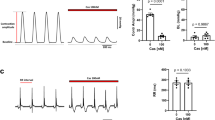
Abstract
ClC chloride channels play essential roles in membrane excitability and maintenance of osmotic balance. Despite the recent crystallization of two bacterial ClC-like proteins, the gating mechanism for these channels remains unclear. In this study we tested scorpion venom for the presence of novel peptide inhibitors of ClC channels, which might be useful tools for dissecting the mechanisms underlying ClC channel gating. Recently, it has been shown that a peptide component of venom from the scorpion L. quinquestriatus hebraeus inhibits the CFTR chloride channel from the intracellular side. Using two-electrode voltage clamp we studied the effect of scorpion venom on ClC-0, -1, and -2, and found both dose- and voltage-dependent inhibition only of ClC-2. Comparison of voltage-dependence of inhibition by venom to that of known pore blockers revealed opposite voltage dependencies, suggesting different mechanisms of inhibition. Kinetic data show that venom induced slower activation kinetics compared to pre-venom records, suggesting that the active component(s) of venom may function as a gating modifier at ClC-2. Trypsinization abolished the inhibitory activity of venom, suggesting that the component(s) of scorpion venom that inhibits ClC-2 is a peptide.







Similar content being viewed by others
Abbreviations
- 9-AC:
-
anthracene-9-carboxylic acid
- CF:
-
cystic fibrosis
- CFTR:
-
cystic fibrosis transmembrane conductance regulator;
- CITx:
-
chlorotoxin
- CPB:
-
2-(p-chlorophenoxy) butyric acid
- CPP:
-
2-(p- chlorophenoxy) proprionic acid
- DIDS:
-
4′4′-Diisothiocyano-2,2′-Stilbene disulfonate
- DMSO:
-
dimethyl sulfoxide;
- DNDS:
-
4,4′-Nitrostilbene- 2,2′-disulfonate
- DPC:
-
diphenylamine 2-carboxylic Acid
- NPPB:
-
5-nitro-2-(3-phenylpropylamino) benzoic acid
- EGTA:
-
ethylene glycol-bis(β-aminoethyl ether)- N,N,N′,N′-tetraacetic acid;
- VSTx:
-
voltage sensing toxin.
References
Adjadlj E., Naudat V., Quiniou E., Wouters D, Sautiere P., Craescu C.T. 1997. Solution structure of Lqh-8/6, a toxin-like peptide from a scorpion venom—structural heterogeneity induced by proline cis/trans isomerization. Eur. J. Biochem. 246:218–227
Aromataris, E.C., Rychkov, G.V., Bennetts, B., Hughes, B.P., Bretag, A.H., Roberts, M.L. 2001. Fast and slow gating of ClC-1: differential effects of 2-(4-chlorophenoxy) propionic acid and dominant negative mutations. Mol. Pharmacol. 60:200–208
Bauer C.K., Steinmeyer K., Schwarz J.R., Jentsch T.J. 1991. Completely functional double-barreled chloride channel expressed from a single Torpedo cDNA. 88:11052–11056
Chen T.Y. 1998. Extracellular zinc ion inhibits ClC-0 chloride channels by facilitating slow gating. J. Gen. Physiol. 12:715–726
Cuppoletti, J., Tewari, K., Sherry, A.M., Kupert, F.Y., Malinoska, D.H. 2001. ClC-2 Cl- channels in lung epithelia: activation by arachidonic acid, amidation, and acid-activated omeprazole. Am. J. Physiol. 281:C46–C54
DeBin J.A., Strichartz G.R. 1991. Chloride channel inhibition by the venom of the scorpion Leiurus quinquestriatus. Toxicon 29:1403–1408
DeBin J.A., Maggio J.E., Strichartz G.R. 1993. Purification and characterization of chlorotoxin, a chloride channel ligand from the venom of the scorpion. Am. J. Physiol. 264:C361–C369
de Santiago, J.A., Nehrke, K., Arreola, J. 2005. Quantitative analysis of the voltage-dependent gating of mouse parotid ClC-2 chloride channel. J. Gen. Physiol. 126:591–603.
