Abstract
Rationale
Intermittent delivery of an important commodity (e.g., food pellets) generates excessive behaviors as an adjunct to the schedule of reinforcement (adjunctive behaviors) that are hypothesized to be due to conflict between engaging and escaping a situation where reinforcement is delivered, but at suboptimal rates.
Objectives
This study characterized the endocrine correlates during schedule-induced polydipsia of water and ethanol using a longitudinal approach in non-human primates.
Methods
Plasma adrenocorticotropic hormone (ACTH) and cortisol were measured in samples from awake cynomolgus monkeys (Macaca fascicularis, 11 adult males) obtained at the onset, mid-day, and offset of their 12-h light cycle. The monkeys were induced to drink water and ethanol (4 % w/v, in water) using a fixed time (FT) 300-s interval schedule of pellet delivery. The induction fluid changed every 30 sessions in the following order: water, 0.5 g/kg ethanol, 1.0 g/kg ethanol, and 1.5 g/kg ethanol. Following induction, ethanol and water were concurrently available for 22 h/day.
Results
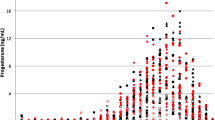
The FT 300-s schedule gradually increased ACTH, but not cortisol, during water induction to a plateau sustained throughout ethanol induction in every monkey. Upon termination of the schedule, ACTH decreased to baseline and cortisol below baseline. Diurnal ACTH and cortisol were unrelated to the dose of ethanol, but ACTH rhythm flattened at 0.5 g/kg/day and remained flattened.
Conclusions
The coincidence of elevated ACTH with the initial experience of drinking to intoxication may have altered the mechanisms involved in the transition to heavy drinking.



Similar content being viewed by others
References
Acosta G, Hasenkamp W, Daunais JB, Friedman DP, Grant KA, Hemby SE (2010) Ethanol self-administration modulation of NMDA receptor subunit and related synaptic protein mRNA expression in prefrontal cortical fields in cynomolgus monkeys. Brain Res 318:144–154
Brett LP, Levine S (1979) Schedule-induced polydipsia suppresses pituitary-adrenal activity in rats. J Comp Physiol Psychol 93:946–956
Born J, Fehm-Wolfsdorf G, Schiebe M, Rockstroh B, Fehm H-L, Voigt KH (1984) Dishabituating effects of an ACTH 4–9 analog in a vigilance task. Pharmacol Biochem Behav 21:513–519
Bowman ME, Lopata A, Jaffe RB, Golos TG, Wickings J, Smith R (2001) Corticotropin-releasing hormone-binding protein in primates. Am J Primatol 53:123–130
Boyd KN, Kumar S, O’Buckley TK, Porcu P, Morrow AL (2010) Ethanol induction of steroidogenesis in rat adrenal and brain is dependent upon pituitary ACTH release and de novo adrenal StAR synthesis. J Neurochem 112:784–796
Brown TG, Flory RK (1972) Schedule-induced escape from fixed interval reinforcement. J Exp Anal Behav 17:395–403
Chrousos GP (2009) Stress and disorders of the stress system. Nat Rev Endocrinol 5:374–381
Cirulli F, van Oers H, De Kloet ER, Levine S (1994) Differential influence of corticosterone and dexamethasone on schedule-induced polydipsia in adrenalectomized rats. Behav Brain Res 65:33–39
Colwell CS (2011) Linking neural activity and molecular oscillation in the SCN. Nat Neurosci 12:553–569
Cuzon Carlson VC, Seabold GK, Helms CM, Garg N, Odagiri M, Rau AR, Daunais J, Alvarez VA, Lovinger DM, Grant KA (2011) Synaptic and morphological neuroadaptations in the putamen associated with long-term, relapsing alcohol drinking in primates. Neuropsychopharmacology 36:2513–2528
