Abstract
Rationale
Stress may elevate ethanol drinking and anxiety associated with ethanol drinking. Studies to identify relevant neurobiological substrates are needed.
Objective
To assess roles of brain regions in corticotrophin releasing factor (CRF) effects on stressor-enhanced, ethanol deprivation-induced drinking and anxiety-like behavior.
Methods
Ethanol-preferring rats (P rats) were exposed to three cycles of a two-bottle choice paradigm with two 2-day deprivation periods that included 1 h exposure to a restraint stressor. To assess the role of CRF and to identify relevant brain regions, a CRF-1 receptor antagonist (SSR125543; 10 ug) was injected into the nucleus accumbens (NAC), amygdala (Amyg), or dorsal raphe nucleus (DRN) prior to exposure to the restraint stressor. In a second study, CRF (0.5 ug) was injected into one of these regions, or the ventral tegmental area (VTA), or paraventricular nucleus of the hypothalamus (PVN).
Results
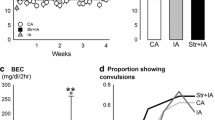
Applying the restraint stressor during deprivation increased voluntary intake and sensitized anxiety-like behavior. Antagonist injection into the NAC prevented increased drinking without affecting anxiety-like behavior, whereas injection into the Amyg or DRN prevented the anxiety-like behavior without affecting drinking. To confirm CRF actions in the stressor effect, CRF was injected into selected brain regions. NAC injections (but not the VTA, Amyg, DRN, or PVN) facilitated drinking but did not change anxiety-like behavior. Injections into the DRN or Amyg (but not PVN or VTA) enhanced anxiety-like behavior.
Conclusions
Results emphasize that a restraint stressor elevates ethanol intake and sensitizes ethanol deprivation-induced anxiety-like behavior through CRF1 receptors in the NAC and Amyg/DRN, respectively.





Similar content being viewed by others
References
Bale TL, Vale WW (2004) CRF and CRF receptors: role in stress responsivity and other behaviors. Ann Rev Pharmacol Toxicol 44:525–557
Ballenger JC, Post RM (1978) Kindling as a model for alcohol withdrawal syndromes. Br J Psychiatr 133:1–14
Besheer J, Grondin JJ, Cannady R, Sharko AC, Faccidomo S, Hodge CW (2010) Metabotropic glutamate receptor 5 activity in the nucleus accumbens is required for the maintenance of ethanol self-administration in a rat genetic model of high alcohol intake. Biol Psychiatry 67(9):812–822
Brady, Lydiard (1993) The association of alcoholism and anxiety. Psychiatr Q 64(2):135–149
Breese GR, Knapp DJ, Overstreet DH (2004) Stress sensitization of ethanol withdrawal-induced reduction in social interaction: inhibition by CRF-1 and benzodiazepine receptor antagonists and a 5-HT1A-receptor agonist. Neuropsychopharmacology 29:470–482
Breese GR, Chu K, Dayas CV, Funk D, Knapp DJ, Koob GF, Lê DA, O’Dell LE, Overstreet DH, Roberts AJ, Sinha R, Valdez GR, Weiss F (2005a) Stress enhancement of craving during sobriety: risk of relapse. Alcohol Clin Exp Res 29:185–195
Breese GR, Overstreet DH, Knapp DJ (2005b) Conceptual framework for the etiology of alcoholism—a “kindling”/stress hypothesis. Psychopharmacology 178:367–380
Breese GR, Overstreet DH, Knapp DJ, Navarro M (2005c) Prior multiple ethanol withdrawals enhance stress-induced anxiety-like behavior: inhibition by CRF1- and benzodiazepine receptor antagonists and a 5-HY1A-receptor agonist. Neuropsychopharmacology 30:1662–1669
Breese GR, Sinha R, Heilig M (2011) Chronic alcohol neuroadaptation and stress contribute to susceptibility for alcohol craving and relapse. Pharmacol Ther 129(2):149–171
Champagne F, Kirouac G (1987) Effects of unavoidable electric shocks on voluntary alcohol consumption in the rat. Percept Mot Skills 64:335–338
