Abstract
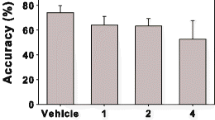
We have recently demonstrated that levodopa acts centrally to induce antinociceptive action against colonic distension through dopamine D2 receptors in rats. Since serotonin (5-HT) and cannabinoid are involved in the regulation of visceral sensation, we hypothesized that they may contribute to levodopa-induced visceral antinociception. We evaluated visceral sensation by colonic distension-induced abdominal withdrawal reflex (AWR) in conscious rats. Subcutaneously administered levodopa increased the threshold of colonic distension-induced AWR; moreover, an intracisternal injection of methiothepin, an unspecific 5-HT receptor antagonist, blocked the levodopa-induced visceral antinociception. Subsequently, we examined the roles of three 5-HT receptor subtypes: 5-HT1A, 5-HT1B, and 5-HT2A, in levodopa-induced visceral antinociception. Ketanserin is a 5-HT2A receptor antagonist that was intracisternally injected and blocked the levodopa-induced antinociception, but neither WAY100635 (5-HT1A receptor antagonist) nor isomoltane (5-HT1B receptor antagonist) did so. Antagonists AM251 (cannabinoid 1 receptor antagonist) or AM630 (cannabinoid 2 receptor antagonist) did not change the levodopa-induced visceral antinociception, suggesting that cannabinoid signaling may not be implicated in levodopa-induced visceral antinociception. We also examined the relation between dopamine D2 and 5-HT2A receptor signaling in the control of visceral sensation. Ketanserin, but not WAY100635, potently blocked the visceral antinociception by quinpirole, which is a dopamine D2 agonist. These results suggest that 5-HT2A receptors in the central nervous system may play specific roles in levodopa-dopamine D2 receptor-induced antinociceptive action against colonic distension.



Similar content being viewed by others
References
Aira Z, Buesa I, Salguerino M, Bilbao J, Aguilera L, Zimmermann M, Azukue JJ (2010) Subtype-specific changes in 5-HT receptor-mediated modulation of C fibre-evoked spinal field potentials are triggered by peripheral nerve injury. Neuroscience 168:831–841. https://doi.org/10.1016/j.neuroscience.2010.04.032
Albizu L, Holloway T, González-Maeso J, Sealfon SC (2011) Functional crosstalk and heteromerization of serotonin 5-HT2A and dopamine D2receptors. Neuropharmacology 61:770–777. https://doi.org/10.1016/j.neuropharm.2011.05.023
Al-Chaer ED, Kawasaki M, Pasricha PJ (2000) A new model of chronic visceral hypersensitivity in adult rats induced by colon irritation during postnatal development. Gastroenterology 119:1276–1285. https://doi.org/10.1053/gast.2000.19576
Ashton JC, Milligan ED (2008) Cannabinoids for the treatment of neuropathic pain: clinical evidence. Curr Opin Investig Drugs 9:65–75
Avila-Rojas SH, Velázquez-Lagunas I, Salinas-Abarca AB, Barragán-Iglesias P, Pineda-Farias JB, Granados-Soto V (2015) Role of spinal 5-HT5A, and 5-HT1A/1B/1D, receptors in neuropathic pain induced by spinal nerve ligation in rats. Brain Res 1622:377–385. https://doi.org/10.1016/j.brainres.2015.06.043
Chia JSM, Omar Farouk AA, Mohamad AS, Sulaiman MR, Perimal EK (2016) Zerumbone alleviates chronic constriction injury-induced allodynia and hyperalgesia through serotonin 5-HT receptors. Biomed Pharmacother 83:1303–1310. https://doi.org/10.1016/j.biopha.2016.08.052
Cirillo C, Vanden Berghe P, Tack J (2011) Role of serotonin in gastrointestinal physiology and pathology. Minerva Endocrinol 36:311–324
Cobacho N, De la Calle JL, González-Escalada JR, Paíno CL (2010) Levodopa analgesia in experimental neuropathic pain. Brain Res Bull 83:304–309. https://doi.org/10.1016/j.brainresbull.2010.08.012
Colpaert FC, Wu WP, Hao JX, Royer I, Sautel F, Wiesenfeld-Hallin Z, Xu XJ (2004) High-efficacy 5-HT1A receptor activation causes a curative-like action on allodynia in rats with spinal cord injury. Eur J Pharmacol 497:29–33. https://doi.org/10.1016/j.ejphar.2004.06.026
Covey DP, Mateo Y, Sulzer D, Cheer JF, Lovinger DM (2017) Endocannabinoid modulation of dopamine neurotransmission. Neuropharmacology 124:52–61. https://doi.org/10.1016/j.neuropharm.2017.04.033
