Abstract
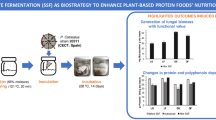
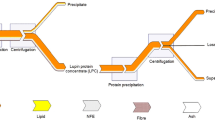
In some European countries, lupin seeds are not used efficiently enough, due to a lack of processing technologies for the preparation of attractive higher-value products; fermentation could be a promising process to increase the value of lupin seeds and their protein isolates. In this study, the influence of a range of factors—seeds of Lupinus angustifolius L. hybrid line (HL) Nos. 1700, 1701, 1800, and 1072, solid state and submerged fermentation methods (FMs) with different Pediococcus pentosaceus strains (05–8, 05–9, and 05–10), and the use of a protein isolation (PI) process—on the total content of phenolic compounds (TPC) and isoflavones, antioxidant properties, trypsin inhibitor activity (TIA), and protein digestibility (PD) of lupin wholemeal and protein isolates was evaluated. In addition, changes in the SDS-PAGE profiles of fermented lupin protein isolates were analysed. The selected Pediococcus strains were found to be suitable starters for the fermentation of lupin seeds. The protein content in isolates ranged from 80.43 to 89.08% on a dry weight basis and was significantly influenced by FM (p = 0.0001). The most common protein fractions in isolates ranged in molecular weight from 15 to 167 kDa. The PD of lupin wholemeal samples was on average 3.13% higher than that of protein isolates. The genistein content of lupin samples showed a significant correlation with PI and HL (p = 0.002 and p = 0.0001, respectively), and a lower genistein content was observed in lupin protein isolates. Fermentation can provide a basis for the development of higher-value products, but the technological parameters should be selected by taking into account that some technological steps (PI) can reduce the amount of genistein.





Similar content being viewed by others
References
Gresta F, De Luca AI, Strano A, Falcone G, Santonoceto C, Anastasi U, Gulisano G (2014) Ital J Agron 9:20–24
Belski R, Jeyakumar H (2012) Adv Food Nutr Res 66:147
Lucas MM, Stoddard FL, Annicchiarico P, Frias J, Martinez-Villaluenga C, Sussmann D, Pueyo JJ (2015) Front Plant Sci 6:705
Czubinski J, Montowska M, Pospiech E, Lampart-Szczapa E (2017) J Sci Food Agric 97:1–8
Melini F, Melini V, Luziatelli F, Ruzzi M (2017) Compr Rev Food Sci Food Saf 16:1101–1122
Bähr M, Fechner A, Kiehntopf M, Jahreis G (2015) Clin Nutr 34:7–14
Arnoldi A, Zanoni C, Lammi C, Boschin G (2015) Crit Rev Plant Sci 34:144–168
Rietjens IMCM., Louisse J, Beekmann (2017) Br J Pharmacol 174:1263–1280
Gupta A, Tiwari SK (2014) Probiotics Antimicrob Proteins 6:73–81
Hur SJ, Lee SY, Kim YC, Choi I, Kim GB (2014) Food Chem 160:346–356
Dordevic TM, Šiler-Marinkovic SS, Dimitrijevic-Brankovic SI (2010) Food Chem 119:957–963
Nagino T, Mitsuyoshi KA, Masuoka N, Chiaki KA, Michitoshi AN, Miyazaki K, Kamachi K, Isozak M, Suzuki C, Kasuga C, Tanaka A (2016) Biosci Microb Food Health 35:9–17
Jackson CJ, Dini JP, Lavandier C, Rupasinghe HP, Faulkner H, Poysa V, Buzzell D, DeGrandis S (2002) Process Biochem 37:1117–1123
Bartkiene E, Bartkevics V, Starkute V, Krungleviciute V, Cizeikiene D, Zadeike D, Juodeikiene G (2016) Zemdirb Agric 103:107–114
Bartkiene E, Bartkevics V, Rusko J, Starkute V, Zadeike D, Juodeikiene G (2016) Int J Food Sci Technol 51:2049–2056
ISO 15214:1998 (1998) Horizontal method for the enumeration of mesophilic lactic acid bacteria. Colony-count technique at 30 °C. International standards. Microbiology of food and animal feeding stuffs. International Organization for Standardization, Ginebra
Muranyi IS, Volke D, Hoffmann R, Eisner P, Herfellner T, Brunnbauer M, Schweiggert-Weisz U (2016) Food Chem 207:6–15