Chaudhri N, Sahuque LL, Cone JJ, Janak PH (2008) Reinstated ethanol-seeking in rats is modulated by environmental context and requires the nucleus accumbens core. Eur J Neurosci 28(11):2288–2298
Chester JA, Blose AM, Zweifel M, Froehlich JM (2004) Effects of stress on alcohol consumption in rats selectively bred for high or low alcohol drinking. Alcohol Clin Exp Res 28:385–393
Dunn AJ, Berridge CW (1990) Physiological and behavioral response to corticotropin-releasing factor administration: is CRF a mediator of anxiety-like behavior or stress responses? Brain Res Brain Res Rev 15:71–100
Eiler WJ 2nd, June HL (2007) Blockade of GABA(A) receptors within the extended amygdala attenuates D(2) regulation of alcohol-motivated behaviors in the ventral tegmental area of alcohol-preferring (P) rats. Neuropharmacology 52(8):1570–1579
File SE, Hyde JR (1978) Can social interaction be used to measure anxiety? Br J Pharmacol 62(1):19–24
File SE, Seth P (2003) A review of 25 years of the social interaction 724 test. Eur J Pharmacol 463:35–53
Funk D, Li Z, Le AD (2006a) Effects of environmental and pharmacological stressors on c-fos and corticotropin releasing factor mRNA in rat brain: relationship to the reinstatement of alcohol seeking. Neuroscience 138:235–243
Funk CK, O’Dell LE, Crawford EF, Koob GF (2006b) Corticotropin-releasing factor within the central nucleus of the amygdala mediates enhanced ethanol self-administration in withdrawn, ethanol-dependent rats. J Neurosci 26:11324–11332
Gremel CH, Cunningham CL (2008) Roles of the nucleus accumbens amygdala in the acquisition and expression of ethanol conditioned behavior in mice. J Neurosci 28(5):1076–1084
Griebel G, Simiand J, Steinberg R, Jung M, Gully D, Roger P, Geslin M, Scatton B, Maffrand JP, Soubrié P (2002) 4-(2-Chloro-4- methoxy-5-methylphenyl)-N-[(1S)-2-cyclopropyl-1-(3-fluoro-4-methylphenyl)ethyl]5- methyl-N-(2-propynyl)-1, 3-thiazol-2-amine hydrochloride (SSR125543A), a potent and selective corticotrophin-releasing factor(1) receptor antagonist. II. Characterization in rodent models of stress-related disorders. J Pharmacol Exp Ther 301:333–345
Hauger RL, Risbrough V, Brauns O, Dautzenberg FM (2006) Corticotropin releasing factor (CRF) receptor signaling in the central nervous system: new molecular targets. CNS Neurol Disord Drug Targets 5(4):453–479
Heilig M, Koob GF (2007) A key role for corticotropin-releasing factor in alcohol dependence. Trends Neurosci 30:399–408
Hiroi R, Neumaier JF (2009) Estrogen decreases 5-HT1B autoreceptor mRNA in selective subregions of rat dorsal raphe nucleus: inverse association between gene expression and anxiety behavior in the open field. Neuroscience 158:456–464
Holahan MR, Kalin NH, Kelley AE (1997) Microinfusion of corticotropin-releasing factor into the nucleus accumbens shell results in increased behavior arousal and oral motor activity. Psychopharmacology 130:189–196
Huang MM, Overstreet DH, Knapp DJ, Angel R, Wills TA, Navarro M, Rivier J, Vale W, Breese GR (2010) Corticotropin-releasing factor (CRF) sensitization of ethanol withdrawal-induced anxiety like behavior is brain site specific and mediated by CRF-1 receptors: relation to stress induced sensitization. J Pharmacol Exp Ther 332:298–307
Imaki T, Shibasaki T, Hotta M, Demura H (1993) Intracerebroventricular administration of corticotropin releasing factor induces c-fos mRNA expression in brain regions related to stress responses: comparison with pattern of c-fos mRNA induction after stress. Brain Res 616:114–125
Kalivas PW, Duffy P, Latimer LG (1987) Neurochemical and behavioral effects of corticotropin-releasing factor in the ventral tegmental area of the rat. J Pharmacol Exp Ther 242:757–763
