Abstract
Objective
The variability in warfarin dose requirement is attributable to genetic and environmental factors. Acenocoumarol (AC) and phenprocoumon (PC) are coumarin derivates widely prescribed in European countries for the prevention and treatment of thromboembolic events. The aim of our study was to investigate the contribution of genes involved in the vitamin K cycle to AC and PC maintenance doses.
Methods
Common single nucleotide polymorphisms (SNPs) in the genes encoding cytochrome P450 family member 2C9 (CYP2C9), vitamin K epoxide reductase complex subunit 1 (VKORC1), γ-glutamyl carboxylase (GGCX), calumenin (CALU) and apolipoprotein E (APOE) were studied in 206 patients receiving AC or PC.
Results
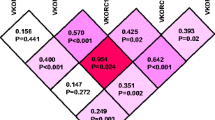
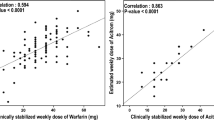
Compared to patients with the VKORC1 C1173C genotype, maintenance doses for AC or PC were reduced to 74.6 or 70.2% in heterozygous C1173T subjects and to 48.6 or 48.1% in homozygous T1173T subjects (P < 0.0001). Furthermore maintenance doses for AC and PC were significantly lower in heterozygous CYP2C9*1*3, CYP2C9*2*3, and in CYP2C9*3*3 homozygote individuals compared to homozygous CYP2C9*1*1 subjects (P = 0.0004 and P = 0.0017, respectively). A multiple regression model including age, sex, last INR, VKORC1, and CYP2C9 genotypes explained ~50% of the variability in AC/PC dose requirements. CALU genotype combinations showed minor effects on PC dose requirements. No associations with AC or PC dose requirements were observed for sequence substitutions in the GGCX or APOE genes.
Conclusion
These results reveal that interindividual variability in weekly AC and PC maintenance dose requirement is mainly dependent on the VKORC1 1173C>T and the CYP2C9*3 alleles. VKORC1 and CYP2C9 genotyping might provide helpful information to prevent serious bleeding events in subjects receiving AC or PC.
Similar content being viewed by others
Abbreviations
- AC:
-
Acenocoumarol
- PC:
-
Phenprocoumon
- SNP:
-
Single nucleotide polymorphism
- CYP2C9:
-
Cytochrome P450 family member 2C9
- VKORC1:
-
Vitamin K epoxide reductase complex subunit 1
- GGCX:
-
γ-Glutamyl carboxylase
- ApoE:
-
Apolipoprotein E
- RFLP:
-
Restriction fragment length polymorphism
- ANOVA:
-
Analysis of variance
References
Cannegieter SC, Torn M, Rosendaal FR (1999) Oral anticoagulant treatment in patients with mechanical heart valves: how to reduce the risk of thromboembolic and bleeding complications. J Intern Med 245(4):369–374
D’Andrea G, D’Ambrosio R, Margaglione M (2008) Oral anticoagulants: pharmacogenetics relationship between genetic and non-genetic factors. Blood Rev 22(3):127–140
He M, Korzekwa KR, Jones JP, Rettie AE, Trager WF (1999) Structural forms of phenprocoumon and warfarin that are metabolized at the active site of CYP2C9. Arch Biochem Biophys 372(1):16–28
Takanashi K, Tainaka H, Kobayashi K et al (2000) CYP2C9 Ile359 and Leu359 variants: enzyme kinetic study with seven substrates. Pharmacogenetics 10(2):95–104
Taube J, Halsall D, Baglin T (2000) Influence of cytochrome P-450 CYP2C9 polymorphisms on warfarin sensitivity and risk of over-anticoagulation in patients on long-term treatment. Blood 96(5):1816–1819
Lindh JD, Holm L, Andersson ML, Rane A (2008) Influence of CYP2C9 genotype on warfarin dose requirements-a systematic review and meta-analysis. Eur J Clin Pharmacol 65(4):365–375
Higashi MK, Veenstra DL, Kondo LM et al (2002) Association between CYP2C9 genetic variants and anticoagulation-related outcomes during warfarin therapy. JAMA 287(13):1690–1698
Aithal GP, Day CP, Kesteven PJ, Daly AK (1999) Association of polymorphisms in the cytochrome P450 CYP2C9 with warfarin dose requirement and risk of bleeding complications. Lancet 353(9154):717–719
Hirsh J, Dalen J, Anderson DR et al (2001) Oral anticoagulants: mechanism of action, clinical effectiveness, and optimal therapeutic range. Chest 119(1 Suppl):8S–21S
