Abstract
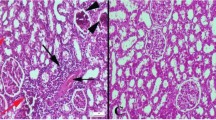
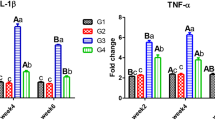
Paraquat (PRQ) is a toxic chemical compound that is very noxious to animals and humans. Gallic acid is a phenolic compound that has antioxidant properties. In this study, we evaluated the ameliorative effect of gallic acid against PRQ-induced renal injury and oxidative stress. In this research, the rats were segregated into six groups. Group 1 is the control group; group 2 received paraquat only; group 3 received gallic acid only; and groups 4, 5, and 6 received paraquat plus gallic acid at doses of 25, 50, and 100 mg/kg bw respectively. Findings of this work displayed that the renal contents of the vitamin C, superoxide dismutase (SOD), and catalase (CAT) significantly reduced and the levels of the serum protein carbonyl, creatinine, serum glutamate pyruvate transaminase (sGPT), urea, serum glutamate oxaloacetate transaminase (sGOT), uric acid, MDA, serum IL-1β, and the kidney IL-1β gene expression were remarkably increased in the group receiving PRQ only compared with that in the control group. On the other hand, treatment with gallic acid after exposure to PRQ led to a significant elevation in renal vitamin C, SOD, and CAT levels plus a remarkable decrease in the serum protein carbonyl, creatinine, sGPT, urea, sGOT, uric acid, MDA, IL-1β, and renal gene expression of IL-1β in comparison with the PRQ-only-treated rats. Histological changes were also ameliorated by gallic acid administration. The data approve that gallic acid diminished the deleterious effects of PRQ exposure. In this regard, our results indicated that the administration of gallic acid could alleviate the noxious effects of PRQ on the antioxidant defense system and renal tissue.


Similar content being viewed by others
References
Akinloye O, Abioye O, Olaojoyetan O, Awosika O, Akinloye D (2013) Dose-dependent effects of paraquat on c-reactive protein, some lipid profile parameters and histology of tissues in male albino rats. IFE J Sci 15:189–195
Awadalla EA (2012) Efficacy of vitamin C against liver and kidney damage induced by paraquat toxicity. Exp Toxicol Pathol 64:431–434
Black AT, Gray JP, Shakarjian MP, Laskin DL, Heck DE, Laskin JD (2008) Increased oxidative stress and antioxidant expression in mouse keratinocytes following exposure to paraquat. Toxicol Appl Pharmacol 231:384–392
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chan B, Lazzaro V, Seale J, Duggin G (1998) The renal excretory mechanisms and the role of organic cations in modulating the renal handling of paraquat. Pharmacol Ther 79:193–203
Danan Jaya BP, Widiastuti EL, Nurcahyani E, Sutyarso S (2017) Taurine and oyster mushroom (Pleurotus Ostreatus) prevents oxidative damage in liver of mice induced by paraquat. Biomed Pharmacol J 10:1993–2000
Fındık H, Tumkaya L, Yılmaz A, Gökhan Aslan M, Okutucu M, Akyildiz K, Mercantepe T (2019) The protective effects of astaxanthin against cisplatin-induced retinal toxicity. Cutan Ocul Toxicol 38:59–65
Flohe L (1984) Superoxide dismutase assays. In: Methods in enzymology, vol 105. Elsevier, pp 93-104
Gil H-W, Seok S-J, Jeong D-S, Yang J-O, Lee E-Y, Hong S-Y (2010) Plasma level of malondialdehyde in the cases of acute paraquat intoxication. Clin Toxicol 48:149–152
Gu S-Y, Yeh T-Y, Lin S-Y, Peng F-C (2016) Unfractionated bone marrow cells attenuate paraquat-induced glomerular injury and acute renal failure by modulating the inflammatory response. Sci Rep 6:23287
Han J-F, Wang S-L, He X-Y, Liu C-Y, Hong J-Y (2006) Effect of genetic variation on human cytochrome p450 reductase-mediated paraquat cytotoxicity. Toxicol Sci 91:42–48
Heidarian E, Soofiniya Y (2011) Hypolipidemic and hypoglycemic effects of aerial part of Cynara scolymus in streptozotocin-induced diabetic rats. J Med Plant Res 5:2717–2723
