Abstract
Rationale
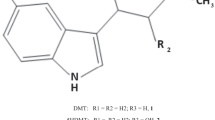
Oleoyl glycine, a little studied fatty acid amide similar in structure to anandamide, interferes with nicotine addiction in mice and acute naloxone-precipitated morphine withdrawal (MWD) in rats. Because endogenous oleoyl glycine is subject to rapid enzymatic deactivation, we evaluated the potential of more stable analogs to interfere with opiate withdrawal.
Objectives
The potential of monomethylated oleoyl glycine (oleoyl alanine, HU595) to interfere with somatic and aversive effects of acute naloxone-precipitated MWD, its duration, and mechanism of action was assessed in male Sprague Dawley rats. The potential of dimethylated oleoyl glycine (HU596) to interfere with the aversive effects of naloxone-precipitated MWD was also investigated.
Results
Oleoyl alanine (HU595) interfered with somatic and aversive effects produced by naloxone-precipitated MWD at equivalent doses (1 and 5 mg/kg, i.p.) as we have reported for oleoyl glycine; however, oleoyl alanine produced a longer lasting (60 min) interference, yet did not produce rewarding or aversive effects on its own and did not modify locomotor activity. HU596 was not effective. The interference with aversive effects of naloxone-precipitated MWD by oleoyl alanine was prevented by both a PPARα antagonist and a CB1 receptor antagonist. Accordingly, the compound was found to inhibit FAAH and activate PPARα in vitro. Finally, oleoyl alanine also reduced acute naloxone-precipitated MWD anhedonia, as measured by decreased saccharin preference.
Conclusions
Oleoyl alanine (also an endogenous fatty acid) may be a more stable and effective treatment for opiate withdrawal than oleoyl glycine.






Similar content being viewed by others
References
Abadji V, Lin S, Taha G, Griffin G, Stevenson LA, Pertwee RG, Makriyannis A (1994) ®-Methanandamide a chiral novel anandamide possessing higher potency and metabolic stability. J. Med Chem 37:1889–1893. https://doi.org/10.1021/jm00038a020
Abdolahi A, Acosta G, Breslin FJ, Hemby SE, Synch WJ (2010) Incubation of nicotine seeding is associated with enhanced protein kinase A-regulated signaling of dopamine- and cAMP regulated phosphorprotein of 32 kDa in the insular cortex. Eur J Neurosci 31:733–741. https://doi.org/10.1111/j.1460-9568.2010.07114.x
Chang JW, Niphakis MJ, Lum KM, Cognetta AB, Wang C, Matthews ML, Niessen S, Buczynski MW, Parsons LH, Cravatt BF (2012) Highly selective inhibitors of monoacylglycerol lipase bearing a reactive group that is bioisosteric with endocannabinoid substrates. Chem Biol 19:579–588
Chang JW, Cognetta AB, Niphakis MJ, Cravatt BF (2013) Proteome-wide reactivity profiling identifies diverse carbamate chemotypes tuned for serine hydrolase inhibition. ACS Chem Biol 8:1590–1599
Contreras M, Ceric F, Torrealba F (2007) Inactivation of the interoceptive insula disrupts drug craving and malaise induced by lithium. Science 26:655–658. https://doi.org/10.1126/science.1145590
Donvito G, Piscitelli F, Muldoon P, Jackson A, Vitale RM, D’Aniello E et al (2019) N-Oleoyl-glycine reduces nicotine reward and withdrawal in mice. Neuropharmacology 148:320–331. https://doi.org/10.1016/j.neuropharm.2018.03.020
Grill HJ, Norgren R (1978) The taste reactivity test: 1 Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res 143:263–279. https://doi.org/10.1016/0006-8993(78)90568-1
Huang SM, Bisogno T, Petros TJ, Chang SY, Zavitsanos PA, Zipkin RE, Sivakuma R, Coop A, Maeda DY, De Petrocellis L, Burstein S, Di Marzo V, Walker JM (2001) Identification of a new class of molecules, the arachidonyl amino acids, and characterization of one member that inhibits pain. J Biol Chem 276:42639–42644. https://doi.org/10.1074/jbc.M107351200
Leishman E, Makie K, Luquet S, Bradshaw HB (2016) Lipidomics profile of a NAPE-PLD KO mouse provides evidence of a broader role of this enzyme in lipid metabolism in the brain. Biochim Biophys Acta 1861:491–500. https://doi.org/10.1016/j.bbalip.2016.03.003
Li M, Hou YY, Lu B, Chen J, Chi ZQ, Liu JG (2009) Expression pattern of neural synaptic plasticity marker-Arc in different brain regions induced by conditioned drug withdrawal from acute morphine-dependent rats. Acta Pharmacologia Sinica 30:282–290. https://doi.org/10.1038/aps.2009.10
Li CL, Zhu N, Meng X-L, Li Y-H, Sui N (2013) Effects of inactivating the agranular or granular insular cortex on the acquisition of the morphine-induced conditioned place preference and naloxone-precipitated conditioned place aversion in rats. J Psychopharmacol 27:837–844. https://doi.org/10.1177/0269881113492028.(
