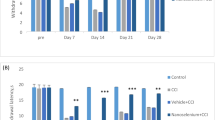
Abstract
The toxicological effects of lead and its compounds have overshadowed its possible health beneficial effects. Currently, the success rate for treating neuropathic pain has been very low. This study investigated the antinociceptive effects of orally administered low dose lead acetate in sciatic nerve ligated Wistar rats. Thirty Wistar rats randomly divided into five groups were used for this study. Chronic constriction injury (CCI) was used to induce neuropathic pain in Wistar rats. Allodynic and hyperalgesic signs were investigated using von Frey filaments and hotplate, respectively. Morris water maze test was used to assess the memory functions of the rats. The study revealed that oral administration of low-dose lead acetate significantly (p < 0.05) increased pain thresholds of ligated rats. CCI enhanced memory function in Wistar rats which was significantly decreased following lead acetate administration. The findings suggest that lead acetate possesses antinociceptive effects in peripherally induced neuropathic pain model in Wistar rats.







Similar content being viewed by others
References
Abdou HM, Hassan MA (2014) Protective role of omega-3 polyunsaturated fatty acid against lead acetate-induced toxicity in liver and kidney of female rats. Biomed Res Int. https://doi.org/10.1155/2014/435857
Abdulmajeed W, Owoyele BV (2015) Anti-nociceptive effects of taurine and caffeine in sciatic nerve ligated wistar rats : involvement of autonomic receptors. J African Assoc Physiol Sci 3:41–48
Bain JR, Mackinnon SE, Hunter DA (1989) Functional evaluation of complete sciatic, peroneal, and posterior tibial nerve lesions in the rat. Plast Reconstr Surg 83:129–138
Bennett GJ, Xie YK (1988) A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33:87–107. https://doi.org/10.1016/0304-3959(88)90209-6
Bondy SC, Hong JS, Tilson HA, Walsh TJ (1985) Effects of triethyl lead on hot-plate responsiveness and biochemical properties of hippocampus. Pharmacol Biochem Behav 22:1007–1011. https://doi.org/10.1016/0091-3057(85)90310-7
Burkey RT, Nation JR, Bratton GR (1994) Chronic lead exposure attenuates ethanol-induced hypoalgesia. Pharmacol Biochem Behav 47:227–231. https://doi.org/10.1016/0091-3057(94)90004-3
Colloca L, Ludman T, Bouhassira D et al (2017) HHS Public Access - Neuropathic Pain. Nat Rev Dis Prim 3:1–45. https://doi.org/10.1038/nrdp.2017.2.Neuropathic
Dableh LJ, Yashpal K, Henry JL (2011) Neuropathic pain as a process: reversal of chronification in an animal model. J Pain Res 4:315–323. https://doi.org/10.2147/JPR.S17882
Dworkin RH, O’Connor AB, Backonja M et al (2007) Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 132:237–251. https://doi.org/10.1016/j.pain.2007.08.033
Fazli-Tabaei S, Fahim M, Zarrindast MR (2003) Effect of acute and chronic lead exposure on apomorphine-induced sniffing in rats. Pharmacol Toxicol 92:88–93. https://doi.org/10.1034/j.1600-0773.2003.920205.x
Finnerup NB, Sindrup SH, Jensen TS (2010) The evidence for pharmacological treatment of neuropathic pain. Pain 150:573–581. https://doi.org/10.1016/j.pain.2010.06.019
Ishola IO, Agbaje EO, Adeyemi OO, Shukla R (2014) Analgesic and anti-inflammatory effects of the methanol root extracts of some selected Nigerian medicinal plants. Pharm Biol 52:1208–1216. https://doi.org/10.3109/13880209.2014.880487
Jiang X, Yan Q, Liu F et al (2018) Chronic trans-astaxanthin treatment exerts antihyperalgesic effect and corrects co-morbid depressive like behaviors in mice with chronic pain. Neurosci Lett 662:36–43. https://doi.org/10.1016/j.neulet.2017.09.064
Kilkenny C, Browne WJ, Cuthill IC et al (2013) Improving bioscience research reporting: the arrive guidelines for reporting animal research. Animals 4:35–44. https://doi.org/10.3390/ani4010035
Lamidi IY, Akefe IO (2017) Mitigate effects of antioxidants in lead toxicity. Clin Pharmacol Toxicol J 1:1–9
Li XY, Ko HG, Chen T et al (2010) Alleviating neuropathic pain hypersensitivity by inhibiting PKMζ in the anterior cingulate cortex. Science (80-) 330:1400–1404. https://doi.org/10.1126/science.1191792
Mahmoud YI, Sayed SS (2016) Effects of L-cysteine on lead acetate induced neurotoxicity in albino mice. Biotech Histochem 91:327–332. https://doi.org/10.3109/10520295.2016.1164897
Migues PV, Hardt O, Wu DC et al (2010) PKM maintains memories by regulating GluR2-dependent AMPA receptor trafficking. Nat Neurosci 13:630–634. https://doi.org/10.1038/nn.2531
Monte-Raso VV, Barbieri CH, Mazzer N et al (2008) Is the sciatic functional index always reliable and reproducible? J Neurosci Methods 170:255–261. https://doi.org/10.1016/j.jneumeth.2008.01.022
Muthuraman A, Diwan V, Jaggi AS et al (2008) Ameliorative effects of Ocimum sanctum in sciatic nerve transection-induced neuropathy in rats. J Ethnopharmacol 120:56–62. https://doi.org/10.1016/j.jep.2008.07.049