da Silva S, Falconi-Sobrinho LL, Dos Anjos-Garcia T, Coimbra NC (2019) 5-Hydroxytryptamine 2A receptors of the dorsal raphe nucleus modulate paniclike behaviours and mediate fear-induced antinociception elicited by neuronal activation in the central nucleus of the inferior colliculus. Behav Brain Res 357–358:71–81. https://doi.org/10.1016/j.bbr.2017.07.016
de Oliveira R, de Oliveira RC, Falconi-Sobrinho LL, da Silva Soares R Jr, Coimbra NC (2017) 5-Hydroxytryptamine2A/2C receptors of nucleus raphe magnus and gigantocellularis/paragigantocellularis pars α reticular nuclei modulate the unconditioned fear-induced antinociception evoked by electrical stimulation of deep layers of the superior colliculus and dorsal periaqueductal grey matter. Behav Brain Res 316:294–304. https://doi.org/10.1016/j.bbr.2016.09.016
Dickey RP, Minton JP (1972) Levodopa relief of bone pain from breast cancer. N Engl J Med 286:843. https://doi.org/10.1056/NEJM197204132861518
Doherty DM, Pickel VM (2000) Ultrastructural localization of the serotonin 2A receptor in dopaminergic neurons in the ventral tegmental area. Brain Res 864:176–185. https://doi.org/10.1016/S0006-8993(00)02062-X
Dupuis A, Wattiez AS, Pinguet J et al (2017) Increasing spinal 5-HT2A receptor responsiveness mediates anti-allodynic effect and potentiates fluoxetine efficacy in neuropathic rats. Evidence for GABA release. Pharmacol Res 118:93–103. https://doi.org/10.1016/j.phrs.2016.09.021
Ertas M, Sagduyu A, Arac N, Uludag B, Ertekin C (1998) Use of levodopa to relieve pain from painful symmetrical diabetic polyneuropathy. Pain 75:257–259. https://doi.org/10.1016/S0304-3959(98)00003-7
González-Hernández A, Martínez-Lorenzana G, Rodríguez-Jiménez J, Rojas-Piloni G, Condés-Lara M (2015) Intracisternal injection of palmitoylethanolamide inhibits the peripheral nociceptive evoked responses of dorsal horn wide dynamic range neurons. J Neural Transm (Vienna) 122:369–374. https://doi.org/10.1007/s00702-014-1255-6
Hosking RD, Zajicek JP (2008) Therapeutic potential of cannabis in pain medicine. Br J Anaesth 101:59–68. https://doi.org/10.1093/bja/aen119
Hoyer D, Clarke DE, Fozard JR et al (1994) International Union of Pharmacology classification of receptors for 5-hydroxytryptamine (serotonin). Pharmacol Rev 46:157–203
Kayser V, Aubel B, Hamon M, Bourgoin S (2002) The antimigraine 5-HT 1B/1D receptor agonists, sumatriptan, zolmitriptan and dihydroergotamine, attenuate pain-related behaviour in a rat model of trigeminal neuropathic pain. Br J Pharmacol 137:1287–1297. https://doi.org/10.1038/sj.bjp.0704979
Kernbaum S, Hauchecorne J (1981) Administration of levodopa for relief of herpes zoster pain. JAMA 246:132–134. https://doi.org/10.1001/jama.1981.03320020024017
Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC (2006) Functional bowel disorders. Gastroenterology 130:1480–1491. https://doi.org/10.1053/j.gastro.2005.11.061
Malik Z, Baik D, Schey R (2015) The role of cannabinoids in regulation of nausea and vomiting, and visceral pain. Curr Gastroenterol Rep 17:429. https://doi.org/10.1007/s11894-015-0429-1
Markoutsaki T, Karantanos T, Gazouli M, Anagnou NP, Karamanolis DG (2011) 5-HT 2A receptor gene polymerisms and irritable bowel syndrome. J Clin Gastroenterol 45:514–517. https://doi.org/10.1097/MCG.0b013e318205e13b
Mayer EA, Gebhart GF (1994) Basic and clinical aspects of visceral hyperalgesia. Gastroenterology 107:271–293. https://doi.org/10.1016/0016-5085(94)90086-8
Nakai K, Nakae A, Oba S, Mashimo T, Ueda K (2010) 5-HT2C receptor agonists attenuate pain-related behaviour in a rat model of trigeminal neuropathic pain. Eur J Pain 14:999–1006. https://doi.org/10.1016/j.ejpain.2010.04.008
Nie L, Di T, Li Y, Cheng P, Li M, Gao J (2018) Blockade of serotonin 5-HT2A receptors potentiates dopamine D2 activation-induced disruption of pup retrieval on an elevated plus maze, but has no effect on D2 blockade-induced one. Pharmacol Biochem Behav 171:74–84. https://doi.org/10.1016/j.pbb.2018.06.004
Nocjar C, Roth BL, Pehek EA (2002) Localization of 5-HT(2A) receptors on dopamine cells in subnuclei of the midbrain A10 cell group. Neuroscience 111:163–176. https://doi.org/10.1016/S0306-4522(01)00593-0