King J, Aguirre C, De Pablo S (1985) J Food Sci 50:82–87
AOAC International (1990) Method 968.06. Official methods of analysis of the Association of Official Analytical Chemists. Gaithersburg, MD, USA
ICC 105/2:1994 (1995) Determination of crude protein in cereals and cereal products for food and for feed. Ed. International Association for Cereal Science and Technology, Vienna
Lqari H, Vioque J, Pedroche J, Millán F (2002) Food Chem 76:349–356
Vaher M, Matso K, Levandi T, Helmja K, Kaljurand M (2010) Procedia Chem 2:76–82
Zhu KX, Lian CX, Guo XN, Peng W, Zhou HM (2011) Food Chem 126:1122–1126
Gobbetti M, De Angelis M, Corsetti A, Di Cagno R (2005) Trends Food Sci Technol 16:57–69
Fritsch C, Vogel RF, Toelstede S (2015) J Appl Microbiol 119:1075–1088
Kumar BV, Vijayendra SVN, Reddy OVS (2015) J Food Sci Technol 52:6112–6124
Jiang J, Chen J, Xiong YL (2009) J Agric Food Chem 57:7576–7583
Schoustra SE, Kasase C, Toarta C, Kassen R, Poulain AJ (2013) PLoS One 8:639–648
Tyl C, Sadler GD (2017) pH and Titratable Acidity. In: Nielsen S (ed) Food Analysis. Springer, Cham, pp 389–406
Feyzi S, Varidi M, Zare F, Varidi MJ (2015) J Sci Food Agric 95:3165–3176
Ballabio C, Peñas E, Uberti F, Fiocchi A, Duranti M, Magni C, Restani P (2013) Pediatr Allergy Immunol 24:270–275
Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A (2013) Antioxid Redox Signal 18:1818–1892
Othman NB, Roblain D, Chammen N, Thonart P, Hamdi M (2009) Food Chem 116:662–669
Gan RY, Shah NP, Wang MF, Lui WY, Corke H (2016) Int J Food Sci Technol 51:875–884
Gujral HS, Sharma P, Bajaj R, Solah V (2012) J Food Sci Technol 18:47–54
Sindhu SC, Khetarpaul N (2001) J Food Compos Anal 14:601–609
Gilani GS, Xiao CW, Cockell KA (2012) Br J Nutr 108:315–332
Wati RK, Theppakorn T, Benjakul S, Rawdkuen S (2010) J Food Sci 75:C223–C228
Wink M (2013) South Afr J Bot 89:164–175
Grela ER, Kiczorowska B, Samolińska W, Matras J, Kiczorowski P, Rybiński W, Hanczakowska E (2017) Eur Food Res Technol 243:1385–1395
Kobayashi H (2013) Front Biosci (Elite Ed) 5:966–973
Dallas D, Sanctuary MR, Qu Y, Khajavi SH, Van Zandt AE, Dyandra M, Frese SA, Barile D, German JB (2017) Crit Rev Food Sci Nutr 57:3313–3331
Feng J, Liu X, Xu Z, Lu Y, Liu Y (2007) Anim Feed Sci Technol 134:295–303
Zhang ZL, Zhou ML, Tang Y, Li FL, Tang YX, Shao JR, Wu YM (2012) Food Res Int 49:389–395
Oomah BD, Tiger N, Olson M, Balasubramanian P (2006) Plant Foods Hum Nutr 61:91–97
Kim SH, Choi KC (2014) J Nutr Biochem 28:70–82
Antosiak A, Milowska K, Maczynska K, Rozalska S, Gabryelak T (2017) Med Chem Res 26:64–73
Danciu C, Oana S, Antal DS, Ardelean F, Chis AR, Soica C, Andrica F, Dehelean C, Brigitha V (2017) New Insights Regarding the Potential Health Benefits of Isoflavones. In: Badria FA (ed) Natural products and cancer drug discovery. InTech. ISBN 978-953-51-3314-8, Print ISBN 978-953-51-3313-1
Liu ZM, Ho SC, Chen YM, Ho S, To K, Tomlinson B, Woo J (2014) Mol Nutr Food Res 58:709–717
Pakalapati G, Li L, Gretz N, Koch E, Wink M (2009) Phytomedicine 16:845–855
Wang HJ, Murphy PA (1996) J Agric Food Chem 44:2377–2383
Acknowledgements
Part of this research was funded by the Baltic-German University Liaison Office project, which is supported by the German Academic Exchange Service (DAAD) with funds from the Foreign Office of the Federal Republic of Germany.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Bartkiene, E., Sakiene, V., Bartkevics, V. et al. Lupinus angustifolius L. lactofermentation and protein isolation: effects on phenolic compounds and genistein, antioxidant properties, trypsin inhibitor activity, and protein digestibility. Eur Food Res Technol 244, 1521–1531 (2018). https://doi.org/10.1007/s00217-018-3066-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-018-3066-8