Abstract
Rationale
Betel quid (BQ) is a common addictive substance in many Asian countries. However, few studies have focused on the influences of BQ on the brain. It remains unclear how BQ can affect structural brain abnormalities in BQ chewers.
Objectives
We aimed to use generalized q-sampling imaging (GQI) to evaluate the impact of the neurological structure of white matter caused by BQ.
Methods
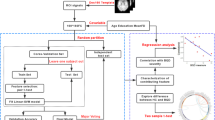
The study population comprised 16 BQ chewers, 15 tobacco and alcohol controls, and 17 healthy controls. We used GQI with voxel-based statistical analysis (VBA) to evaluate structural brain and connectivity abnormalities in the BQ chewers compared to the tobacco and alcohol controls and the healthy controls. Graph theoretical analysis (GTA) and network-based statistical (NBS) analysis were also performed to identify the structural network differences among the three groups.
Results
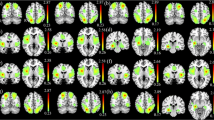
Using GQI, we found increases in diffusion anisotropy in the right anterior cingulate cortex (ACC), the midbrain, the bilateral angular gyrus, the right superior temporal gyrus (rSTG), the bilateral superior occipital gyrus, the left middle occipital gyrus, the bilateral superior and inferior parietal lobule, and the bilateral postcentral and precentral gyrus in the BQ chewers when compared to the tobacco and alcohol controls and the healthy controls. In GTA and NBS analyses, we found more connections in connectivity among the BQ chewers, particularly in the bilateral anterior cingulum.
Conclusions
Our results provided further evidence indicating that BQ chewing may lead to brain structure and connectivity changes in BQ chewers.





Similar content being viewed by others
References
Bales A, Peterson MJ, Ojha S, Upadhaya K, Adhikari B, Barrett B (2009) Associations between betel nut (Areca catechu) and symptoms of schizophrenia among patients in Nepal: a longitudinal study. Psychiatry Res 169:203–211
Benegal V, Rajkumar RP, Muralidharan K (2008) Does areca nut use lead to dependence? Drug Alcohol Depend 97:114–121
Bigler ED, Mortensen S, Neeley ES, Ozonoff S, Krasny L, Johnson M, Lu J, Provencal SL, McMahon W, Lainhart JE (2007) Superior temporal gyrus, language function, and autism. Dev Neuropsychol 31:217–238
Blood AJ, Iosifescu DV, Makris N, Perlis RH, Kennedy DN, Dougherty DD, Kim BW, Lee MJ, Wu S, Lee S, Calhoun J, Hodge SM, Fava M, Rosen BR, Smoller JW, Gasic GP, Breiter HC, Phenotype Genotype Project on Addiction and Mood Disorder (2010) Microstructural abnormalities in subcortical reward circuitry of subjects with major depressive disorder. PLoS One 5:e13945
Buckner RL, Andrews-Hanna JR, Schacter DL (2008) The brain’s default network. Ann N Y Acad Sci 1124:1–38
Bullmore ET, Bassett DS (2011) Brain graphs: graphical models of the human brain connectome. Annu Rev Clin Psychol 7:113–140
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198
Chen CH, Chen WJ, Cheng AT (2005) New approach to the validity of the Alcohol Use Disorders Identification Test: stratum-specific likelihood ratios analysis. Alcohol Clin Exp Res 29:602–608
Chen F, Zhong Y, Zhang Z, Xu Q, Liu T, Pan M, Li J, Lu G (2015) Gray matter abnormalities associated with betel quid dependence: a voxel-based morphometry study. Am J Transl Res 7:364–374