Obata H, Saito S, Sakurazaqa, Sasaki M, Usui T, Goto F (2004) Antiallodynic effects of intrathecally administered 5-HT(2C) receptor agonists in rats with nerve injury. Pain 108:163–169. https://doi.org/10.1016/j.pain.2003.12.019
Ochi T, Goto T (2000) The antinociceptive effect induced by FR140423 is mediated through spinal 5-HT2A and 5-HT3 receptors. Eur J Pharmacol 409:167–172. https://doi.org/10.1016/s0014-2999(00)00832-3
Okumura T, Nozu T, Kumei S, Takakusaki K, Miyagishi S, Ohhira M (2015a) Antinociceptive action against colonic distension by brain orexin in conscious rats. Brain Res 1598:12–17. https://doi.org/10.1016/j.brainres.2014.12.021
Okumura T, Nozu T, Kumei S, Takakusaki K, Miyagishi S, Ohhira M (2015b) Involvement of dopaminergic system in the central orexin-induced antinociceptive action against colonic distension in conscious rats. Neurosci Lett 605:34–38. https://doi.org/10.1016/j.neulet.2015.08.013
Okumura T, Nozu T, Kumei S, Takakusaki K, Miyagishi S, Ohhira M (2016) Levodopa acts centrally to induce an antinociceptive action against colonic distension through activation of D2 dopamine receptors and the orexinergic system in the brain in conscious rats. J Pharmacol Sci 130:123–127. https://doi.org/10.1016/j.jphs.2016.01.007
Okumura T, Nozu T, Kumei S, Ohhira M (2018a) Role of the cannabinoid signaling in the brain orexin- and ghrelin-induced visceral antinociception in conscious rats. J Pharmacol Sci 137:230–232. https://doi.org/10.1016/j.jphs.2018.06.001
Okumura T, Nozu T, Kumei S, Takakusaki K, Ohhira M (2018b) Ghrelin acts centrally to induce an antinociceptive action during colonic distension through the orexinergic, dopaminergic and opioid systems in conscious rats. Brain Res 1686:48–54. https://doi.org/10.1016/j.brainres.2018.02.024
Patel R, Dickenson AH (2018) Modality selective roles of pro-nociceptive spinal 5-HT2A and 5-HT3 receptors in normal and neuropathic states. Neuropharmacology 143:29–37. https://doi.org/10.1016/j.neuropharm.2018.09.028
Pazos A, Cortes R, Palacios JM (1985) Quantitative autoradiographic mapping of serotonin receptors in the rat brain. II. Serotonin-2 receptors. Brain Res 346:231–249. https://doi.org/10.1016/0006-8993(85)90857-1
Pichon X, Wattiez AS, Becamel C, Ehrlich I, Bockaert J, Eschalier A, Marin P, Courteix C (2010) Disrupting 5-HT(2A) receptor/PDZ protein interactions reduces hyperalgesia and enhances SSRI efficacy in neuropathic pain. Mol Ther 18:1462–1470. https://doi.org/10.1038/mt.2010.101
Posserud I, Syrous A, Lindstrom L, Tack J, Abrahamsson H, Simren M (2007) Altered rectal perception in irritable bowel syndrome is associated with symptom severity. Gastroenterology 133:1113–1123. https://doi.org/10.1053/j.gastro.2007.07.024
Shimizu T, Iwata S, Morioka H, Masuyama T, Fukuda T, Nomoto M (2004) Antinociceptive mechanism of L-DOPA. Pain 110:246–249. https://doi.org/10.1016/j.pain.2004.03.038
Song Z, Meyerson BA, Linderoth B (2011) Spinal 5-HT receptors that contribute to the pain-relieving effects of spinal cord stimulation in a rat model of neuropathy. Pain 152:1666–1673. https://doi.org/10.1016/j.pain.2011.03.012
Whitfield AC, Moore BT, Daniels RN (2014) Classics in chemical neuroscience: levodopa. ACS Chem Neurosci 5:1192–1197. https://doi.org/10.1021/cn5001759
Funding
This work was supported in part by grants-in-aid from the Ministry of Education, Science, Sports and Culture of Japan (19K08410 (TO) and 18K07896 (TN)).
Author information
Authors and Affiliations
Contributions
TO and TN designed the experiments. TO, TN, MI, SI collected the data. TO, SK, MO analyzed the data and TO and TN wrote the paper.
Corresponding author
Ethics declarations
Approval was obtained from the Research and Development and Animal Care committees at Asahikawa Medical University (No. 13030) for all of the experiments conducted in this study.
Conflict of interest
The authors declare that they have no conflict interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Okumura, T., Nozu, T., Ishioh, M. et al. 5-HT2A receptors but not cannabinoid receptors in the central nervous system mediate levodopa-induced visceral antinociception in conscious rats. Naunyn-Schmiedeberg's Arch Pharmacol 393, 1419–1425 (2020). https://doi.org/10.1007/s00210-020-01842-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01842-0