Wu SM, Stafford DW, Frazier LD et al (1997) Genomic sequence and transcription start site for the human gamma-glutamyl carboxylase. Blood 89(11):4058–4062
Li T, Chang CY, Jin DY et al (2004) Identification of the gene for vitamin K epoxide reductase. Nature 427(6974):541–544
Rost S, Fregin A, Ivaskevicius V et al (2004) Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2. Nature 427(6974):537–541
Wang D, Chen H, Momary KM et al (2008) Regulatory polymorphism in vitamin K epoxide reductase complex subunit 1 (VKORC1) affects gene expression and warfarin dose requirement. Blood 112(4):1013–1021
Geisen C, Watzka M, Sittinger K et al (2005) VKORC1 haplotypes and their impact on the inter-individual and inter-ethnical variability of oral anticoagulation. Thromb Haemost 94(4):773–779
Reitsma PH, van der Heijden JF, Groot AP et al (2005) A C1173T dimorphism in the VKORC1 gene determines coumarin sensitivity and bleeding risk. PLoS Med 2(10):e312
Wadelius M, Chen LY, Downes K et al (2005) Common VKORC1 and GGCX polymorphisms associated with warfarin dose. Pharmacogenomics J 5(4):262–270
Gonzalez-Conejero R, Corral J, Roldan V et al (2007) The genetic interaction between VKORC1 c1173t and calumenin a29809g modulates the anticoagulant response of acenocoumarol. J Thromb Haemost 5(8):1701–1706
Wajih N, Sane DC, Hutson SM, Wallin R (2004) The inhibitory effect of calumenin on the vitamin K-dependent gamma-carboxylation system. Characterization of the system in normal and warfarin-resistant rats. J Biol Chem 279(24):25276–25283
Khan T, Wynne H, Wood P et al (2004) Dietary vitamin K influences intra-individual variability in anticoagulant response to warfarin. Br J Haematol 124(3):348–354
Lamon-Fava S, Sadowski JA, Davidson KW et al (1998) Plasma lipoproteins as carriers of phylloquinone (vitamin K1) in humans. Am J Clin Nutr 67(6):1226–1231
Saupe J, Shearer MJ, Kohlmeier M (1993) Phylloquinone transport and its influence on gamma-carboxyglutamate residues of osteocalcin in patients on maintenance hemodialysis. Am J Clin Nutr 58(2):204–208
Kimmel SE, Christie J, Kealey C et al (2008) Apolipoprotein E genotype and warfarin dosing among Caucasians and African Americans. Pharmacogenomics J 8(1):53–60
Klein TE, Altman RB, Eriksson N et al. (2009) Estimation of the warfarin dose with clinical and pharmacogenetic data. N Engl J Med 360(8):753–764
Baxter K (ed) (2007) Stockley’s drug interactions: a source book of interactions, their mechanisms, clinical importance and management, 8 ed. Pharmaceutical Press, London
Lacy CF, Armstrong LL, Goldman MP, Lance LL (eds) (2009) Drug information handbook: a comprehensive resource for all clinicians and healthcare professionals, 18th ed. Lexi-Comp, Hudson, OH
Oberkofler H, Linnemayr V, Weitgasser R et al (2004) Complex haplotypes of the PGC-1alpha gene are associated with carbohydrate metabolism and type 2 diabetes. Diabetes 53(5):1385–1393
Au N, Rettie AE (2008) Pharmacogenomics of 4-hydroxycoumarin anticoagulants. Drug Metab Rev 40(2):355–375
Ufer M (2005) Comparative pharmacokinetics of vitamin K antagonists: warfarin, phenprocoumon and acenocoumarol. Clin Pharmacokinet 44(12):1227–1246
Bodin L, Verstuyft C, Tregouet DA et al (2005) Cytochrome P450 2C9 (CYP2C9) and vitamin K epoxide reductase (VKORC1) genotypes as determinants of acenocoumarol sensitivity. Blood 106(1):135–140
Qazim B, Stollberger C, Krugluger W et al (2008) Dependency of phenprocoumon dosage on polymorphisms in the VKORC1 and CYP2C9 genes. J Thromb Thrombolysis 28:211–214
Schalekamp T, Brasse BP, Roijers JF et al (2007) VKORC1 and CYP2C9 genotypes and phenprocoumon anticoagulation status: interaction between both genotypes affects dose requirement. Clin Pharmacol Ther 81(2):185–193
D’Andrea G, D’Ambrosio RL, Di Perna P et al (2005) A polymorphism in the VKORC1 gene is associated with an interindividual variability in the dose-anticoagulant effect of warfarin. Blood 105(2):645–649