Chen VC, Shen CY, Liang SH, Li ZH, Tyan YS, Liao YT, Huang YC, Lee Y, McIntyre RS, Weng JC (2016) Assessment of abnormal brain structures and networks in major depressive disorder using morphometric and connectome analyses. J Affect Disord 205:103–111
Cheng Y, Xu J, Yu H, Nie B, Li N, Luo C, Li H, Liu F, Bai Y, Shan B, Xu L, Xu X (2014) Delineation of early and later adult onset depression by diffusion tensor imaging. PLoS One 9:e112307
Chu NS (2002) Neurological aspects of areca and betel chewing. Addict Biol 7:111–114
Chuang C, Chang C, Chang C (2007) The workplace relevant factors of betel quid chewing among transportation workers in central Taiwan. Taiwan Public Health 26:433–442
Ersche KD, Jones PS, Williams GB, Turton AJ, Robbins TW, Bullmore ET (2012) Abnormal brain structure implicated in stimulant drug addiction. Science 335:601–604
Ersche KD, Jones PS, Williams GB, Smith DG, Bullmore ET, Robbins TW (2013a) Distinctive personality traits and neural correlates associated with stimulant drug use versus familial risk of stimulant dependence. Biol Psychiatry 74:137–144
Ersche KD, Williams GB, Robbins TW, Bullmore ET (2013b) Meta-analysis of structural brain abnormalities associated with stimulant drug dependence and neuroimaging of addiction vulnerability and resilience. Curr Opin Neurobiol 23:615–624
Everitt BJ, Parkinson JA, Olmstead MC, Arroyo M, Robledo P, Robbins TW (1999) Associative processes in addiction and reward the role of amygdala-ventral striatal subsystems. Ann N Y Acad Sci 877:412–438
Fagerström K-O (1978) Measuring degree of physical dependence to tobacco smoking with reference to individualization of treatment. Addict Behav 3:235–241
Fornito A, Zalesky A, Bullmore ET (2010) Network scaling effects in graph analytic studies of human resting-state FMRI data. Front Syst Neurosci 4:22
Goldstein RZ, Bechara A, Garavan H, Childress AR, Paulus MP, Volkow ND (2009) The neurocircuitry of impaired insight in drug addiction. Trends Cogn Sci 13:372–380
Gong Q, He Y (2015) Depression, neuroimaging and connectomics: a selective overview. Biol Psychiatry 77:223–235
Greicius MD, Krasnow B, Reiss AL, Menon V (2003) Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci 100:253–258
Gupta PC, Ray CS (2004) Epidemiology of betel quid usage. Ann Acad Med Singap 33:31–36
Heatherton TF, Wagner DD (2011) Cognitive neuroscience of self-regulation failure. Trends Cogn Sci 15:132–139
Ho MC, Wang CK (2010) Can betel nut chewing affect the UFOV size after sleep deprivation? Chinese Journal of Psychology 52:445–456
Ho MC, Wang CK (2011) The effect of betel nut chewing on contour and object masking. Attention, Perception & Psychophysics 73:2583–2593
Ho MC, Li RH, Tang TC (2015) Betel nut chewing effects on sustained attention and inhibitory control after sleep deprivation. Aust J Psychol 67:222–230
Hosseini SM, Hoeft F, Kesler SR (2012) GAT: a graph-theoretical analysis toolbox for analyzing between-group differences in large-scale structural and functional brain networks. PLoS One 7:e40709
Huang C-L, Lin H-H, Wang H-H (2006) The psychometric properties of the Chinese version of the Fagerstrom test for nicotine dependence. Addict Behav 31:2324–2327
Huang X, Liu Z, Mwansisya TE, Pu W, Zhou L, Liu C, Chen X, Rohrbaugh R, Marienfeld C, Xue Z (2016) Betel quid chewing alters functional connectivity in frontal and default networks: a resting-state fMRI study. Journal of Magnetic Resonance Imaging.
