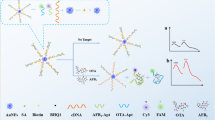
Abstract
This paper describes a novel fluorescence label-free aptasensor to detect aflatoxin B1 (AFB1) by utilizing SYBR Gold, aptamer, and single-walled carbon nanohorns (SWCNHs). In the presence of AFB1, the conformation of AFB1-specific aptamer went through and the spatial structure of this specific aptamer was transformed accordingly. Due to the resistance of the transformed aptamer when adsorbed on the surface of SWCNHs, the protection of the fluorescence of SYBR Gold was accomplished. Consequently, concentrations of AFB1 showed a strong association with fluorescence intensity. The detection limit (LOD) of AFB1 was 1.89 ng/mL, while the linear range was 5–200 ng/mL and fluorescence intensity satisfactorily correlated (R2 = 0.9919) with the logarithm of AFB1 concentration.
Graphical abstract






Similar content being viewed by others
References
Qian J, Ren C, Wang C, An K, Cui H, Hao N, Wang K. Gold nanoparticles mediated designing of versatile aptasensor for colorimetric/electrochemical dual-channel detection of aflatoxin B1. Biosens Bioelectron. 2020;166. https://doi.org/10.1016/j.bios.2020.112443.
Li Y, Sun L, Zhao Q. Development of aptamer fluorescent switch assay for aflatoxin B1 by using fluorescein-labeled aptamer and black hole quencher 1-labeled complementary DNA. Anal Bioanal Chem. 2018;410(24):6269–77. https://doi.org/10.1007/s00216-018-1237-x.
Zhang ZH, Li MY, Wang Z, Zuo HX, Wang JY, Xing Y, Jin C, Xu G, Piao L, Piao H, Ma J, Jin X. Convallatoxin promotes apoptosis and inhibits proliferation and angiogenesis through crosstalk between JAK2/STAT3 (T705) and mTOR/STAT3 (S727) signaling pathways in colorectal cancer. Phytomedicine. 2020;68. https://doi.org/10.1016/j.phymed.2020.153172
Li Y, Liu D, Zhu C, Shen X, Liu Y, You T. Sensitivity programmable ratiometric electrochemical aptasensor based on signal engineering for the detection of aflatoxin B1 in peanut. J Hazard Mater. 2020;387. https://doi.org/10.1016/j.jhazmat.2019.122001
Rahimi F, Roshanfekr H, Peyman H. Ultra-sensitive electrochemical aptasensor for label-free detection of Aflatoxin B1 in wheat flour sample using factorial design experiments. Food Chem. 2021;343. https://doi.org/10.1016/j.foodchem.2020.128436
Goud KY, Catanante G, Hayat A, Satyanarayana M, Gobi KV, Marty JL. Disposable and portable electrochemical aptasensor for label free detection of aflatoxin B1 in alcoholic beverages. Sensor Actuat B-Chem. 2016;235:466–73. https://doi.org/10.1016/j.snb.2016.05.112.
Chen J, Wen J, Zhuang L, Zhou S. An enzyme-free catalytic DNA circuit for amplified detection of aflatoxin B1 using gold nanoparticles as colorimetric indicators. Nanoscale. 2016;8(18):9791–7. https://doi.org/10.1039/c6nr01381c.
Wu J, Zeng L, Li N, Liu C, Chen J. A wash-free and label-free colorimetric biosensor for naked-eye detection of aflatoxin B1 using G-quadruplex as the signal reporter. Food Chem. 2019;298. 125034
Zhang J, Xia YK, Chen M, Wu DZ, Cai SX, Liu MM, He WH, Chen JH. A fluorescent aptasensor based on DNA-scaffolded silver nanoclusters coupling with Zn(II)-ion signal-enhancement for simultaneous detection of OTA and AFB(1). Sensor Actuat B-Chem. 2016;235:79–85. https://doi.org/10.1016/j.snb.2016.05.061.
Abnous K, Danesh NM, Alibolandi M, Ramezani M, Emrani AS, Zolfaghari R, Taghdisi SM. A new amplified pi-shape electrochemical aptasensor for ultrasensitive detection of aflatoxin B-1. Biosens Bioelectron. 2017;94:374–9. https://doi.org/10.1016/j.bios.2017.03.028.
Chen L, Wen F, Li M, Guo X, Li S, Zheng N, Wang J. A simple aptamer-based fluorescent assay for the detection of Aflatoxin B-1 in infant rice cereal. Food Chem. 2017;215:377–82. https://doi.org/10.1016/j.foodchem.2016.07.148.