Kampov-Polevoy AB, Matthews DB, Gause L, Morrow AL, Overstreet DH (2000) P rats develop physical dependence on alcohol via voluntary drinking: changes in seizure thresholds, anxiety, and patterns of alcohol drinking. Alcohol Clin Exp Res 24:278–284
Knapp DJ, Overstreet DH, Angel RA, Navarro M, Breese GR (2007) The amygdala regulates the antianxiety-like behavior sensitization effect of flumazenil during repeated chronic ethanol or repeated stress. Alcohol Clin Exp Res 31:1872–1882
Knapp DJ, Whitman BA, Wills TA, Angel RA, Overstreet DH, Criswell HE, Ming Z, Breese GR (2011) Cytokine involvement in stress may depend on corticotrophin releasing factor to sensitize ethanol withdrawal anxiety. Brain Behav Immun (in press)
Koob GF (2003) Alcoholism, allostasis and beyond. Alcohol Clin Exp Res 27:232–243
Koob GF (2008) A role for brain stress systems in addiction. Neuron 59:11–34
Koob GF, Bloom FE (1985) Corticotropin-releasing factor and behavior. Fed Proc 44:259–263
Koob GF, Heinrichs SC (1999) A role for corticotropin releasing factor and urocortin in behavioral responses to stressors. Brain Res 848:141–152
Koob GF, Le Moal M (1997) Drug abuse: hedonic homeostatic dysregulation. Science 278:52–58
Koob GF, Kenneth Lloyd G, Mason BJ (2009) Development of pharmacotherapies for drug addiction: a Rosetta stone approach. Nat Rev Drug Discov 8(6):500–515
Lack AK, Ariwodala OJ, Chappell AM, Weiner JL, McCool BA (2008) Ethanol inhibition of kainate receptor-mediated excitatory neurotransmission in the rat basolateral nucleus of the amygdala. Neuropharmacology 55:661–668
Lee EH, Tsai MJ (1989) The hippocampus and amygdala mediate the locomotor stimulating effects of corticotropin-releasing factor in mice. Behav Neural Biol 52:412–423
Lowry CA, Hale MW, Plant A, Windle RJ, Shanks N, Wood SA, Ingram CD, Renner KJ, Lightman SL, Summers CH (2009) Fluoxetine inhibits corticotropin-releasing factor (CRF)-induced behavioural responses in rats. Stress 12:225–239
Lynch WJ, Kushner MG, Rawleigh JM, Fiszdon J, Carroll ME (1999) The effects of restraint stress on voluntary ethanol consumption in rats. Exp Clin Psychopharmacol 7:318–323
McBride WJ, Schultz JA, Kimpel MW, McClintick JN, Wang M, You J, Rodd ZA (2009) Differential effects of ethanol in the nucleus accumbens shell of alcohol-preferring (P), alcohol-non-preferring (NP) and Wistar rats: a proteomics study. Pharmacol Biochem Behav 92:304–313
Meloni EG, Reedy CL, Cohen BM, Carlezon WA Jr (2008) Activation of raphe efferents to the medial prefrontal cortex by CRF: correlation with anxiety-like behavior. Biol Psychiatry 63:832–839
Nadal R, Chappell AM, Samson HH (1998) Effects of nicotine and mecamylamine microinjection into the nucleus accumbens on ethanol and sucrose self-administration. Alcohol Clin Exp Res 22:1190–1198
Nie H, Rewal M, Gill TM, Ron D, Janak PH (2011) Extrasynaptic {delta}-containing GABAA receptors in the nucleus accumbens dorsomedial shell contribute to alcohol intake. Proc Natl Acad Sci USA 108(11):4459–4464
Overstreet DH, McArthur RA, Rezvani AH, Post C (1997) Selective inhibition of alcohol intake in diverse alcohol-preferring rat strains by the 5-HT2A antagonists amperozide and FG5974. Alcohol Clin Exp Res 21:1448–1454
Overstreet DH, Knapp DJ, Breese GR (2002) Accentuated decreases in social interaction in rats subjected to repeated ethanol withdrawals. Alcohol Clin Exp Res 26:1259–1269
Overstreet DH, Knapp DJ, Moy SS, Breese GR (2003) A 5-HT1A agonist and a 5-HT2C antagonist reduce social interaction deficit induced by multiple ethanol withdrawals in rats. Psychopharmacology 167:344–352