Dantzer R, Terlouw C, Morméde P, Le Moal M (1988) Schedule-induced polydipsia experience decreases plasma corticosterone levels but increases plasma prolactin levels. Physiol Behav 43:275–279
DeCarolis NA, Myracle A, Erbach J, Glowa J, Flores P, Riley AL (2003) Strain-dependent differences in schedule-induced polydipsia: an assessment in Lewis and Fischer rats. Pharmacol Biochem Behav 74:755–763
De Wied D (1961) The significance of the antidiuretic hormone in the release mechanism of corticotropin. Endocrinology 68:956–970
Falk JL (1961) Production of polydipsia in normal rats by an intermittent food schedule. Science 133:195–196
Falk JL (1977) The origin and function of adjunctive behavior. Anim Learn Behav 5:325–335
Falk JL (1998) Drug abuse as an adjunctive behavior. Drug Alcohol Dep 52:91–98
Flores P, Pellón R (2000) Antipunishment effects of diazepam on two levels of suppression of schedule-induced drinking in rats. Pharmacol Biochem Behav 67:207–214
Flory RK (1971) The control of schedule-induced polydipsia: frequency and magnitude of reinforcement. Learn Motiv 2:215–227
Fry W, Kelleher RT, Cook L (1960) A mathematical index of performance on fixed-interval schedules of reinforcement. J Exp Anal Behav 3:193–199
Gallagher TF, Yoshida K, Roffwarg HD, Fukushima DK, Weitzman ED, Hellman L (1973) ACTH and cortisol secretory patterns in man. J Clin Endocrinol Metab 36:1058–1068
Grant KA, Johanson CE (1988) The nature of the scheduled reinforcer and adjunctive drinking in nondeprived rhesus monkeys. Pharmacol Biochem Behav 29:295–301
Grant KA, Leng X, Green HL, Szeliga KT, Rogers LSM, Gonzales SW (2008) Drinking typography established by scheduled induction predicts chronic heavy drinking in a monkey model of ethanol self-administration. Alcohol Clin Exp Res 32:1824–1838
Helms CM, McClintick M, Grant KA (2012a) Social rank, chronic ethanol self-administration and hypothalamic–pituitary–adrenal axis response in monkeys. Psychopharmacology 224:133–143
Helms CM, Rossi DJ, Grant KA (2012b) Neurosteroid influences on sensitivity to ethanol. Front Endocrinol 3:1–19
Hermes MLHJ, Coderre EM, Buijs RM, Renaud LP (1996) GABA and glutamate mediate rapid neurotransmission from suprachiasmatic nucleus to hypothalamic paraventricular nucleus in rat. J Physiol 496:749–757
Herod SM, Dettmer AM, Novak MA, Meyer JS, Cameron JL (2011) Sensitivity to stress-induced reproductive dysfunction is associated with a selective but not a generalized increase in activity of the adrenal axis. Am J Physiol Endocrinol Metab 300:E28–E36
Holgert H, Åman K, Cozzari C, Hartman BK, Brimijoin S, Emson P, Goldstein M, Hökfelt T (1995) The cholinergic innervation of the adrenal gland and its relation to enkephalin and nitric oxide synthase. Neuroreport 6:2576–2580
Holgert H, Dagerlind Å, Hökfelt T (1998) Immunohistochemical characterization of the peptidergic innervation of the rat adrenal gland. Horm Met Res 30:315–322
Knott TK, Dopico AM, Dayanithi G, Lemos J, Treistman SN (2002) Integrated channel plasticity contributes to alcohol tolerance in neurohypophysial terminals. Mol Pharmacol 62:135–142
Krystal JH, Petrakis IL, Krupitsky E, Schütz C, Trevisan L, S’Souza C (2003) NMDA receptor antagonism and the ethanol intoxication signal: from alcoholism risk to pharmacotherapy. Ann NY Acad Sci 1003:176–184
Kuramoto H, Kondo H, Fujita T (1987) Calcitonin gene-related peptide (CGRP)-like immunoreactivity in scattered chromaffin cells and nerve fibers in the adrenal gland of rats. Cell Tissue Res 247:309–315
