Abstract
Purpose
Previous studies have shown that tetrahydrocannabinol (THC), the main psychoactive component of cannabis, can impair cognitive abilities. There is also some evidence that cannabidiol (CBD), the most abundant non-intoxicating constituent of cannabis, can attenuate these effects. The purpose of this study was to investigate the effects of THC:CBD oromucosal spray (with equal parts THC and CBD) on cognition compared with control conditions in human studies.
Methods
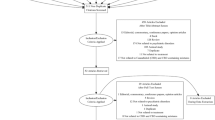
A systematic literature search was performed on four major bibliographic databases. Studies were included in the present review if they evaluated the cognitive effects of THC:CBD oromucosal spray compared with a control condition.
Results
Ten studies were identified (7 on patients with multiple sclerosis, 1 on those with Huntington, and 2 on healthy volunteers) with 510 participants in total. There was considerable heterogeneity among the studies in terms of dose and duration of administration. All studies have used an equal or nearly equal dose of THC and CBD.
Conclusions
Although the results across studies were somewhat inconsistent, most evidence revealed that there is no significant difference between THC:CBD oromucosal spray and control treatments in terms of cognitive outcomes. However, more trials are needed with longer follow-up periods, and dose considerations, particularly comparing lower and higher doses of the spray.


Similar content being viewed by others
Data availability
All data in relation to this study are presented in this manuscript.
References
Crean RD, Crane NA, Mason BJ (2011) An evidence based review of acute and long-term effects of cannabis use on executive cognitive functions. J Addict Med 5(1):1
Volkow ND, Swanson JM, Evins AE, DeLisi LE, Meier MH, Gonzalez R, Bloomfield MA, Curran HV, Baler R (2016) Effects of cannabis use on human behavior, including cognition, motivation, and psychosis: a review. JAMA Psychiat 73(3):292–297
Wright MJ Jr, Vandewater SA, Taffe MA (2013) Cannabidiol attenuates deficits of visuospatial associative memory induced by Δ9tetrahydrocannabinol. Br J Pharmacol 170(7):1365–1373
Curran VH, Brignell C, Fletcher S, Middleton P, Henry J (2002) Cognitive and subjective dose-response effects of acute oral Δ9-tetrahydrocannabinol (THC) in infrequent cannabis users. Psychopharmacology 164(1):61–70
Schoedel AK, Jane Harrison S (2012) Subjective and physiological effects of oromucosal sprays containing cannabinoids (nabiximols): potentials and limitations for psychosis research. Curr Pharm Des 18(32):5008–5014
Castelli L, Prosperini L, Pozzilli C (2019) Balance worsening associated with nabiximols in multiple sclerosis. Mult Scler J 25(1):113–117
Boggs DL, Nguyen JD, Morgenson D, Taffe MA, Ranganathan M (2018) Clinical and preclinical evidence for functional interactions of cannabidiol and Δ9-tetrahydrocannabinol. Neuropsychopharmacology 43(1):142–154
Solowij N, Broyd SJ, Beale C, Prick J-A, Greenwood L-m, Van Hell H, Suo C, Galettis P, Pai N, Fu S (2018) Therapeutic effects of prolonged cannabidiol treatment on psychological symptoms and cognitive function in regular cannabis users: a pragmatic open-label clinical trial. Cannabis Cannabinoid Res 3(1):21–34
Roser P, Juckel G, Rentzsch J, Nadulski T, Gallinat J, Stadelmann AM (2008) Effects of acute oral Δ9-tetrahydrocannabinol and standardized cannabis extract on the auditory P300 event-related potential in healthy volunteers. Eur Neuropsychopharmacol 18(8):569–577
Woelfl T, Rohleder C, Mueller JK, Lange B, Reuter A, Schmidt AM, Koethe D, Hellmich M, Leweke FM (2020) Effects of cannabidiol and delta-9-tetrahydrocannabinol on emotion, cognition, and attention: a double-blind, placebo-controlled, randomized experimental trial in healthy volunteers. Front Psych 11:576877
Arkell TR, Lintzeris N, Kevin RC, Ramaekers JG, Vandrey R, Irwin C, Haber PS, McGregor IS (2019) Cannabidiol (CBD) content in vaporized cannabis does not prevent tetrahydrocannabinol (THC)-induced impairment of driving and cognition. Psychopharmacology 236(9):2713–2724
Alessandria G, Meli R, Infante MT, Vestito L, Capello E, Bandini F (2020) Long-term assessment of the cognitive effects of nabiximols in patients with multiple sclerosis: a pilot study. Clin Neurol Neurosurg 196:105990
Russo M, Rifici C, Sessa E, D’Aleo G, Bramanti P, Calabrò RS (2015) Sativex-induced neurobehavioral effects: causal or concausal? A practical advice! DARU J Pharm Sci 23(1):1–2
