Abstract
Rationale
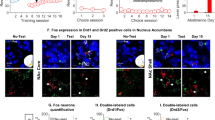
Behavioral and anatomical data suggest that the ventral striatum, consisting of the nucleus accumbens and olfactory tubercle, is functionally heterogeneous. Cocaine and d-amphetamine appear to be more rewarding when administered into the medial olfactory tubercle or medial accumbens shell than into their lateral counterparts, including the accumbens core.
Objectives
We sought to determine whether rats self-administer the popular recreational drug (±)-3,4-methylenedioxymethamphetamine (MDMA) into ventrostriatal subregions and whether the medial olfactory tubercle and medial accumbens shell mediate MDMA’s positive reinforcing effects more effectively than their lateral counterparts.
Results
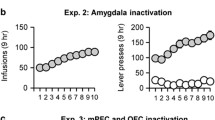
Rats receiving 30 mM MDMA into the medial olfactory tubercle, medial accumbens shell, or accumbens core, but not the lateral tubercle or lateral shell, showed higher self-administration rates than rats receiving vehicle. The medial shell supported more vigorous self-administration of MDMA at higher concentrations than the core or medial olfactory tubercle. In addition, intra-medial shell MDMA self-administration was disrupted by co-administration of the D1 or D2 receptor antagonists SCH 23390 (1–3 mM) or raclopride (3–10 mM).
Conclusions
Our data suggest that the ventral striatum is functionally heterogeneous. The medial accumbens shell appears to be more important than other ventrostriatal subregions in mediating the positive reinforcing effects of MDMA via both D1- and D2-type receptors. Together with previous data, our data also suggest that unidentified actions of MDMA interfere with the positive reinforcing effects of dopamine in the medial olfactory tubercle.






Similar content being viewed by others
References
Bari AA, Pierce RC (2005) D1-like and D2 dopamine receptor antagonists administered into the shell subregion of the rat nucleus accumbens decrease cocaine, but not food, reinforcement. Neuroscience 135:959–968
Bossert JM, Poles GC, Wihbey KA, Koya E, Shaham Y (2007) Differential effects of blockade of dopamine D1-family receptors in nucleus accumbens core or shell on reinstatement of heroin seeking induced by contextual and discrete cues. J Neurosci 27:12655–12663
Carlezon WA Jr, Devine DP, Wise RA (1995) Habit-forming actions of nomifensine in nucleus accumbens. Psychopharmacology 122:194–197
Daniela E, Brennan K, Gittings D, Hely L, Schenk S (2004) Effect of SCH 23390 on (+/−)-3,4-methylenedioxymethamphetamine hyperactivity and self-administration in rats. Pharmacol Biochem Behav 77:745–750
De La Garza R 2nd, Fabrizio KR, Gupta A (2007) Relevance of rodent models of intravenous MDMA self-administration to human MDMA consumption patterns. Psychopharmacology (Berl) 189:425–434
Fenu S, Spina L, Rivas E, Longoni R, Di Chiara G (2006) Morphine-conditioned single-trial place preference: role of nucleus accumbens shell dopamine receptors in acquisition, but not expression. Psychopharmacology (Berl) 187:143–153
Fibiger HC, Phillips AG (1986) Reward, motivation, cognition: psychobiology of mesotelencephalic dopamine systems. In: Mountcastle VB, Bloom FE, Geiger SR (eds) Handbook of physiology: vol. 4. The nervous system. American Physiological Society, Bethesda, pp 647–675
Green AR, Mechan AO, Elliott JM, O’Shea E, Colado MI (2003) The pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). Pharmacol Rev 55:463–508
Ikemoto S (2003) Involvement of the olfactory tubercle in cocaine reward: intracranial self-administration studies. J Neurosci 23:9305–9311
Ikemoto S (2007) Dopamine reward circuitry: two projection systems from the ventral midbrain to the nucleus accumbens–olfactory tubercle complex. Brain Res Rev 56:27–78
Ikemoto S, Sharpe LG (2001) A head-attachable device for injecting nanoliter volumes of drug solutions into brain sites of freely moving rats. J Neurosci Methods 110:135–140