Zhang X, Tang Q, Mi T, Zhao S, Wen K, Guo L, Mi J, Zhang S, Shi W, Shen J, Ke Y, Wang Z. Dual-wavelength fluorescence polarization immunoassay to increase information content per screen: Applications for simultaneous detection of total aflatoxins and family zearalenones in maize. Food Control. 2018;87:100–8. https://doi.org/10.1016/j.foodcont.2017.12.002.
Zhan S, Hu J, Li Y, Huang X, Xiong Y. Direct competitive ELISA enhanced by dynamic light scattering for the ultrasensitive detection of aflatoxin B-1 in corn samples. Food Chem. 2021;342. https://doi.org/10.1016/j.foodchem.2020.128327
Xu Z, Long L-l, Chen Y-q, Chen M-L, Cheng Y-H. A nanozyme-linked immunosorbent assay based on metal-organic frameworks (MOFs) for sensitive detection of aflatoxin B-1. Food Chem. 2021;338. https://doi.org/10.1016/j.foodchem.2020.128039
Sang FM, Zhang X, Liu J, Yin SY, Zhang ZZ. A label-free hairpin aptamer probe for colorimetric detection of adenosine triphosphate based on the anti-aggregation of gold nanoparticles. Spectrochim Acta Part a. 2019;217:122–7. https://doi.org/10.1016/j.saa.2019.03.081.
Zhao Y, Liu R, Sun W, Lv L, Guo Z. Ochratoxin A detection platform based on signal amplification by Exonuclease III and fluorescence quenching by gold nanoparticles. Sensor Actuat B-Chem. 2018;255:1640–5. https://doi.org/10.1016/j.snb.2017.08.176.
Roushani M, Ghanbari K. A novel aptasensor based on gold nanorods/ZnS QDs-modified electrode for evaluation of streptomycin antibiotic. Anal Method. 2018;10(43):5197–204. https://doi.org/10.1039/c8ay01815d.
Ji DY, Wang HQ, Ge J, Zhang L, Li JJ, Bai DM, Chen J, Li ZH. Label-free and rapid detection of ATP based on structure switching of aptamers. Anal Biochem. 2017;526:22–8. https://doi.org/10.1016/j.ab.2017.03.011.
Gao J, Yao X, Chen Y, Gao Z, Zhang J. Near-Infrared Light-Induced Self-Powered Aptasensing Platform for Aflatoxin B1 Based on Upconversion Nanoparticles-Doped Bi2S3 Nanorods. Anal chem. 2021;93(2):677–82. https://doi.org/10.1021/acs.analchem.0c04248.
Nasirian V, Chabok A, Barati A, Rafienia M, Arabi MS, Shamsipur M. Ultrasensitive aflatoxin B1 assay based on FRET from aptamer labelled fluorescent polymer dots to silver nanoparticles labeled with complementary DNA. Microchim Acta. 2017;184(12):4655–62. https://doi.org/10.1007/s00604-017-2508-5.
Barth I, Conteduca D, Reardon C, Johnson S, Krauss TF. Common-path interferometric label-free protein sensing with resonant dielectric nanostructures. Light-Sci Appl. 2020;9 (1). https://doi.org/10.1038/s41377-020-0336-6
Cao Z, Yao B, Qin C, Yang R, Guo Y, Zhang Y, Wu Y, Bi L, Chen Y, Xie Z, Peng G, Huang S-W, Wong CW, Rao Y. Biochemical sensing in graphene-enhanced microfiber resonators with individual molecule sensitivity and selectivity. Light-Sci Appl. 2019;8. https://doi.org/10.1038/s41377-019-0213-3
Riedel M, Holzel S, Hille P, Schormann J, Eickhoff M, Lisdat F. InGaN/GaN nanowires as a new platform for photoelectrochemical sensors - detection of NADH. Biosens Bioelectron. 2017;94:298–304. https://doi.org/10.1016/j.bios.2017.03.022.