Krueger C, Tian L (2004) A comparison of the general linear mixed model and repeated measures ANOVA using a dataset with multiple missing data points. Biol Res Nurs 6:151–1516
Lamas E, Pellón R (1995) Food-deprivation effects on punished schedule-induced drinking in rats. J Exp Anal Behav 64:47–60
Levine E (2000) The hypothalamus as a major integrating center. In: Conn PM, Freeman ME (eds) Neuroendocrinology in physiology and medicine, 1st edn. Humana Press Inc., Totowa, pp 75–93
Levine R, Levine S (1989) Role of the pituitary-adrenal hormones in the acquisition of schedule-induced polydipsia. Behav Neurosci 103:621–637
Lin W, Singer G, Irby D (1990) Effect of hypophysectomy on schedule-induced wheel running. Pharmacol Biochem Behav 35:739–742
Lin W, Singer G, Papasava M (1988) The role of adrenal corticosterone in schedule-induced wheel running. Pharmacol Biochem Behav 30:101–106
López-Grancha M, López-Crespo G, Venero C, Cañadas F, Sánchez-Santed F, Sandi C, Flores P (2006) Differences in corticosterone level due to inter-food interval length: implications for schedule-induced polydipsia. Horm Behav 49:166–172
Lydersen T, Perkins D, Thome S, Lowman E (1980) Choice of timeout during response-independent food schedules. J Exp Anal Behav 33:59–76
McElroy B, Zakaria A, Glass JD, Prosser RA (2009) Ethanol modulates mammalian circadian clock phase resetting through extrasynaptic GABA receptor activation. Neurosci 164:842–848
Meier AH (1976) Daily variation in concentration of plasma corticosteroid in hypophysectomized rats. Endocrinology 98:1475–1479
Mittleman G, Blaha CD, Phillips AG (1992) Pituitary-adrenal and dopaminergic modulation of schedule-induced polydipsia: behavioral and neurochemical evidence. Behav Neurosci 106:408–420
Mittleman G, Jones GH, Robbins TW (1988) The relationship between schedule-induced polydipsia and pituitary-adrenal activity: pharmacological and behavioral manipulations. Behav Brain Res 28:315–324
Nader MA, Woolverton WL (1992) Further characterization of adjunctive behavior generated by schedules of cocaine self-administration. Behav Pharmacol 3:65–74
National Research Council (1996) Guide for the care and use of laboratory animals. National Academy Press, Washington, D.C, p 125
Pederson RC, Brownie AC, Ling N (1980) Pro-adrenocorticotropin/endorphin-derived peptides: coordinate action on adrenal steroidogenesis. Science 208:1044–1046
Pitts RC, Malagodi EF (1996) Effects of reinforcement amount on attack induced under a fixed-interval schedule in pigeons. J Exp Anal Behav 65:93–110
Platt B, Beyer CE, Schechter LE, Rosenzweig-Lipson S (2008) Schedule-induced polydipsia: a rat model of obsessive-compulsive disorder. Curr Protoc Neurosci 43:9.27.1–9.27.8
Roper TJ (1978) Diversity and substitutability of adjunctive activities under fixed-interval schedules of food reinforcement. J Exp Anal Behav 30:83–96
Samson HH, Falk JL (1974) Schedule-induced ethanol polydipsia: enhancement by saccharin. Pharmacol Biochem Behav 2:835–838
Shelton KL, Young JE, Grant KA (2001) A multiple schedule model of limited access drinking in the cynomolgus macaque. Behav Pharmacol 12:559–573
Shimura M, Harata N, Tamai M, Akaike N (1996) Allosteric modulation of GABAA receptors in acutely dissociated neurons of the suprachiasmatic nucleus. Am J Physiol 270:C1726–C1734
Shively CA (1998) Social subordination stress, behavior, and central monoaminergic function in female cynomolgus monkeys. Biol Psychiatry 44:882–891