IARC (2004) Betel-quid and areca-nut chewing and some areca-nut-derived nitrosamines IARC monographs on the evaluation of carcinogenic risks to humans, Lyon
Jensen JH, Helpern JA, Ramani A, Lu H, Kaczynski K (2005) Diffusional kurtosis imaging: the quantification of non-Gaussian water diffusion by means of magnetic resonance imaging. Magn Reson Med 53:1432–1440
Jou RJ, Minshew NJ, Keshavan MS, Vitale MP, Hardan AY (2010) Enlarged right superior temporal gyrus in children and adolescents with autism. Brain Res 1360:205–212
Koob GF (2006) The neurobiology of addiction: a neuroadaptational view relevant for diagnosis. Addiction 101:23–30
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacology 35:217–238
Lee CH, Chiang SL, Ko AMS, Hua CH, Tsai MH, Warnakulasuriya S, Ibrahim SO, Zain RB, Ling TY, Huang CL (2014) Betel-quid dependence domains and syndrome associated with betel-quid ingredients among chewers: an Asian multi-country evidence. Addiction 109:1194–1204
Li RH, Ho MC, Tang TC, Chang CF (2012) Development of the Betel Nut Dependency Scale (BNDS). Chinese Journal of Psychology 54:331–348
Liu T, Li J, Huang S, Zhao Z, Yang G, Pan M, Li C, Chen F, Pan S (2015) Neurochemical abnormalities in anterior cingulate cortex on betel quid dependence: a 2D (1)H MRS investigation. Am J Transl Res 7:2795–2804
Liu T, Li J, Zhao Z, Zhong Y, Zhang Z, Xu Q, Yang G, Lu G, Pan S, Chen F (2016a) Betel quid dependence is associated with functional connectivity changes of the anterior cingulate cortex: a resting-state fMRI study. J Transl Med 14:1
Liu T, Li J-j, Zhao Z-y, Yang G-s, Pan M-j, Li C-q, Pan S-y, Chen F (2016b) Altered spontaneous brain activity in betel quid dependence: a resting-state functional magnetic resonance imaging study. Medicine 95.
Lo CY, He Y, Lin CP (2011) Graph theoretical analysis of human brain structural networks. Rev Neurosci 22:551–563
McClernon FJ, Kozink RV, Lutz AM, Rose JE (2009) 24-h smoking abstinence potentiates fMRI-BOLD activation to smoking cues in cerebral cortex and dorsal striatum. Psychopharmacology 204:25–35
MHW (2008) 905 betel nut chewing
Moerel M, De Martino F, Formisano E (2014) An anatomical and functional topography of human auditory cortical areas. Front Neurosci 8:225
Naqvi NH, Rudrauf D, Damasio H, Bechara A (2007) Damage to the insula disrupts addiction to cigarette smoking. Science 315:531–534
Noël X, Brevers D, Bechara A (2013) A neurocognitive approach to understanding the neurobiology of addiction. Current opinion in neurobiology.
Ota M, Noda T, Sato N, Hattori K, Hori H, Sasayama D, Teraishi T, Nagashima A, Obu S, Higuchi T, Kunugi H (2014) White matter abnormalities in major depressive disorder with melancholic and atypical features: a diffusion tensor imaging study. Psychiatry and clinical neurosciences.
Robinson TE, Berridge KC (2003) Addiction. Annu Rev Psychol 54:25–53
Rose JE, McClernon FJ, Froeliger B, Behm FM, Preud’homme X, Krystal AD (2011) Repetitive transcranial magnetic stimulation of the superior frontal gyrus modulates craving for cigarettes. Biol Psychiatry 70:794–799
Salamone JD, Correa M, Farrar A, Mingote SM (2007) Effort-related functions of nucleus accumbens dopamine and associated forebrain circuits. Psychopharmacology 191:461–482
Saunders JB, Aasland OG, Babor TF, Grant M (1993) Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 88:791–804
Shen CY, Tyan YS, Kuo LW, Wu CW, Weng JC (2015) Quantitative evaluation of rabbit brain injury after cerebral hemisphere radiation exposure using generalized q-sampling imaging. PLoS One 10:e0133001
Srivastava S, Bhatia MS, Bhargava SK, Kumari R, Chandra S (2015) A diffusion tensor imaging study using a voxel-based analysis, region-of-interest method to analyze white matter abnormalities in first-episode, treatment-naive major depressive disorder. The Journal of neuropsychiatry and clinical neurosciences: appineuropsych15050120.
Thomason ME, Thompson PM (2011) Diffusion imaging, white matter, and psychopathology. Annu Rev Clin Psychol 7:63–85
Tuch DS (2004) Q-ball imaging. Magn Reson Med 52:1358–1372
Verdejo-Garcia A, Clark L, Dunn BD (2012) The role of interoception in addiction: a critical review. Neuroscience & Biobehavioral Reviews.
