Abstract
Objective: Deflazacort, a synthetic oxazoline derivative of prednisolone, has been suggested as having major advantages over other glucocorticoids, as it is claimed to cause fewer adverse effects at equivalent antiinflammatory potency. The assumed equipotency ratio of deflazacort versus other glucocorticoids is critical for this assumption.
Methods:
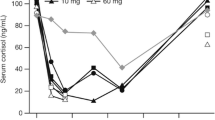
In a randomized cross-over study we compared the acute effects of deflazacort and prednisolone on serum cortisol, osteocalcin, insulin and blood cells (eosinophils and lymphocytes) in normal subjects. On seven occasions separated by a wash out period ≥ 1 week all participants received placebo, prednisolone (8 mg, 20 mg, 40 mg) and deflazacort (12 mg, 30 mg, 60 mg). The medication was given orally at 20.00 h as a single dose. Blood was collected at 8.00 h before and after each medication. Log (dose) response relationships were calculated and were used to compare the drugs.
Results:
The following equipotent dose ratios (mg deflazacort: mg prednisolone) were found: osteocalcin suppression 1.54, cortisol suppression 2.27, suppression of eosinophils 1.14 and lymphocytes 2.77. As parallelism between regression curves was rejected, equipotency could not be calculated for insulin. In 3 subjects even the highest dose of deflazacort failed to suppress serum cortisol.
Conclusion:
Our study highlights the difficulties of establishing equipotency ratios for glucocorticoids. It casts doubts on the generally assumed equipotency dose ratio of deflazacort vs prednisolone, as both for cortisol and lymphocytes the 95% CI was > 1.2. Thus, reduced adverse effects during deflazacort therapy may be a consequence of lower effective glucocorticoid dosage.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 11 December 1995 /Accepted in revised form: 16 March 1996
Rights and permissions
About this article
Cite this article
Babadjanova, G., Allolio, B., Vollmer, M. et al. Comparison of the pharmacodynamic effects of deflazacort and prednisolone in healthy subjects. E J Clin Pharmacol 51, 53–57 (1996). https://doi.org/10.1007/s002280050160
Issue Date:
DOI: https://doi.org/10.1007/s002280050160