Shively CA, Grant KA, Register TC (2002) Effects of long-term moderate alcohol consumption on agonistic and affiliative behavior of socially housed female cynomolgus monkeys (Macaca fascicularis). Psychopharmacology 165:1–8
Spruijit BM, Josephy M, Van Rijzingen I, Maaswinkel H (1994) The ACTH(4–9) analog Org2766 modulates the behavioral changes induced by NMDA and the NMDA receptor antagonist AP5. J Neurosci 14:3225–3230
Stöhr T, Szuran T, Welzl H, Pliska V, Feldon J, Pryce CR (2000) Lewis/Fischer rat strain differences in endocrine and behavioural responses to environmental challenge. Pharmacol Biochem Behav 67:809–819
Tazi A, Dantzer R, Morméde P, Le Moal M (1986) Pituitary-adrenal correlates of schedule-induced polydipsia and wheel running in rats. Behav Brain Res 19:249–256
Ulrich-Lai YM et al (2006) Adrenal splanchnic innervation contributes to the diurnal rhythm of plasma corticosterone in rats by modulating adrenal sensitivity to ACTH. Am J Physiol Regul Integr Comp Physiol 290:R1128–R1135
van Wimersma Greidanus TB, Croiset G, de Wied D (2000) Neuroendocrine regulation of learning and memory. In: Conn PM, Freeman ME (eds) Neuroendocrinology in physiology and medicine. Humana Press Inc, Totowa, pp 353–370
Vivian JA, Green HL, Young JE, Majerksy LS, Thomas BW, Shively CA, Tobin JR, Nader MA, Grant KA (2001) Induction and maintenance of ethanol self-administration in cynomolgus monkeys (Macaca fascicularis): long-term characterization of sex and individual differences. Alcohol Clin Exp Res 25:1087–1097
Vivian JA, Waters CA, Szeliga KT, Jordan K, Grant KA (2002) Characterization of the discriminative stimulus effects of N-methyl-D-aspartate ligands under different ethanol training conditions in the cynomolgus monkey (Macaca fascicularis). Psychopharmacology 162:273–281
Wallace M, Singer G, Finlay J, Gibson S (1983) The effects of 6-OHDA lesions of the nucleus accumbens septum on schedule-induced drinking, wheel running and corticosterone levels in the rat. Pharmacol Biochem Behav 18:129–136
Wand GS, Dobs AS (1991) Alterations in the hypothalamic–pituitary–adrenal axis in actively drinking alcoholics. J Clin Endocrinol Metab 72:1290–1295
Watts AG, Swanson LW, Sanchez-Watts G (1987) Efferent projections of the suprachiasmatic nucleus: I. Studies using anterograde transport of Phaseolus vulgaris leucoagglutinin in the rat. J Comp Neurol 258:204–229
Wolthuis OL, de Wied D (1976) The effect of ACTH-analogues on motor behavior and visual evoked responses in rats. Pharmacol Biochem Behav 4:273–278
Woods RJ, Grossman A, Saphier P, Kennedy K, Ur E, Behan D, Potter E, Vale W, Lowry PJ (1994) Association of human corticotropin-releasing hormone to its binding protein in blood may trigger clearance of the complex. J Clin Endocrinol Metab 78:73–76
Acknowledgments
The authors wish to thank Erin E. Shannon, Sarah Thornton, Natalie Maners, and Dr. Patrizia Porcu for research and technical expertise. This research was supported by NIH Grants AA109431, AA010760, OD011092, and AA013510.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Helms, C.M., Gonzales, S.W., Green, H.L. et al. Diurnal pituitary-adrenal activity during schedule-induced polydipsia of water and ethanol in cynomolgus monkeys (Macaca fascicularis). Psychopharmacology 228, 541–549 (2013). https://doi.org/10.1007/s00213-013-3052-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3052-6