Devuyst O., Guggino W.B. 2002. Chloride channels in the kidney: lessons from knockout animals. Am. J. Physiol. 283:F1176–F1191
Doyle O., D.A., Morais Cabral J., Pfuetzner R.A., Kuo A., Gulbis J.M., Cohen S.L., Chait B.T., MacKinnon R. 1998. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280:69–77
Dutzler R., Campbell E.B., MacKinnon R. 2003. Gating the selectivity filter in ClC chloride channels. Science 300:108–112
Dutzler, R..Campbell E.B., Cadene M., Chait B.J., MacKinnon R. 2002. X-ray structure of a ClC chloride channel at 3.0 A reveals the molecular basis of anion selectivity. Nature 415:287–294
Fahlke C., Yu H.T., Beck C., Rhodes T.H., George Jr. A.L. 1997. Pore-forming segments in voltage-gated chloride channels. Nature 390:529–532
Fuller M.F., Zhang Z.R., Cui G., Kubanek J., McCarty N.A. 2004. Inhibition of CFTR channels by a peptide toxin of scorpion venom. Am. J. Physiol. 287:C1328–C1341
Furukawa, T., Ogura, T., Katayama, Y., Hiraoka, M. 1998. Characteristics of rabbit ClC-2 current expressed in Xenopus oocytes and its contribution to volume regulation. Am. J. Physiol. 274:C500–C512
Gross A., MacKinnon R. 1996. Agitoxin footprinting the shaker potassium channel pore. Neuron 16:399–406
Gyomorey K., Garami F., Galley K., Rommens J.M., Bear C.E. 2000. Non-CFTR chloride channels likely contribute to secretion in the murine small intestine. Am. J. Physiol. 219:CL787–C1794
Haug K., Warnstedt M., Alekov A.K., Sander T., Ramirez A, Poser B., Maljevic S., Hebeisen S., Kubisch C., Rebstock J., Horvath S., Hallmann K., Dullinger J.S., Rau B., Haverkamp F., Beyenburg S., Schulz H., Janz D., Giese B., Muller-Newen G., Propping P., Elger C.E., Fahlke C., Lerche H., Heils A. 2003. Mutations: in ClCN2 encoding a voltage-gated chloride channel are associated with idiopathic generalized epilepsies. Nat. Genet. 33:527–532
Hechenberger M., Schwappach B., Fischer W.N., Frommer W.B., Jentsch T.J., Steinmeyer K. 1996. A family of putative chloride channels from Arabidopsis and functional complementation of a yeast strain with a ClC gene disruption. J. Biol. Chem. 271:33632–33638
Jentsch T.J., Stein V, Weinreich F., Zdebik A.A. 2002. Molecular structure and physiological function of chloride channels. Physiol. Rev. 82: 503–568
Jordt S.E., Jentsch T.J. 1997. Molecular dissection of gating in the ClC-2 chloride channel. EMBO J 16:1582–1592
Koch M.C., Steinmeyer K., Lorenz C., Ricker K., Wolf F., Otto M., Zoll B., Lehmann-Horn F., Grzeschik K.H., Jentsch T.J. 1992. The skeletal muscle chloride channel in dominant and recessive human myotonia. Science 257: 797–800
Lee HC, Wang J.M, paq Swartz K.J. 2003. Interaction between extracellular Hanatoxin and the resting conformation of the voltage-sensor paddle in Kv channels. Neuron 40:527–536
Li-Smerin Y., Swartz K.J. 1998. Gating modifier toxins reveal a conserved structural motif in voltage-gated Ca2+ and K+ channels. Proc. Natl. Acad. Sci. USA 95:8585–8589
Lipecka J., Ball M., Thomas A., Fanen P., Edelman A., Fritsch J. 2002. Distribution of ClC-2 chloride channel in rat and human epithelial tissues. Am. J. Physiol. 282:C805–C816
Lloyd, S.E., Pearce, S.H., Fisher, S.E., Steinmeyer, K., Schwappach, B., Scheinman, S.J., Harding, B., Bolino, A., Devoto, M., Goodyer, P., Rigden, S.P., Wrong, O., Jentsch, T.J., 24. Craig, I.W., Thakker R.V. 1996. A common molecular basis for three inherited kidney stone diseases. Nature 379:445–449
Machaca, K., Hartzell H.C. 1998. Asymmetrical distribution of Ca2+-activated Cl− channels in Xenopus oocytes. Biophys. J. 74:1286–1295, 1998. Erratum in: Biophys. J. 74:3313
Maduke M., Miller C., Mindell J.A. 2000. A decade of CLC chloride channels: structure, mechanism, and many unsettled questions. Annu. Rev. Biophys. Biomol. Struct. 29:411–438
Maertens C., Wei L., Tytgat J., Droogmans G., Nilius B. 2000. Chlorotoxin does not inhibit volume-regulated, calcium-activated and cyclic AMP-activated chloride channels. Br. J. Pharmacol. 129:791–801
McCarty N.A., Zhang Z.R. 2001. Identification of a region of strong discrimination in the pore of CFTR. Am. J. Physiol. 281:L852–L867
McDonough S.I. 2003. Peptide toxin inhibition of voltage-gated calcium channels: Selectivity and Mechanisms In: Calcuim Channel Pharmacology, ed. McDonough SI. Plenum, New York
McDonough S.I., Boland L.M., Mintz I.M., Bean B.P. 2002. Interactions among toxins that inhibit N-type and P-type calcium channels. J. Gen. Physiol. 119:313–328