Etezadi D, Warner JB, Ruggeri FS, Dietler G, Lashuel HA, Altug H. Nanoplasmonic mid-infrared biosensor for in vitro protein secondary structure detection. Light-Sci Appl. 2017;6. https://doi.org/10.1038/lsa.2017.29
Jung H, Park M, Kang M, Jeong K-H. Silver nanoislands on cellulose fibers for chromatographic separation and ultrasensitive detection of small molecules. Light-Sci Applications. 2016;5. https://doi.org/10.1038/lsa.2016.9
Peng G, Li X, Cui F, Qiu Q, Chen X, Huang H. Aflatoxin B1 Electrochemical Aptasensor Based on Tetrahedral DNA Nanostructures Functionalized Three Dimensionally Ordered Macroporous MoS2-AuNPs Film. Acs Appl Mater Interf. 2018;10(21):17551–9. https://doi.org/10.1021/acsami.8b01693.
Li H, Xu SL, Quan JS, Yung BC, Pang JX, Zhou CG, Cho YA, Zhang MZ, Liu SJ, Muthusamy N, Chan KK, Byrd JC, Lee LJ, Marcucci G, Lee RJ. CD33-Targeted Lipid Nanoparticles (aCD33LNs) for Therapeutic Delivery of GTI-2040 to Acute Myelogenous Leukemia. Mol Pharmaceut. 2015;12(6):2010–8. https://doi.org/10.1021/mp5008212.
Hu X, Saravanakumar K, Jin T, Wang M-H. Mycosynthesis, characterization, anticancer and antibacterial activity of silver nanoparticles from endophytic fungus Talaromyces purpureogenus. Int J Nanomed. 2019;14:3427–38. https://doi.org/10.2147/ijn.s200817.
Lv L, Cui C, Liang C, Quan W, Wang S, Guo Z. Aptamer-based single-walled carbon nanohorn sensors for ochratoxin A detection. Food Control. 2016;60:296–301. https://doi.org/10.1016/j.foodcont.2015.08.002.
Liu J, Xiong RH, Brans T, Lippens S, Parthoens E, Zanacchi FC, Magrassi R, Singh SK, Kurungot S, Szunerits S, Bove H, Ameloot M, Fraire JC, Teirlinck E, Samal SK, De Rycke R, Houthaeve G, De Smedt SC, Boukherroub R, Braeckmans K. Repeated photoporation with graphene quantum dots enables homogeneous labeling of live cells with extrinsic markers for fluorescence microscopy. Light-Sci Appl. 2018;7. https://doi.org/10.1038/s41377-018-0048-3
Xin H, Li B. Optical orientation and shifting of a single multiwalled carbon nanotube. Light-Sci Appl. 2014;3. https://doi.org/10.1038/lsa.2014.86
Lim S-H, Li C-H, Jeong Y-I, Jang W-Y, Choi J-M, Jung S. Enhancing Radiotherapeutic Effect With Nanoparticle-Mediated Radiosensitizer Delivery Guided By Focused Gamma Rays In Lewis Lung Carcinoma-Bearing Mouse Brain Tumor Models. Int J Nanomed. 2019;14:8861–74. https://doi.org/10.2147/ijn.s227894.
Liu X, Ying Y, Ping J. Structure, synthesis, and sensing applications of single-walled carbon nanohorns. Biosens Bioelectron. 2020;167. https://doi.org/10.1016/j.bios.2020.112495
Moreno-Lanceta A, Medrano-Bosch M, Melgar-Lesmes P. Single-Walled Carbon Nanohorns as Promising Nanotube-Derived Delivery Systems to Treat Cancer. Pharmaceutics. 2020;12(9). https://doi.org/10.3390/pharmaceutics12090850
Wang H, Pan L, Liu Y, Ye Y, Yao S. Electrochemical sensing of nitenpyram based on the binary nanohybrid of hydroxylated multiwall carbon nanotubes/single-wall carbon nanohorns. J Electroanal Chem. 2020;862. https://doi.org/10.1016/j.jelechem.2020.113955
Ekwueme EC, Rao R, Mohiuddin M, Pellegrini M, Lee YS, Reiter MP, Jackson J, Freeman JW. Single-walled carbon nanohorns modulate tenocyte cellular response and tendon biomechanics. J Biomed Mater Res Part B. 2020;108(5):1907–14. https://doi.org/10.1002/jbm.b.34532.