Heidarian E, Rafieian-Kopaei M, Khoshdel A, Bakhshesh M (2014) Metabolic effects of berberine on liver phosphatidate phosphohydrolase in rats fed on high lipogenic diet: an additional mechanism for the hypolipidemic effects of berberine. Asian Pac J Trop Biomed 4:S429–S435
Hu X, Liang Y, Zhao H, Zhao M (2019) Effects of AT-RvD1 on paraquat-induced acute renal injury in mice. Int Immunopharmacol 67:231–238
Hynes MJ, Coinceanainn MÓ (2001) The kinetics and mechanisms of the reaction of iron (III) with gallic acid, gallic acid methyl ester and catechin. J Inorg Biochem 85:131–142
Jadon A, Bhadauria M, Shukla S (2007) Protective effect of Terminalia belerica Roxb. and gallic acid against carbon tetrachloride induced damage in albino rats. J Ethnopharmacol 109:214–218
Kähkönen MP, Hopia AI, Vuorela HJ, Rauha J-P, Pihlaja K, Kujala TS, Heinonen M (1999) Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem 47:3954–3962
Karimi-Khouzani O, Heidarian E, Amini SA (2017) Anti-inflammatory and ameliorative effects of gallic acid on fluoxetine-induced oxidative stress and liver damage in rats. Pharmacol Rep 69:830–835
Kazłowska K, Hsu T, Hou C-C, Yang W-C, Tsai G-J (2010) Anti-inflammatory properties of phenolic compounds and crude extract from Porphyra dentata. J Ethnopharmacol 128:123–130
Kilic K, Sakat MS, Akdemir FNE, Yildirim S, Saglam YS, Askin S (2019) Protective effect of gallic acid against cisplatin-induced ototoxicity in rats. Braz J Otorhinolaryngol 85:267–274
Kimbrough RD (1974) Toxic effects of the herbicide paraquat. Chest 65:65S–67S
Koekkoek W, van Zanten AR (2016) Antioxidant vitamins and trace elements in critical illness. Nutr Clin Pract 31:457–474
Liu Z, Wang X, Wang Y, Zhao M (2017) NLRP3 inflammasome activation regulated by NF-κB and DAPK contributed to paraquat-induced acute kidney injury. Immunol Res 65:687–698
Medinas DB, Augusto O (2010) Mechanism of the peroxidase activity of superoxide dismutase 1. Free Radic Biol Med 49:682 author reply 683
Mølck AM, Friis C (1998) Transport of paraquat by isolated renal proximal tubular segments from rabbits. Pharmacol Toxicol 83:208–213
Mulay SR et al. (2012) Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1β secretion. J Clin Invest 123
Nabavi SM, Habtemariam S, Nabavi SF, Sureda A, Daglia M, Moghaddam AH, Amani MA (2013) Protective effect of gallic acid isolated from Peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat’s kidney. Mol Cell Biochem 372:233–239
Nouri A, Heidarian E (2019a) Ameliorative effects of N-acetyl cysteine on diclofenac-induced renal injury in male rats based on serum biochemical parameters, oxidative biomarkers, and histopathological study. J Food Biochem 43:e12950
Nouri A, Heidarian E (2019b) Nephroprotective effect of silymarin against diclofenacinduced renal damage and oxidative stress in male rats. J Herbmed Pharmacol
Nouri A, Heidarian E, Amini-Khoei H, Abbaszadeh S, Basati G (2019) Quercetin through mitigation of inflammatory response and oxidative stress exerts protective effects in rat model of diclofenac-induced liver toxicity. J Pharm Pharmacogn Res 7:200–212
Omaye ST, Turnbull JD, Sauberlich HE (1979) Selected methods for the determination of ascorbic acid in animal cells, tissues, and fluids. In: Methods in enzymology, vol 62. Elsevier, pp 3-11
Padma S, Sundaram PS (2020) Determining an accurate method to estimate GFR in renal transplant recipients with stable serum creatinine levels. Iran J Nucl Med 28:21–27
Padma VV, Sowmya P, Felix TA, Baskaran R, Poornima P (2011) Protective effect of gallic acid against lindane induced toxicity in experimental rats. Food Chem Toxicol 49:991–998
Prashad D, Chambers D, Beadle D (1981) Changes in renal function associated with paraquat dichloride toxicity in the domestic fowl. Gen Pharmacol Vasc S 12:291–293
Ramkumar K, Vijayakumar RS, Vanitha P, Suganya N, Manjula C, Rajaguru P, Sivasubramanian S, Gunasekaran P (2014) Protective effect of gallic acid on alloxan-induced oxidative stress and osmotic fragility in rats. Hum Exp Toxicol 33:638–649