Wade DT, Robson P, House H, Makela P, Aram J (2003) A preliminary controlled study to determine whether whole-plant cannabis extracts can improve intractable neurogenic symptoms. Clin Rehabil 17(1):21–29
Nicholson AN, Turner C, Stone BM, Robson PJ (2004) Effect of Δ-9-tetrahydrocannabinol and cannabidiol on nocturnal sleep and early-morning behavior in young adults. J Clin Psychopharmacol 24(3):305–313
Schoedel KA, Chen N, Hilliard A, White L, Stott C, Russo E, Wright S, Guy G, Romach MK, Sellers EM (2011) A randomized, double-blind, placebo-controlled, crossover study to evaluate the subjective abuse potential and cognitive effects of nabiximols oromucosal spray in subjects with a history of recreational cannabis use. Hum Psychopharmacol Clin Exp 26(3):224–236
López-Sendón Moreno JL, García Caldentey J, Trigo Cubillo P, Ruiz Romero C, García Ribas G, Arias A, Alonso M, García de Yébenes MJ, Tolón RM, Galve-Roperh I (2016) A double-blind, randomized, cross-over, placebo-controlled, pilot trial with Sativex in Huntington’s disease. J Neurol 263(7):1390–1400
Wade DT, Makela P, Robson P, House H, Bateman C (2004) Do cannabis-based medicinal extracts have general or specific effects on symptoms in multiple sclerosis? A double-blind, randomized, placebo-controlled study on 160 patients. Mult Scler J 10(4):434–441
Rog DJ, Nurmikko TJ, Friede T, Young CA (2005) Randomized, controlled trial of cannabis-based medicine in central pain in multiple sclerosis. Neurology 65(6):812–819
Aragona M, Onesti E, Tomassini V, Conte A, Gupta S, Gilio F, Pantano P, Pozzilli C, Inghilleri M (2009) Psychopathological and cognitive effects of therapeutic cannabinoids in multiple sclerosis: a double-blind, placebo controlled, crossover study. Clin Neuropharmacol 32(1):41–47
Vachová M, Novotná A, Mares J, Taláb R, Fiedler J, Lauder H, Taylor L, Etges T, Wright S, Nováková I (2014) A multicentre, double-blind, randomised, parallel-group, placebo-controlled study of effect of long-term sativexand# 174; treatment on cognition and mood of patients with spasticity due to multiple sclerosis. J Multiple Sclerosis 1(2):1–8
Russo M, De Luca R, Torrisi M, Rifici C, Sessa E, Bramanti P, Naro A, Calabrò R (2016) Should we care about sativex-induced neurobehavioral effects? A 6-month follow-up study. Age (y) 42 (8.9):43–49
Cohen K, Weizman A, Weinstein A (2019) Positive and negative effects of cannabis and cannabinoids on health. Clin Pharmacol Ther 105(5):1139–1147
Karschner EL (2010) Pharmacodynamic and pharmacokinetic characterization of sativex and oral THC. University of Maryland, Baltimore
Desrosiers NA, Ramaekers JG, Chauchard E, Gorelick DA, Huestis MA (2015) Smoked cannabis’ psychomotor and neurocognitive effects in occasional and frequent smokers. J Anal Toxicol 39(4):251–261
Gage SH (2019) Cannabis and psychosis: triangulating the evidence. Lancet Psychiat 6(5):364–365
Hellemans KG, Wilcox J, Nino JN, Young M, McQuaid RJ (2019) Cannabis use, anxiety, and perceptions of risk among Canadian undergraduates: the moderating role of gender. Can J Addict 10(3):22–29
Bloomfield MA, Hindocha C, Green SF, Wall MB, Lees R, Petrilli K, Costello H, Ogunbiyi MO, Bossong MG, Freeman TP (2019) The neuropsychopharmacology of cannabis: a review of human imaging studies. Pharmacol Ther 195:132–161
Lim SY, Sharan S, Woo S (2020) Model‐based analysis of cannabidiol dose‐exposure relationship and bioavailability. Pharmacother: J Human Pharmacol Drug Ther 40(4):291–300
Romero-Sandoval EA, Kolano AL, Alvarado-Vázquez PA (2017) Cannabis and cannabinoids for chronic pain. Curr Rheumatol Rep 19(11):1–10
Vermersch P (2011) Sativex®(tetrahydrocannabinol+ cannabidiol), an endocannabinoid system modulator: basic features and main clinical data. Expert Rev Neurother 11(sup4):15–19
Rekand T (2014) THC: CBD spray and MS spasticity symptoms: data from latest studies. Eur Neurol 71(Suppl. 1):4–9
Nikravesh M, Jafari Z, Mehrpour M, Kazemi R, Shavaki YA, Hossienifar S, Azizi MP (2017) The paced auditory serial addition test for working memory assessment: psychometric properties. Med J Islam Repub Iran 31:61
Brooks JBB, Giraud VO, Saleh YJ, Rodrigues SJ, Daia LA, Fragoso YD (2011) Paced auditory serial addition test (PASAT): a very difficult test even for individuals with high intellectual capability. Arq Neuropsiquiatr 69:482–484