Limebeer CL, Vemuri VK, Bedard H, Lang ST, Ossenkopp KP, Makriyannis A, Parker LA (2010) Inverse agonism of cannabinoid CB1 receptors potentiates LiCl-induced nausea in the conditioned gaping model in rats. Br J Pharmacol 161:336–349. https://doi.org/10.1111/j.1476-5381.2010.00885.x
Limebeer CL, Rock EM, Sharkey KA, Parker LA (2018) Nausea-induced 5-HT release in the interoceptive insular cortex and regulation by monoacylglycerol lipase (MAGL) inhibition and cannabidiol. eNeuro 5(4):ENEU18.2018. https://doi.org/10.1523/ENEURO.0256-18.2018
McDonald RV, Parker LA, Siegel S (1997) Conditioned sucrose aversions produced by naloxone-precipitated withdrawal from acutely administered morphine. Pharmacol Biochem Behav 58:1003–1008. https://doi.org/10.1016/S0091-3057(97)00313-4
Napadow V, Sheehan JD, Kim J, Lacount LT, Park K, Kaptchuk TJ et al (2013) The brain circuitry underlying the temporal evolution of nausea in humans. Cereb Cortex 23:806–813. https://doi.org/10.1093/cercor/bhs073
Naqvi NH, Rudrauf D, Damasio H, Bechara A (2007) Damage to the insula disrupts addiction to cigarette smoking. Science 315:531–534. https://doi.org/10.1126/science.1135926
Parker LA (2014) Conditioned flavor avoidance and conditioned gaping: rat models of conditioned nausea. Eur J Pharmacol 722:122–133. https://doi.org/10.1016/j.ejphar.2013.09.070
Penfield W, Faulk ME (1955) The insula: further observations on its function. Brain 78:445–470. https://doi.org/10.1093/brain/78.4.445
Petrie GN, Wills KL, Piscitelli F, Smoum R, Limebeer CL, Rock EM et al (2019) Oleoyl glycine: interference with the aversive effects of acute naloxone-precipitated morphine withdrawal, but not morphine reward, in male Sprague-Dawley rats. Psychopharmacology (Berl) 236:2623-2633.N. https://doi.org/10.1007/s00213-019-05237-9
Rock EM, Ayoub S, Limebeer CL, Gene A, Wills K, DeVuono M, Smoum R, Di Marzo V, Lichtman AH, Mechoulam R, Parker LA (2020) Acute naloxone-precipitated morphine withdrawal elicits nausea-like somatic behaviors in rats in a manner suppressed by N-oleoylglycine. Psychopharmacology. 237:375-384.https://doi.org/10.1007/s00213-019-05373-2
Scheggi S, De Montis MG, Gambarana C (2018) Making sense of rodent models of anhedonia. The International Journal of Neuropsychopharmacology 21:1049–1065. https://doi.org/10.1093/ijnp/pyy083
Sclocco R, Kim J, Garcia RG, Sheehan JD, Beissner F, Bianchi AM, Cerutti S, Kuo B, Barbieri R, Napadow V (2016) Brain circuitry supporting multi-organ autonomic outflow in response to nausea. Cereb Cortex 26:485–497. https://doi.org/10.1109/EMBC.2015.7319439
Tortoriello G, Rhodes BP, Takacs SM, Stuart JM, Basnet A, Raboune S, Widlanski TS, Doherty P, Harkany T, Bradshaw HB (2013) Targeted lipidomics in Drosophila melanogaster identifies novel 2-monoacylglycerols and N-acylamides. PLoS One 8:e67865. https://doi.org/10.1371/journal.pone.0067865
Tuerke KJ, Limebeer CL, Fletcher PJ, Parker LA (2012) Double dissociation between regulation of conditioned disgust and taste avoidance by serotonin availability at the 5-HT3 receptor in the posterior and anterior insular cortex. J Neurosci 32:13709–13717. https://doi.org/10.1523/JNEUROSCI.2042-12.2012
Wills KL, Petrie GN, Millett G, Limebeer CL, Rock EM, Niphakis MJ, Cravatt BF, Parker LA (2016) Double dissociation of monoacylglycerol lipase inhibition and CB1 antagonism in the central amygdala, basolateral amygdala, and the interoceptive insular cortex on the affective properties of acute naloxone-precipitated morphine withdrawal in rats. Neuropsychopharmacology 41:1865–1873. https://doi.org/10.1038/npp.2015.356
Funding
The research was supported by grants from the Natural Sciences and Engineering Research Council of Canada (03629) and PlantExt to LAP and from PlantExt to RM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal procedures were approved by the Animal Care Committee of the University of Guelph and adhered to the guidelines of the Canadian Council of Animal Care.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ayoub, S.M., Smoum, R., Farag, M. et al. Oleoyl alanine (HU595): a stable monomethylated oleoyl glycine interferes with acute naloxone precipitated morphine withdrawal in male rats. Psychopharmacology 237, 2753–2765 (2020). https://doi.org/10.1007/s00213-020-05570-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05570-4