Overstreet DH, Knapp DJ, Breese GR (2004) Modulation of multiple ethanol withdrawal induced anxiety-like behavior by CRF and CRF1 receptors. Pharmacol Biochem Behav 77:405–413
Overstreet DH, Knapp DJ, Breese GR (2005) Pharmacological modulation of repeated ethanol withdrawal-induced anxiety-like behavior differs in alcohol-preferring P and Sprague–Dawley rats. Pharmacol Biochem Behav 81:122–130
Overstreet DH, Knapp DJ, Angel RA, Navarro M, Breese GR (2006) Reduction in repeated ethanol-withdrawal-induced anxiety-like behavior by site-selective injections of 5-HT(1A) and 5-HT(2C) ligands. Psychopharmacology 187:1–12
Overstreet DH, Knapp DJ, Breese GR (2007) Drug challenges reveal differences in mediation of stress facilitation of voluntary alcohol drinking and withdrawal-induced anxiety-like behavior in alcohol-preferring P rats. Alcohol Clin Exp Res 31:1473–1481
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates, 5th edn. Elsevier Academic Press, Oxford
Pérez de la Mora M, Lara-García D, Jacobsen KX, Vázquez-García M, Crespo-Ramírez M, Flores-Gracia C, Escamilla-Marvan E, Fuxe K (2006) Anxiolytic-like effects of the selective metabotropic glutamate receptor 5 antagonist MPEP after its intra-amygdaloid microinjection in three different non-conditioned rat models of anxiety. Eur J Neurosci 23(10):2749–2759
Pohorecky LA (1990) Interaction of ethanol and stress: research with experimental animals—an update. Alcohol Alcohol 25:263–276
Pohorecky LA (1991) Stress and alcohol interaction: an update of human research. Alcohol Clin Exp Res 15:438–459
Primus RJ, Yevich E, Baltazar C, Gallager DW (1997) Autoradiographic localization of CRF1 and CRF2 binding sites in adult rat brain. Neuropsychopharmacology 17(5):308–316
Rassnick S, Heinrichs SC, Britton KT, Koob GF (1993) Microinjection of a corticotropin releasing factor antagonist into the central nucleus of the amygdala reverses anxiogenic-like effects of ethanol withdrawal. Brain Res 605:25–32
Regier DA, Farmer ME, Rae DS, Locke BZ, Keith SJ, Judd LL, Goodwin FK (1990) Lifetime community prevalence rates from the epidemiological catchment area program survey. J Am Med Assoc 264(19):2511–2518
Rewal M, Jurd R, Gill TM, He DY, Ron D, Janak PH (2009) Alpha-4 containing GABAA receptors in the nucleus accumbens mediate moderate intake of alcohol. J Neurosci 29:543–549
Rodd ZA, Melendez RI, Bell RL, Kuc KA, Zhang Y, Murphy JM, McBride WJ (2004) Intracranial self-administration of ethanol within the ventral tegmental area of male Wistar rats: evidence for involvement of dopamine neurons. J Neurosci 24:1050–1057
Ryabinin AE, Criado JR, Henriksen SJ, Bloom FE, Wilson MC (1997) Differential sensitivity of c-Fos expression in hippocampus and other brain regions to moderate and low doses of alcohol. Mol Psychiatry 2(1):32–43
Samson HH, Chappell A (2003) Dopaminergic involvement in medial prefrontal cortex and core of the nucleus accumbens in the regulation of ethanol self-administration: a dual-site microinjection study in the rat. Physiol Behav 79(4–5):581–590
Silberman Y, Bajo M, Chappell AM, Christian DT, Cruz M, Diaz MR, Kash T, Lack AK, Messing RO, Siggins GR, Winder D, Roberto M, McCool BA, Weiner JL (2009) Neurobiological mechanisms contributing to alcohol-stress-anxiety interactions. Alcohol 43(7):509–519
Sinclair JD, Senter RJ (1968) Development of an alcohol-deprivation effect in rats. Q J Stud Alcohol 29:863–867
Sinha R (2001) How does stress increase risk of drug abuse and relapse? Psychopharmacology 158:343–359
Sinha R (2008) Chronic stress, drug use, and vulnerability to addiction. Ann NY Acad Sci 1141:105–130