McDonough S.I., Lampe R.A., Keith R.A., Bean B.P. 1997. Voltage-dependent inhibition of N- and P-type calcium channels by the peptide toxin omega-grammotoxin-SIA. Mol. Pharmacol. 52:1095–1104
Murray C.B., Chu S., Zeitlin P.L. 1996. Gestational and tissue-specific regulation of ClC-2 chloride channel expression. Am. J. Physiol. 21:L829–L837
Naranjo D., Miller C. 1996. A strongly interacting pair of residues on the contact surface of charybdotoxin and a Shaker K+ channel. Neuron 16:123–130
Niemeyer M.I., Cid L.P., Zuniga L., Catalan M., Sepulveda F.V. 2003. A conserved pore-lining glutamate as a voltage- and chloride-dependent gate in the ClC-2 chloride channel. J. Physiol. 553:873–879
Norton R.S., Pallaghy P.K. 1998. The cystine knot structure of ion channel toxins and related polypeptides. Toxicon 36:1573–1583
Pusch M., Accardi A., Liantonio A., Ferrera L., De Luca A., Camerino D.C., Conti F. 2001. Mechanism of block of single protopores of the Torpedo chloride channel ClC-0 by 2-(p-chlorophenoxy) butyric acid (CPB). J. Gen. Physiol. 118:45–62
Pusch M., Liantonio A., Bertorello L., Accardi A., De Luca A., Pierno S., Tortorella V., Camerino D.C. 2000. Pharmacological characterization of chloride channels belonging to the ClC family by the use of chiral Clofibric acid derivatives. Mol. Pharmacol. 58:498–507
Pusch M., Jordt S.E., Stein V., Jentsch T.J. 1999. Chloride dependence of hyperpolarization-activated chloride channel gates. J. Physiol. 5l5:341–353
Qu Z., Hartzell H.C. 2000. Anion permeation in Ca2+-activated Cl− channels. J. Gen. Physiol. 116:825–844
Ruta V., Jiang Y., Lee A., Chen J., MacKinnon R. 2003. Functional analysis of an archaebacterial voltage-dependent K+ channel. Nature 422:180–185
Sherry A.M., Malinowska D.H., Morris R.E., Ciraolo G.M., Cuppoletti J. 2004. Localization of ClC-2 Cl- channels in rabbit gastric mucosa. Am. J. Physiol. 280:CL599–CL606
Sidach S.S., Mintz I.M. 2002. Kurtoxin, a gating modifier of neuronal high- and low-threshold Ca2+ channels. J.Neurosci. 22:2023–2034
Simon D.B., Bindra R.S., Mansfield T.A., Nelson-Williams C., Mendonca E., Stone R., Schurman S., Nayir A., Alpay H., Bakkaloglu A., Rodriguez-Soriano J., Morales J.M., Sanjadv S.A., Taylor C.M., Pilz D., Brem A., Trachtman H., Griswold W., Richard G.A., John E., Lifton R.P. 1997. Mutations in the chloride channel gene, CLCNKB, cause Barrier’s syndrome type III. Nat. Genet. 17:171–178
Steinmeyer K., Klocke R., Ortland C., Gronemeier M., Jockusch H., Grander S., Jentsch T.J. 1991. Inactivation of muscle chloride channel by transposon insertion in myotonic mice. Nature 354:304–308
Stobrawa S.M., Breiderhoff T., Takamori S., Engel D., Schweizer M., Zdebik A.A., Bosl M.R., Ruether K., Jahn H., Draguhn A., Jahn R., Jentsch T.J. 2001. Disruption of ClC-3, a chloride channel expressed on synaptic vesicles, leads to a loss of the hippocampus. Neuron 29:185–196
Swartz K.J., MacKinnon R. 1997. Hanatoxin modifies the gating of a voltage-dependent K+ channel through multiple binding sites. Neuron 18:665–673
Swartz K.J., MacKinnon R. 1997. Mapping the receptor site for Hanatoxin, a gating modifier of voltage-dependent K+ channels. Neuron 18:675–682
Zdebik A.A., Cuffe J.E., Bertog M, Korbmacher C, Jentsch T.J. 2004. Additional disruption of the ClC-2 Cl− channel does not exacerbate the Cystic Fibrosis phenotype of Cystic Fibrosis Transmembrane Conductance Regulator mouse models. J. Biol. Chem. 219:22276–22283
Zuniga L., Niemeyer M.I., Varela D., Catalan M., Cid LP., Sepulveda F.V. 2004. The voltage-dependent ClC-2 chloride channel has a dual gating mechanism. J. Physiol. 555:671–682
Acknowledgements
We thank Kate Hubbard for contributing to initial experiments. We thank Dr. Steve Harvey and Dr. Robert Lee for critically reading an early version of the manuscript. This work was supported by the National Institute of Health (NIH DK 56481 and DK 066409). During the performance of this work, N.A.M. was an Established Investigator of the American Heart Association.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thompson, C., Fields, D., Olivetti, P. et al. Inhibition of ClC-2 Chloride Channels by a Peptide Component or Components of Scorpion Venom. J Membrane Biol 208, 65–76 (2005). https://doi.org/10.1007/s00232-005-0818-8
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s00232-005-0818-8