Guo Z, Tian J, Cui C, Wang Y, Yang H, Yuan M, Yu H. A label-free aptasensor for turn-on fluorescent detection of ochratoxin a based on SYBR gold and single walled carbon nanohorns. Food Control. 2021;123. https://doi.org/10.1016/j.foodcont.2020.107741
Guo M, Hou Q, Waterhouse GIN, Hou J, Ai S, Li X. A simple aptamer-based fluorescent aflatoxin B1 sensor using humic acid as quencher. Talanta. 2019;205. https://doi.org/10.1016/j.talanta.2019.120131
Lerdsri J, Chananchana W, Upan J, Sridara T, Jakmunee J. Label-free colorimetric aptasensor for rapid detection of aflatoxin B1 by utilizing cationic perylene probe and localized surface plasmon resonance of gold nanoparticles. Sensor Actuat B-Chem. 2020;320. https://doi.org/10.1016/j.snb.2020.128356
Zhang C, Dou X, Zhang L, Sun M, Zhao M, OuYang Z, Kong D, Antonio FL, Yang M. A Rapid Label-Free Fluorescent Aptasensor PicoGreen-Based Strategy for Aflatoxin B-1 Detection in Traditional Chinese Medicines. Toxins. 2018;10 (3). https://doi.org/10.3390/toxins10030101
Lv L, Li D, Liu R, Cui C, Guo Z. Label-free aptasensor for ochratoxin A detection using SYBR Gold as a probe. Sensor Actuat B-Chem. 2017;246:647–52. https://doi.org/10.1016/j.snb.2017.02.143.
Li H, Guo Z, Xie W, Sun W, Ji S, Tian J, Lv L. A label-free fluorometric aptasensor for adenosine triphosphate (ATP) detection based on aggregation-induced emission probe. Anal Biochem. 2019;578:60–5. https://doi.org/10.1016/j.ab.2019.05.009.
Sabet FS, Hosseini M, Khabbaz H, Dadmehr M, Ganjali MR. FRET-based aptamer biosensor for selective and sensitive detection of aflatoxin B1 in peanut and rice. Food Chem. 2017;220:527–32. https://doi.org/10.1016/j.foodchem.2016.10.004.
Zhu S, Liu Z, Zhang W, Han S, Hu L, Xu G. Nucleic acid detection using single-walled carbon nanohorns as a fluorescent sensing platform. Chem Commun. 2011;47(21):6099–101. https://doi.org/10.1039/c1cc10952a.
Guo Z, Lv L, Cui C, Wang Y, Ji S, Fang J, Yuan M, Yu H. Detection of aflatoxin B(1)with a new label-free fluorescent aptasensor based on exonuclease I and SYBR Gold. Anal Methods. 2020;12(22):2928–33. https://doi.org/10.1039/d0ay00967a.
Li A, Tang L, Song D, Song S, Ma W, Xu L, Kuang H, Wu X, Liu L, Chen X, Xu C. A SERS-active sensor based on heterogeneous gold nanostar core-silver nanoparticle satellite assemblies for ultrasensitive detection of aflatoxinB1. Nanoscale. 2016;8(4):1873–8. https://doi.org/10.1039/c5nr08372a.
Kong D, Liu L, Song S, Suryoprabowo S, Li A, Kuang H, Wang L, Xu C. A gold nanoparticle-based semi-quantitative and quantitative ultrasensitive paper sensor for the detection of twenty mycotoxins. Nanoscale. 2016;8(9):5245–53. https://doi.org/10.1039/c5nr09171c.
Gell RM, Carbone I. HPLC quantitation of aflatoxin B-1 from fungal mycelium culture. J Microbio Methods. 2019;158:14–7. https://doi.org/10.1016/j.mimet.2019.01.008.
Hosseini M, Khabbaz H, Dadmehr M, Ganjali MR, Mohamadnejad J. Aptamer-based Colorimetric and Chemiluminescence Detection of Aflatoxin B1 in Foods Samples. Acta Chim Slov. 2015;62(3):721–8.
Joo M, Baek SH, Cheon SA, Chun HS, Choi SW, Park TJ. Development of aflatoxin B-1 aptasensor based on wide-range fluorescence detection using graphene oxide quencher. Colloid Surface B. 2017;154:27–32. https://doi.org/10.1016/j.colsurfb.2017.03.010.
Funding
This work was supported by the Department of Sciences and Technology of Jilin Province(20200403044SF), the State Key Research and Development Plan (2018YFD0400104), the Higher Education Discipline Innovation Project (111 Project, D18012), the Department of Education of Jilin Province (JJKH20200516KJ), and the Training Program of Ronghe College, Yanbian University (012021081).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qi, X., Lv, L., Wei, D. et al. Detection of aflatoxin B1 with a new label-free fluorescence aptasensor based on PVP-coated single-walled carbon nanohorns and SYBR Gold. Anal Bioanal Chem 414, 3087–3094 (2022). https://doi.org/10.1007/s00216-022-03938-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-022-03938-3