Abstract
Background
Patients with atrial fibrillation (AF) who are on long-term direct oral anticoagulants (DOAC) with low anti-Xa or anti-IIa levels may be at higher risk of recurrent stroke. However, no prospective post-marketing study has investigated these DOAC plasma levels at the time of embolic stroke. The aim of this study was to assess the anti-Xa (rivaroxaban, apixaban) and anti-IIa (dabigatran) plasma levels in DOAC-treated AF patients at the time of acute embolic stroke.
Patients and methods
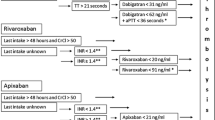
We prospectively identified 43 patients with AF on long-term DOAC who experienced embolic strokes. We compared the DOAC plasma levels of these patients with a control sample of 57 patients who tolerated long-term therapeutic dose DOAC therapy without any adverse event. DOAC levels were assessed with drug-specific anti-Xa chromogenic analysis (rivaroxaban, apixaban) and with Hemoclot Thrombin Inhibitor assay (dabigatran).
Results
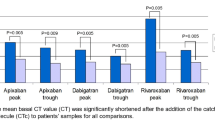
Dabigatran-treated patients with stroke had significantly lower anti-IIa levels when compared with the trough (40.7 ± 36.9 vs. 85.4 ± 57.2 ng/mL, p < 0.05) and peak samples of the controls (40.7 ± 36.9 vs. 138.8 ± 78.7 ng/mL, p < 0.001). Similarly, there were significantly lower anti-Xa levels in apixaban-treated patients with stroke compared to the trough control samples (72.4 ± 46.7 vs. 119.9 ± 81.7 ng/mL, p < 0.05), and in rivaroxaban- and apixaban-treated patients when compared to peak control samples (rivaroxaban: 42.7 ± 31.9 vs. 177.6 ± 38.6 ng/mL, p < 0.001; apixaban: 72.4 ± 46.7 vs. 210.9 ± 88.7 ng/mL, p < 0.001).
Conclusion
This observational study showed significantly lower anti-IIa and anti-Xa plasma levels in AF patients with embolic stroke compared to those who tolerated long-term therapeutic dose DOAC therapy.
Similar content being viewed by others
Data availability
All data are available at corresponding author upon reasonable request.
References
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, Pogue J, Reilly PA, Themeles E, Varrone J, Wang S, Alings M, Xavier D, Zhu J, Diaz R, Lewis BS, Darius H, Diener HC, Joyner CD, Wallentin L, Steering Committee RE-LY, Investigators (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361:1139–1151
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM, Investigators ROCKETAF (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:883–891
Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, Al-Khalidi HR, Ansell J, Atar D, Avezum A, Bahit MC, Diaz R, Easton JD, Ezekowitz JA, Flaker G, Garcia D, Geraldes M, Gersh BJ, Golitsyn S, Goto S, Hermosillo AG, Hohnloser SH, Horowitz J, Mohan P, Jansky P, Lewis BS, Lopez-Sendon JL, Pais P, Parkhomenko A, Verheugt FW, Zhu J, Wallentin L, ARISTOTLE Committees and Investigators (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365:981–992
Reilly PA, Lehr T, Haertter S, Connolly SJ, Yusuf S, Eikelboom JW, Ezekowitz MD, Nehmiz G, Wang S, Wallentin L, Investigators RE-LY (2014) The effect of dabigatran plasma concentrations and patient characteristics on the frequency of ischemic stroke and major bleeding in atrial fibrillation patients: the RE-LY Trial (Randomized Evaluation of Long-Term Anticoagulation Therapy). J Am Coll Cardiol 63:321–328
Testa S, Paoletti O, Legnani C, Dellanoce C, Antonucci E, Cosmi B, Pengo V, Poli D, Morandini R, Testa R, Tripodi A, Palareti G (2018) Low drug levels and thrombotic complications in high-risk atrial fibrillation patients treated with direct oral anticoagulants. J Thromb Haemost 16:842–848
Schmitz EM, Boonen K, van den Heuvel DJ, van Dongen JL, Schellings MW, Emmen JM, van der Graaf F, Brunsveld L, van de Kerkhof D (2014) Determination of dabigatran, rivaroxaban and apixaban by ultra-performance liquid chromatography–tandem mass spectrometry (UPLC-MS/MS) and coagulation assays for therapy monitoring of novel direct oral anticoagulants. J Thromb Haemost 12:1636–1646
Slavik L, Lukes J, Friedecky D, Zhanelova M, Nemcova M, Ulehlova J, Prochazkova J, Hlusi A, Palova M, Vaclavik J (2018) Multianalyte determination of NOACs using LC-MS/MS and comparison with functional coagulation assays. Clin Lab 64:1611–1621