Nikfar S, Abdollahi M, Sharifzadeh M, Eftekhar N (1998) Interaction between lead acetate and morphine on antinociception in mice by formalin test. Gen Pharmacol 30:489–493. https://doi.org/10.1016/S0306-3623(97)00283-8
Ossipov MH, Lai J, King T et al (2004) Antinociceptive and nociceptive actions of opioids. J Neurobiol 61:126–148. https://doi.org/10.1002/neu.20091
Oyagbemi AA, Omobowale TO, Akinrinde AS et al (2015) Lack of reversal of oxidative damage in renal tissues of lead acetate-treated rats. Environ Toxicol 30:1235–1243. https://doi.org/10.1002/tox.21994
Rainville P (2002) Distributed representation of nociception underlying the experience of pain. Curr Opin Neurobiol 12:195–204
Sacktor TC (2010) 11 - Progress - How Does Pkmζ Maintain Long-Term Memory.pdf
Salvemini D, Little JW, Doyle T, Neumann WL (2011) Roles of reactive oxygen and nitrogen species in pain. Free Radic Biol Med 51:951–966. https://doi.org/10.1016/j.freeradbiomed.2011.01.026
Sanders T, Liu Y, Buchner V, Tchounwou PB (2010) Neurotoxic effects and biomarkers of leaf exposure: a review. Revis Environ Heal 24:15–45
Sandkühler J, Lee J (2013) How to erase memory traces of pain and fear. Trends Neurosci 36:343–352. https://doi.org/10.1016/j.tins.2013.03.004
Sayed Aly Mohamed Metwally E (2015) Anatomical and histological study of the effect of lead on hepatocytes of albino rats. Int J Biomed Mater Res 3:34. https://doi.org/10.11648/j.ijbmr.20150304.11
Tilo G, Susanne F, Garret AF (2006) Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities. J Clin Invest 116:4–15. https://doi.org/10.1172/JCI27291.The
Tiwari V, Guan Y, Raja SN (2014) Modulating the delicate Glial-Neuronal Interactions in neuropathic pain: promises and potential caveats. Neurosci Biobehav Rev 45:19–27. https://doi.org/10.1016/j.neubiorev.2014.05.002
Umar AH, Suleiman I, Muhammad H (2016) Effect of low dose lead ( Pb ) administration on tail immersion test and formalin-induced pain in Wistar Rats : possible modulatory role of cobalt ( II ) chloride. Niger J Physiol Sci 31:161–164
Vickers C, Paterson A (1986) Two types of chronic lead treatment in C57BL/6 mice: interaction with behavioural determinants of pain. Life Sci 39:47–53. https://doi.org/10.1016/0024-3205(86)90436-4
Weng H-R, Gao M, Maixner DW (2014) Glycogen synthase kinase 3 beta regulates glial glutamate transporter protein expression in the spinal dorsal horn in rats with neuropathic pain. Exp Neurol 252:18–27. https://doi.org/10.1016/j.expneurol.2013.11.018
Woolf CJ, Mannion RJ (1999) Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 353:1959–1964. https://doi.org/10.1016/S0140-6736(99)01307-0
Yan X, Weng HR (2013) Endogenous interleukin-1β in neuropathic rats enhances glutamate release from the primary afferents in the spinal dorsal horn through coupling with presynaptic N-methyl-D-aspartic acid receptors. J Biol Chem 288:30544–30557. https://doi.org/10.1074/jbc.M113.495465
Zhang MT, Wang B, Jia YN et al (2017) Neuroprotective effect of liquiritin against neuropathic pain induced by chronic constriction injury of the sciatic nerve in mice. Biomed Pharmacother 95:186–198. https://doi.org/10.1016/j.biopha.2017.07.167
Zhu YF, Wu Q, Henry JL (2012) Changes in functional properties of A-type but not C-type sensory neurons in vivo in a rat model of peripheral neuropathy. J Pain Res 5:175–192. https://doi.org/10.2147/JPR.S26367
Zurich MG, Eskes C, Honegger P et al (2002) Maturation-dependent neurotoxicity of lead acetate in vitro: Implication of glial reactions. J Neurosci Res 70:108–116. https://doi.org/10.1002/jnr.10367
Acknowledgment
Our special appreciation goes to Mr. Muritala Sharafadeen and Bashir Abubakar of the Department of Physiology, University of Ilorin, for their immense technical support during this study.
Funding
No grants were received for this project. It was self-sponsored.
Author information
Authors and Affiliations
Contributions
BVO conceived and designed the research. AOB, KA, DA, and SA conducted experiments and analyzed data. BVO and AOB contributed new reagents and analytical tools. BVO, AOB, and MA supervised the research and wrote the manuscript. All authors read and approved the manuscript and all data were generated in-house and no paper mill was used.
Corresponding author
Ethics declarations
The study was conducted in accordance with the ARRIVE guidelines (Kilkenny et al. 2013). The protocol was also assessed and approved by the University of Ilorin Ethical Review Committee (UERC/ASN/2019/1744) before the commencement of the research.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(XLSX 12 kb)
Rights and permissions
About this article
Cite this article
Owoyele, B., Bakare, A., Ayinla, M.T. et al. Antinociceptive effects of lead acetate in sciatic nerve chronic constriction injury model of peripheral neuropathy in male Wistar rats. Naunyn-Schmiedeberg's Arch Pharmacol 394, 117–125 (2021). https://doi.org/10.1007/s00210-020-01951-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01951-w