Rieder MJ, Reiner AP, Gage BF et al (2005) Effect of VKORC1 haplotypes on transcriptional regulation and warfarin dose. N Engl J Med 352(22):2285–2293
Thijssen HH, Flinois JP, Beaune PH (2000) Cytochrome P4502C9 is the principal catalyst of racemic acenocoumarol hydroxylation reactions in human liver microsomes. Drug Metab Dispos 28(11):1284–1290
Li C, Schwarz UI, Ritchie MD et al. (2008) Relative contribution of CYP2C9 and VKORC1 genotypes and early INR response to the prediction of warfarin sensitivity during initiation of therapy. Blood 113:3925–3930
Ufer M, Kammerer B, Kahlich R et al (2004) Genetic polymorphisms of cytochrome P450 2C9 causing reduced phenprocoumon (S)-7-hydroxylation in vitro and in vivo. Xenobiotica 34(9):847–859
Kirchheiner J, Ufer M, Walter EC et al (2004) Effects of CYP2C9 polymorphisms on the pharmacokinetics of R- and S-phenprocoumon in healthy volunteers. Pharmacogenetics 14(1):19–26
Visser LE, van Schaik RH, van Vliet M et al (2004) The risk of bleeding complications in patients with cytochrome P450 CYP2C9*2 or CYP2C9*3 alleles on acenocoumarol or phenprocoumon. Thromb Haemost 92(1):61–66
Hummers-Pradier E, Hess S, Adham IM et al (2003) Determination of bleeding risk using genetic markers in patients taking phenprocoumon. Eur J Clin Pharmacol 59(3):213–219
Schalekamp T, Oosterhof M, van Meegen E et al (2004) Effects of cytochrome P450 2C9 polymorphisms on phenprocoumon anticoagulation status. Clin Pharmacol Ther 76(5):409–417
Caldwell MD, Awad T, Johnson JA et al (2008) CYP4F2 genetic variant alters required warfarin dose. Blood 111(8):4106–4112
Vecsler M, Loebstein R, Almog S et al (2006) Combined genetic profiles of components and regulators of the vitamin K-dependent gamma-carboxylation system affect individual sensitivity to warfarin. Thromb Haemost 95(2):205–211
Wadelius M, Chen LY, Eriksson N et al (2007) Association of warfarin dose with genes involved in its action and metabolism. Hum Genet 121(1):23–34
Kimura R, Miyashita K, Kokubo Y et al (2007) Genotypes of vitamin K epoxide reductase, gamma-glutamyl carboxylase, and cytochrome P450 2C9 as determinants of daily warfarin dose in Japanese patients. Thromb Res 120(2):181–186
Pudota BN, Hommema EL, Hallgren KW et al (2001) Identification of sequences within the gamma-carboxylase that represent a novel contact site with vitamin K-dependent proteins and that are required for activity. J Biol Chem 276(50):46878–46886
Visser LE, Trienekens PH, De Smet PA et al (2005) Patients with an ApoE epsilon4 allele require lower doses of coumarin anticoagulants. Pharmacogenet Genomics 15(2):69–74
Cooper GM, Johnson JA, Langaee TY et al (2008) A genome-wide scan for common genetic variants with a large influence on warfarin maintenance dose. Blood 112(4):1022–1027
Teichert M, Eijgelsheim M, Rivadeneira F et al (2009) A genome-wide association study of acenocoumarol maintenance dosage. Hum Mol Genet 18(19):3758–3768
Wadelius M, Chen LY, Lindh JD et al (2009) The largest prospective warfarin-treated cohort supports genetic forecasting. Blood 113(4):784–792
Acknowledgments
The technical assistance of I. Kremser, H. Schnaitl, and E. Wölbl is greatly acknowledged. This study was supported by grants from the Forschungsfonds der Paracelsus Medizinischen Privatuniversität Salzburg (Project 07/06/034), the Land Salzburg, and the Verein für Medizinische Forschung Salzburg, Austria.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Table I
Relationship between APOE, GGCX and Calumenin genotypes and mean acenocoumarol weekly dose requirement (DOC 49 kb)
Table II
Relationship between APOE, GGCX and Calumenin genotypes and mean phenprocoumon weekly dose requirement (DOC 49 kb)
Rights and permissions
About this article
Cite this article
Cadamuro, J., Dieplinger, B., Felder, T. et al. Genetic determinants of acenocoumarol and phenprocoumon maintenance dose requirements. Eur J Clin Pharmacol 66, 253–260 (2010). https://doi.org/10.1007/s00228-009-0768-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-009-0768-7