Ikemoto S, Wise RA (2004) Mapping of chemical trigger zones for reward. Neuropharmacology 47:190–201
Ikemoto S, Glazier BS, Murphy JM, McBride WJ (1997) Role of dopamine D1 and D2 receptors in the nucleus accumbens in mediating reward. J Neurosci 17:8580–8587
Ikemoto S, Qin M, Liu ZH (2005) The functional divide for primary reinforcement of D-amphetamine lies between the medial and lateral ventral striatum: is the division of the accumbens core, shell and olfactory tubercle valid. J Neurosci 25:5061–5065
Ito R, Robbins TW, Everitt BJ (2004) Differential control over cocaine-seeking behavior by nucleus accumbens core and shell. Nat Neurosci 7:389–397
Kankaanpaa A, Meririnne E, Lillsunde P, Seppala T (1998) The acute effects of amphetamine derivatives on extracellular serotonin and dopamine levels in rat nucleus accumbens. Pharmacol Biochem Behav 59:1003–1009
Koob GF (1992) Drugs of abuse: anatomy, pharmacology and function of reward pathways. Trends Pharmacol Sci 13:177–184
McBride WJ, Murphy JM, Ikemoto S (1999) Localization of brain reinforcement mechanisms: intracranial self-administration and intracranial place-conditioning studies. Behav Brain Res 101:129–152
Parrott AC (2001) Human psychopharmacology of Ecstasy (MDMA): a review of 15 years of empirical research. Hum Psychopharmacol 16:557–577
Parrott AC (2004) Is ecstasy MDMA? A review of the proportion of ecstasy tablets containing MDMA, their dosage levels, and the changing perceptions of purity. Psychopharmacology (Berl) 173:234–241
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Elsevier, Burlington
Pierce RC, Kumaresan V (2006) The mesolimbic dopamine system: the final common pathway for the reinforcing effect of drugs of abuse. Neurosci Biobehav Rev 30:215–238
Pope HG Jr, Ionescu-Pioggia M, Pope KW (2001) Drug use and life style among college undergraduates: a 30-year longitudinal study. Am J Psychiatry 158:1519–1521
Rodd-Henricks ZA, McKinzie DL, Li TK, Murphy JM, McBride WJ (2002) Cocaine is self-administered into the shell but not the core of the nucleus accumbens of Wistar rats. J Pharmacol Exp Ther 303:1216–1226
Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (2001) Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 39:32–41
Sellings LH, Clarke PB (2003) Segregation of amphetamine reward and locomotor stimulation between nucleus accumbens medial shell and core. J Neurosci 23:6295–6303
Sellings LH, McQuade LE, Clarke PB (2006a) Characterization of dopamine-dependent rewarding and locomotor stimulant effects of intravenously-administered methylphenidate in rats. Neuroscience 141:1457–1468
Sellings LH, McQuade LE, Clarke PB (2006b) Evidence for multiple sites within rat ventral striatum mediating cocaine conditioned place preference and locomotor activation. J Pharmacol Exp Ther 317:1178–1187
Spina L, Fenu S, Longoni R, Rivas E, Di Chiara G (2006) Nicotine-conditioned single-trial place preference: selective role of nucleus accumbens shell dopamine D1 receptors in acquisition. Psychopharmacology (Berl) 184:447–455
White SR, Obradovic T, Imel KM, Wheaton MJ (1996) The effects of methylenedioxymethamphetamine (MDMA, “Ecstasy”) on monoaminergic neurotransmission in the central nervous system. Prog Neurobiol 49:455–479
Wise RA, Bozarth MA (1987) A psychomotor stimulant theory of addiction. Psychol Rev 94:469–492
Yamamoto BK, Spanos LJ (1988) The acute effects of methylenedioxymethamphetamine on dopamine release in the awake-behaving rat. Eur J Pharmacol 148:195–203
Acknowledgment
This work was supported by the Intramural Research Program of the National Institute on Drug Abuse, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, R., Qin, M., Liu, ZH. et al. Intracranial self-administration of MDMA into the ventral striatum of the rat: differential roles of the nucleus accumbens shell, core, and olfactory tubercle. Psychopharmacology 198, 261–270 (2008). https://doi.org/10.1007/s00213-008-1131-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-008-1131-x