Volkow ND, Wang G-J, Fowler JS, Tomasi D (2012) Addiction circuitry in the human brain. Annu Rev Pharmacol Toxicol 52:321
Walton ME, Behrens TE, Buckley MJ, Rudebeck PH, Rushworth MF (2010) Separable learning systems in the macaque brain and the role of orbitofrontal cortex in contingent learning. Neuron 65:927–939
Wang Y, Wang Q, Haldar JP, Yeh FC, Xie M, Sun P, Tu TW, Trinkaus K, Klein RS, Cross AH, Song SK (2011) Quantification of increased cellularity during inflammatory demyelination. Brain : a journal of neurology 134:3590–3601
Wang X, Cusick MF, Wang Y, Sun P, Libbey JE, Trinkaus K, Fujinami RS, Song SK (2014) Diffusion basis spectrum imaging detects and distinguishes coexisting subclinical inflammation, demyelination and axonal injury in experimental autoimmune encephalomyelitis mice. NMR Biomed 27:843–852
Watts DJ, Strogatz SH (1998) Collective dynamics of “small-world” networks. Nature 393:440–442
Wedeen VJ, Hagmann P, Tseng WY, Reese TG, Weisskoff RM (2005) Mapping complex tissue architecture with diffusion spectrum magnetic resonance imaging. Magn Reson Med 54:1377–1386
Winstock AR (2002) Areca nut-abuse liability, dependence and public health. Addict Biol 7:133–138
Winstock AR, Trivedy CR, Warnakulasuriya KA, Peters TJ (2000) A dependency syndrome related to areca nut use: some medical and psychological aspects among areca nut users in the Gujarat community in the UK. Addict Biol 5:173–179
Wise T, Radua J, Nortje G, Cleare AJ, Young AH, Arnone D (2015) Voxel-based meta-analytical evidence of structural disconnectivity in major depression and bipolar disorder. Biological psychiatry.
Yeh FC, Wedeen VJ, Tseng WY (2010) Generalized q-sampling imaging. IEEE Trans Med Imaging 29:1626–1635
Zhang J, Wang J, Wu Q, Kuang W, Huang X, He Y, Gong Q (2011) Disrupted brain connectivity networks in drug-naive, first-episode major depressive disorder. Biol Psychiatry 70:334–342
Zhang H, Schneider T, Wheeler-Kingshott CA, Alexander DC (2012) NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. NeuroImage 61:1000–1016
Zhang H, Wang Y, Lu T, Qiu B, Tang Y, Ou S, Tie X, Sun C, Xu K (2013) Differences between generalized q-sampling imaging and diffusion tensor imaging in the preoperative visualization of the nerve fiber tracts within peritumoral edema in brain. Neurosurgery 73:1044–1053 discussion 1053
Acknowledgements
The authors appreciate the full support from the Department of Medical Imaging, Chung Shan Medical University Hospital, Taichung, Taiwan. The authors would like to thank Jau-Yang Lin for his assistance in experimental preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent was obtained from all participants; the study was approved by the Institutional Review Board of Chung Shan Medical University Hospital. All participants underwent assessment established by psychologists at Chung Shan Medical University, Taichung, Taiwan.
Funding
This study was supported by the research programs NSC103-2420-H-040-001-MY2 and MOST105-2410-H-040-001-MY3, which were sponsored by the Ministry of Science and Technology, Taipei, Taiwan.
Conflict of interest
The authors declare that they have no conflict of interest.
Appendix
Appendix
The BNDS consists of the following three factors: craving and desire (e.g., if possible, I may chew BQ right now), withdrawal response (e.g., when BQ is not available, I feel so upset that my work and activities are disturbed), and tasting habits (e.g., I care about types, textures, and the feeling that comes from chewing BQ).The eigenvalues of the three factors accounted for 63.10% of total variances, and α coefficients of reliability were between 0.73 to 0.89. Most of model-fit indexes showed good fitting results (RMSEA = 0.070, SRMR = 0.038, AGFI = 0.90, GFI = 0.94, NFI = 0.97, NNFI = 0.98, CFI = 0.98, IFI = 0.98, RFI = 0.96), suggesting optimal construct validity of the scale. The scale also offered criterion-related validity for reference. For example, the BNDS score correlated positively to the number of days per week on which chewing occurred and the average number of BQ chewed per day. The BNDS score correlated negatively to the extent of willing to quit chewing BQ.
Rights and permissions
About this article
Cite this article
Weng, JC., Kao, TW., Huang, GJ. et al. Evaluation of structural connectivity changes in betel-quid chewers using generalized q-sampling MRI. Psychopharmacology 234, 1945–1955 (2017). https://doi.org/10.1007/s00213-017-4602-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4602-0