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. In: Methods in enzymology, vol 233. Elsevier, pp 357-363
Rifaioglu MM, Sefil F, Gokce H, Nacar A, Dorum BA, Davarci M (2015) Protective effects of caffeic acid phenethyl ester on the dose-dependent acute nephrotoxicity with paraquat in a rat model. Environ Toxicol 30:375–381
Sahu N, Mishra G, Chandra HK, Nirala SK, Bhadauria M (2020) Naringenin mitigates antituberculosis drugs induced hepatic and renal injury in rats. J Tradit Complement Med 10:26–35
Seok S-J, Gil H-W, Jeong D-S, Yang J-O, Lee E-Y, Hong S-Y (2009) Paraquat intoxication in subjects who attempt suicide: why they chose paraquat. Korean J Intern Med 24:247–251
Sepulveda JL (2019) Challenges in routine clinical chemistry testing analysis of small molecules. In: Accurate results in the clinical laboratory. Elsevier, pp 101–140
Shahzad CM, Mohafez OM, Rasool ST, Nair AB (2017) Effect of vitamin C on N, N′-dimethyl-4, 4′-bipyridinium dichloride-induced hepatic and renal toxicity in Swiss albino mice. Trop J Pharm Res 16:2645–2649
Sharifinasab Z, Banaee M, Mohiseni M, Noori A (2016) Vitamin C and chitosan alleviate toxic effects of paraquat on some biochemical parameters in hepatocytes of common carp. Iran J Toxicol 10:31–40
Sharifi-Rigi A, Heidarian E (2019a Protective and anti-inflammatory effects of silymarin on paraquat-induced nephrotoxicity in rats). J Herbmed Pharmacol 8
Sharifi-Rigi A, Heidarian E (2019b) Therapeutic potential of Origanum vulgare leaf hydroethanolic extract against renal oxidative stress and nephrotoxicity induced by paraquat in rats. Avicenna J Phytomed 9:563
Tan D, Wang Y, Bai B, Yang X, Han J (2015) Betanin attenuates oxidative stress and inflammatory reaction in kidney of paraquat-treated rat. Food Chem Toxicol 78:141–146
Vale J, Meredith T, Buckley B (1987) Paraquat poisoning: clinical features and immediate general management. Hum Toxicol 6:41–47
Valipour P, Heidarian E, Khoshdel A, Gholami AM (2016) Protective effects of hydroalcoholic extract of Ferulago angulata against gentamicin-induced nephrotoxicity in rats
Vesey DA, Cheung CW, Cuttle L, Endre ZA, Gobé G, Johnson DW (2002) Interleukin-1β induces human proximal tubule cell injury, α-smooth muscle actin expression and fibronectin production. Kidney Int 62:31–40
Wang Z, Ge Y, Bao H, Dworkin L, Peng A, Gong R (2013) Redox-sensitive glycogen synthase kinase 3β-directed control of mitochondrial permeability transition: rheostatic regulation of acute kidney injury. Free Radic Biol Med 65:849–858
Wei T, Tian W, Liu F, Xie G (2014) Protective effects of exogenous β-hydroxybutyrate on paraquat toxicity in rat kidney. Biochem Biophys Res Commun 447:666–671
Xu J-j, J-t Z, Tang L, Lin Q-m (2017) Intravenous injection of Xuebijing attenuates acute kidney injury in rats with paraquat intoxication. World J Emerg Med 8:61–64
Zhang J, Lv G, Zhao Y (2011) The significance of serum xanthine oxidase and oxidation markers in acute paraquat poisoning in humans. Clin Biochem 44:221–225
Author information
Authors and Affiliations
Contributions
Ali Nouri and Fatemeh Heibati performed the experiments and wrote the manuscript; Esfandiar Heidarian analyzed the data and edited the manuscript; Ali Nouri and Esfandiar Heidarian designed the study and wrote the manuscript. The experiments were performed in-house, and no paper mill was used.
Corresponding author
Ethics declarations
The whole protocol of the research was approved by Shahrekord University of Medical Sciences Ethics Committee, Shahrekord, Iran (ethics number, IR. SKUMS. REC. 1397. 175).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nouri, A., Heibati, F. & Heidarian, E. Gallic acid exerts anti-inflammatory, anti-oxidative stress, and nephroprotective effects against paraquat-induced renal injury in male rats. Naunyn-Schmiedeberg's Arch Pharmacol 394, 1–9 (2021). https://doi.org/10.1007/s00210-020-01931-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01931-0