cockrell CA, Finlayson B et al. (1984) Crystal retention by injured urothelium of the rat urinary bladder. J Urol 132: 153
Khan SR, Finlayson B, Hackett RL (1982) Experimental calcium oxalate nephrolithiasis in the rat. Role of the renal papilla. Am J Pathol 107: 59
Tessitore N, Ortalda V, Fabris A et al. (1985) Renal acidification defects in patients with recurrent calcium nephrolithiasis. Nephron 41: 325
Sutton RA, Walker VR (1980) Responses to hydrochlorothiazide and acetazolamide in patients with calcium stones. Evidence suggesting a defect in renal tubular function. N Engl J Med 302: 709
Lee JB, Crowshaw K, Takman BH, Attrep KA (1967) The identification of prostag-landins E2, F2a and A2 from rabbit kidney medulla. Biochem J 105: 1251
Daniels EG, Hinman JW, Leach BE, Muirhead EE (1967) Identification of prostaglandin E2 as the principal vasodepressor lipid of rabbit renal medulla. Nature 215: 1298
Vane JR (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature 231: 232
Dunn MD, Hood VL (1977) Prostaglandins and the kidney: an editorial review. Am J Physiol 233: 169
Herschman HR (1996) Prostaglandin synthase 2. Biochim Biophys Acta 1299: 125
Dubois RN, Abramson SB, Crofford L, Gupta RA, Simon LS, Van de Putte LB, Lipsky PE (1998) Cyclooxygenase in biology and disease. FASEB J 12: 1063
Laudanno OM, Cesolari JA, Esnarriaga J, Rista L, Piombo G, Rocaspana A (2001) Gastrointestinal damage induced by celecoxib and rofecoxib in rats. Dig Dis Sci 46: 779
Chahwala SB, Harpur ES (1983) Gentamicin-induced hypercalciuria in the rat. Acta Pharmacol Toxicol 53: 358
Farell G, Huang E, Kim SY, Horstkorte R, Lieske JC (2004) Modulation of proliferating renal epithelial cell affinity for calcium oxalate monohydrate crystals. J Am Soc Nephrol 15: 3052
Bosomworth MP, Aparicio SR, Hay AW (1999) Urine N-acetyl-{beta}-D-glucosaminidase—a marker of tubular damage? Nephrol Dial Transplant 14: 620
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jeong, B.C., Park, M.Y., Kwak, C. et al. An animal model of calcium oxalate urolithiasis based on a cyclooxygenase 2 selective inhibitor. Urol Res 33, 453–459 (2005). https://doi.org/10.1007/s00240-005-0507-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-005-0507-1