Velayudhan L, McGoohan K, Bhattacharyya S (2021) Safety and tolerability of natural and synthetic cannabinoids in adults aged over 50 years: a systematic review and meta-analysis. PLoS Med 18(3):e1003524
Morgan CJ, Schafer G, Freeman TP, Curran HV (2010) Impact of cannabidiol on the acute memory and psychotomimetic effects of smoked cannabis: naturalistic study. Br J Psychiatry 197(4):285–290
Cadden M, Arnett P (2015) Factors associated with employment status in individuals with multiple sclerosis. Int J MS care 17(6):284–291
Kronenberger WG, Henning SC, Ditmars AM, Roman AS, Pisoni DB (2018) Verbal learning and memory in prelingually deaf children with cochlear implants. Int J Audiol 57(10):746–754
Jiang JM, Seng EK, Zimmerman ME, Sliwinski M, Kim M, Lipton RB (2017) Evaluation of the reliability, validity, and predictive validity of the subscales of the perceived stress scale in older adults. J Alzheimers Dis 59(3):987–996
Terzaghi M, Sinforiani E, Zucchella C, Zambrelli E, Pasotti C, Rustioni V, Manni R (2008) Cognitive performance in REM sleep behaviour disorder: a possible early marker of neurodegenerative disease? Sleep Med 9(4):343–351
Horner MD, Teichner G, Kortte KB, Harvey RT (2002) Construct validity of the Babcock Story recall test. Appl Neuropsychol 9(2):114–116
Moran EE, Bressman SB, Ortega RA, Raymond D, Nichols WC, Palmese CA, Elango S, Swan M, Shanker V, Perera I (2021) Cognitive functioning of glucocerebrosidase (GBA) non-manifesting carriers. Front Neurol 12:635958
Ryazanskaya G, Khudyakova M, Buivolova O (2020) Automated analysis of verbal fluency task in healthy speakers: preliminary results. The Night Whites Language Workshop: The Fifth Saint Petersburg Winter Workshop on Experimental Studies of Speech and Language (Night Whites 2019), pp 91–91
Tan EJ, Neill E, Rossell SL (2015) Assessing the relationship between semantic processing and thought disorder symptoms in schizophrenia. J Int Neuropsychol Soc 21(8):629–638
Engell T, Lorås HW, Sigmundsson H (2020) Window view of nature after brief exercise improves choice reaction time and heart rate restoration. New Ideas Psychol 58:100781
Kretschmer V, Griefahn B, Schmidt K (2011) Bright light and night work: effects on selective and divided attention in elderly persons. Light Res Technol 43(4):473–486
Rigney U, Kimber S, Hindmarch I (1999) The effects of acute doses of standardized Ginkgo biloba extract on memory and psychomotor performance in volunteers. Phytother Res 13(5):408–415
Gerstenecker A, Martin R, Marson DC, Bashir K, Triebel KL (2016) Introducing demographic corrections for the 10/36 spatial recall test. Int J Geriatr Psychiatry 31(4):406–411
Pokryszko-Dragan A, Zagrajek M, Slotwinski K, Bilinska M, Gruszka E, Podemski R (2016) Event-related potentials and cognitive performance in multiple sclerosis patients with fatigue. Neurol Sci 37(9):1545–1556
Hu S, Shi J, Xiong W, Li W, Fang L, Feng H (2017) Oxiracetam or fastigial nucleus stimulation reduces cognitive injury at high altitude. Brain Behav 7(10):e00762
Mikicin M, Nowacka-Dobosz S, Mróz A, Kuk A, Zagórska-Pachucka A (2019) Are there correlations between attention, physical endurance and anthropometric parameters of athletes? Biomed Hum Kinet 11(1):103–109
Bauer B (2017) Perceptual averaging of line length: effects of concurrent digit memory load. Atten Percept Psychophys 79(8):2510–2522
Smith JA, Zhao W, Yu M, Rumfelt KE, Moorjani P, Ganna A, Dey AB, Lee J, Kardia SL (2020) Association between episodic memory and genetic risk factors for Alzheimer’s disease in south Asians from the longitudinal aging study in India–diagnostic assessment of dementia (LASI-DAD). J Am Geriatr Soc 68:S45–S53
Acknowledgements
This study was supported by Kurdistan University of Medical Sciences, Sanandaj, Iran (Grant ID: 1400/09/17).
Funding
This study was funded by Kurdistan University of Medical Sciences (Grant ID: 1400/09/17).
Author information
Authors and Affiliations
Contributions
EM, MG, MR, and HB designed the study and prepared the data. MA and PD led the drafting of the manuscript to which all authors contributed through editing and revision. All authors had access to the data sets and agreed to the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Motaghi, E., Ghasemi-Pirbaluti, M., Rashidi, M. et al. The effect of tetrahydrocannabinol:cannabidiol oromucosal spray on cognition: a systematic review. Eur J Clin Pharmacol 79, 371–381 (2023). https://doi.org/10.1007/s00228-023-03454-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-023-03454-y