Samoš M, Stančiaková L, Ivanková J, Staško J, Kovář F, Dobrotová M, Galajda P, Kubisz P, Mokáň M (2015) Monitoring of dabigatran therapy using Hemoclot(®) Thrombin Inhibitor assay in patients with atrial fibrillation. J Thromb Thrombolysis 39:95–100
Samoš M, Bolek T, Stančiaková L, Škorňová I, Bánovčin P Jr, Kovář F, Staško J, Galajda P, Kubisz P, Mokáň M (2018) Anti-Xa activity in oral factor Xa inhibitor-treated patients with atrial fibrillation and a higher risk of bleeding: a pilot study. Blood Coagul Fibrinolysis 29:369–373
Wada S, Inoue M, Matsuki T, Okata T, Kumamoto M, Tagawa N, Yoshimura S, Okamoto A, Miyata T, Ihara M, Toyoda K, Koga M (2020) Concentrations of dabigatran administered after acute ischemic stroke. J Neurol Sci 411:116704
Macha K, Marsch A, Siedler G, Breuer L, Strasser EF, Engelhorn T, Schwab S, Kallmünzer B (2019) Cerebral ischemia in patients on direct oral anticoagulants. Stroke 50:873–879
Sugrue A, Sanborn D, Amin M, Farwati M, Sridhar H, Ahmed A, Mehta R, Siontis KC, Mulpuru SK, Deshmukh AJ, Gersh BJ, Asirvatham SJ, Madhavan M (2020) Inappropriate dosing of direct oral anticoagulants in patients with atrial fibrillation. Am J Cardiol. https://doi.org/10.1016/j.amjcard.2020.12.062
Di Gennaro L, Monaco M, Riccio C, De Candia E, Alberelli MA, di Martino C, Basso M, Ferretti MA, Lancellotti S, De Cristofaro R (2021) Direct oral anticoagulants and therapeutic adherence: do not let your guard down. Acta Cardiol. https://doi.org/10.1080/00015385.2021.1908702
Bolek T, Samoš M, Škorňová I, Galajda P, Staško J, Kubisz P, Mokáň M (2019) Proton pump inhibitors and dabigatran therapy: impact on gastric bleeding and dabigatran plasma levels. Semin Thromb Hemost 45:846–850
Kanuri SH, Kreutz RP (2019) Pharmacogenomics of novel direct oral anticoagulants: newly identified genes and genetic variants. J Pers Med 9:7
Antonijevic NM, Zivkovic ID, Jovanovic LM, Matic DM, Kocica MJ, Mrdovic IB, Kanjuh VI, Culafic MD (2017) Dabigatran—metabolism, pharmacologic properties and drug interactions. Curr Drug Metab 18:622–635
Kvasnicka T, Malikova I, Zenahlikova Z, Kettnerova K, Brzezkova R, Zima T, Ulrych J, Briza J, Netuka I, Kvasnicka J (2017) Rivaroxaban—metabolism, pharmacologic properties and drug interactions. Curr Drug Metab 18:636–642
Kubisz P, Stanciakova L, Dobrotova M, Samos M, Mokan M, Stasko J (2017) Apixaban—metabolism, pharmacologic properties and drug interactions. Curr Drug Metab 18:609–621
Acknowledgements
The authors would like to thank Mr. Paul McCullough for proofreading and A/Prof. Marián Grendár, PhD. for the assistance with statistical analysis.
Funding
This study was supported by research project APVV (Slovak Research and Development Agency) 16–0020, by research project of Research Agency of Slovak Ministry of Education, Science and Sports (VEGA) 1/0090/20, and by research and development project ITMS2014 + : 313011V344.
Author information
Authors and Affiliations
Contributions
V.N., I.Š., T.B., M.S., J.S., and E.K. designed the study. V.N., A.P., T.B., and M.S. drafted the manuscript. A.P., T.B., J.D., Š.S., L.B., G.H., and M.S. collected, analyzed, and interpreted the clinical data. I.Š. supervised anti-Xa activity assessment; I.Š. and L.S. assessed the anti-Xa activity, analyzed, and interpreted the laboratory data. Š.S., G.H., J.S., P.K., E.K., and M.M. revised the manuscript critically. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and informed consent
This research was done according to ethical standards and was approved by the local ethical committee (Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava). The patients agreed to participate in the research and signed informed consent for study participation.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nosáľ, V., Petrovičová, A., Škorňová, I. et al. Plasma levels of direct oral anticoagulants in atrial fibrillation patients at the time of embolic stroke: a pilot prospective multicenter study. Eur J Clin Pharmacol 78, 557–564 (2022). https://doi.org/10.1007/s00228-022-03280-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-022-03280-8