Sommer WH, Rimondini R, Hansson AC, Hipskind PA, Gehlert DR, Barr CS, Heilig MA (2008) Upregulation of voluntary alcohol intake, behavioral sensitivity to stress, and amygdala crhr1 expression following a history of dependence. Biol Psychiatry 63(2):139–145
Tohyama M, Takatsuji K (1998) Atlas of neuroactive substances and their receptors in the rat. Oxford University Press, New York
Van Pett K, Viau V, Bittencourt JC, Chan RK, Li HY, Arias C, Prins GS, Perrin M, Vale W, Sawchenko PE (2000) Distribution of mRNAs encoding CRF receptors in brain and pituitary of rat and mouse. J Comp Neurol 428(2):191–212
Vengeliene V, Siegmund S, Singer MV, Sinclair JD, Li TK, Spanagel R (2003) A comparative study on alcohol-preferring rat lines: effects of deprivation and stress phases on voluntary alcohol intake. Alcohol Clin Exp Res 27:1048–1054
Wand G (2005) The anxious amygdala: CREB signaling and predisposition to anxiety and alcoholism. J Clin Invest 115:2762–2773
Wang B, You ZB, Rice KC, Wise RA (2007) Stress-induced relapse to cocaine seeking: roles for the CRF(2) receptor and CRF-binding protein in the ventral tegmental area of the rat. Psychopharmacology (Berl) 193(2):283–294
Zhou FC, Anthony B, Dunn KW, Lindquist WB, Xu ZC, Deng P (2007) Chronic alcohol drinking alters neuronal dendritic spines in the brain reward center nucleus accumbens. Brain Res 1134(1):148–161
Zorrilla EP, Koob GF (2004) The therapeutic potential of CRF1 antagonists for anxiety. Expert Opin Investig Drugs 13:799–828
Acknowledgments
This work was supported by NIAAA [AA11605, AA14949] as well as by funds from the Center for Alcohol Studies. Drs. Knapp and Overstreet contributed equally to this work. We thank T-K Li and Lawrence Lumeng at Indiana University Medical Center for sending the original breeding stock to establish our colony of P rats used in this investigation. We also thank Kui-Ling Huang for technical assistance. The authors confirm that they have no financial relationship with the NIH or Synofi other than funding this research (NIH) or providing the gift of the SSR compound (Synofi). The authors also confirm that these experiments comply with all relevant laws of the United States.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
213_2011_2366_MOESM1_ESM.ppt
Supplemental Figure Legend. Microinjection targeting based on the brain atlas of Paxinos and Watson (2005). See “Methods” in text for coordinate details for each brain region. Red circles represent the ideal center point (anterior-posterior, dorso-ventral, and medial-lateral) based on the coordinates detailed in Table 1. For the schematics at left, dark hatched circles represent approximate outer limits of injections deemed acceptable. Other circles indicate representative locations of injections in regions of experimental groups in which CRF (green circles) or the CRF antagonist (black circles) were active. Numbers in the lower right of each schematic reflect the plate number from the atlas. Photomicrographs representing injection sites for each region appear in the right column. The straight hatched lines in the photomicrographs represent the approximate angle of approach to the sites of interest where darkened dye markings can be seen. a Accumbens, b paraventricular nucleus of the hypothalamus, c amygdala, d ventral tegmental area, e dorsal raphe nucleus (PPT 2.04 mb)
Rights and permissions
About this article
Cite this article
Knapp, D.J., Overstreet, D.H., Huang, M. et al. Effects of a stressor and corticotrophin releasing factor on ethanol deprivation-induced ethanol intake and anxiety-like behavior in alcohol-preferring P rats. Psychopharmacology 218, 179–189 (2011). https://doi.org/10.1007/s00213-011-2366-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-011-2366-5