Abstract
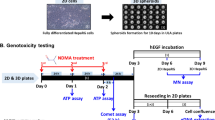
The in vitro micronucleus (MN) assay is a component of most test batteries used in assessing potential genotoxicity. Our previous study adapted metabolically competent HepaRG cells to the high-throughput (HT) flow-cytometry-based MN assay for genotoxicity assessment (Guo et al. in J Toxicol Environ Health A 83:702–717, 2020b, https://doi.org/10.1080/15287394.2020.1822972). We also demonstrated that, compared to HepaRG cells grown as two-dimensional (2D) cultures, 3D HepaRG spheroids have increased metabolic capacity and improved sensitivity in detecting DNA damage induced by genotoxicants using the comet assay (Seo et al. in ALTEX 39:583–604, 2022, https://doi.org/10.14573/altex.22011212022). In the present study, we have compared the performance of the HT flow-cytometry-based MN assay in HepaRG spheroids and 2D HepaRG cells by testing 34 compounds, including 19 genotoxicants or carcinogens and 15 compounds that show different genotoxic responses in vitro and in vivo. 2D HepaRG cells and spheroids were exposed to the test compounds for 24 h, followed by an additional 3- or 6-day incubation with human epidermal growth factor to stimulate cell division. The results demonstrated that HepaRG spheroids showed generally higher sensitivity in detecting several indirect-acting genotoxicants (require metabolic activation) compared to 2D cultures, with 7,12-dimethylbenzanthracene and N-nitrosodimethylamine inducing higher % MN formation along with having significantly lower benchmark dose values for MN induction in 3D spheroids. These data suggest that 3D HepaRG spheroids can be adapted to the HT flow-cytometry-based MN assay for genotoxicity testing. Our findings also indicate that integration of the MN and comet assays improved the sensitivity for detecting genotoxicants that require metabolic activation. These results suggest that HepaRG spheroids may contribute to New Approach Methodologies for genotoxicity assessment.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
References
Barranger A, Le Hegarat L (2022) Towards better prediction of xenobiotic genotoxicity: CometChip technology coupled with a 3D model of HepaRG human liver cells. Arch Toxicol 96(7):2087–2095. https://doi.org/10.1007/s00204-022-03292-4
Beal MA, Audebert M, Barton-Maclaren T et al (2022) Quantitative in vitro to in vivo extrapolation of genotoxicity data provides protective estimates of in vivo dose. Environ Mol Mutagen. https://doi.org/10.1002/em.22521
Chen R, Lin YT, Fornace AJ Jr, Li HH (2022) A high-throughput and highly automated genotoxicity screening assay. Altex 39(1):71–81. https://doi.org/10.14573/altex.2102121
Chevereau M, Glatt H, Zalko D, Cravedi JP, Audebert M (2017) Role of human sulfotransferase 1A1 and N-acetyltransferase 2 in the metabolic activation of 16 heterocyclic amines and related heterocyclics to genotoxicants in recombinant V79 cells. Arch Toxicol 91(9):3175–3184. https://doi.org/10.1007/s00204-017-1935-8
Cimino MC (2006) Comparative overview of current international strategies and guidelines for genetic toxicology testing for regulatory purposes. Environ Mol Mutagen 47(5):362–390. https://doi.org/10.1002/em.20216
Cirit M, Stokes CL (2018) Maximizing the impact of microphysiological systems with in vitro-in vivo translation. Lab Chip 18(13):1831–1837. https://doi.org/10.1039/c8lc00039e
Conway GE, Shah UK, Llewellyn S et al (2020) Adaptation of the in vitro micronucleus assay for genotoxicity testing using 3D liver models supporting longer-term exposure durations. Mutagenesis 35(4):319–330. https://doi.org/10.1093/mutage/geaa018
Dasari S, Ganjayi MS, Meriga B (2018) Glutathione S-transferase is a good biomarker in acrylamide induced neurotoxicity and genotoxicity. Interdiscipl Toxicol 11(2):115–121. https://doi.org/10.2478/intox-2018-0007
Delos M (2021) Cell culture models as an in vitro alternative to study the absorption and biotransformation of drugs and mycotoxins in humans and animals. Ghent University, Ghent, Belgium. https://libstore.ugent.be/fulltxt/RUG01/003/010/626/RUG01-003010626_2021_0001_AC.pdf. Accessed 02/22/2023
EPA (2022) TSCA Chemical Substance Inventory. https://www.epa.gov/tsca-inventory. Accessed 11/28/2022
FDA (2000) Redbook 2000: IV.C.1. Short-term tests for genetic toxicity. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/IngredientsAdditivesGRASPackaging/ucm078321htm. Accessed 12/27/2022
Fenech M (2000) The in vitro micronucleus technique. Mutat Res 455(1–2):81–95. https://doi.org/10.1016/S0027-5107(00)00065-8
Fenech M (2007) Cytokinesis-block micronucleus cytome assay. Nat Protoc 2(5):1084–1104. https://doi.org/10.1038/nprot.2007.77
Fenech M (2008) The micronucleus assay determination of chromosomal level DNA damage. Methods Mol Biol 410:185–216. https://doi.org/10.1007/978-1-59745-548-0_12
Guo X, Seo JE, Li X, Mei N (2020a) Genetic toxicity assessment using liver cell models: past, present, and future. J Toxicol Environ Health Part B 23(1):27–50. https://doi.org/10.1080/10937404.2019.1692744
Guo X, Seo JE, Petibone D et al (2020b) Performance of HepaRG and HepG2 cells in the high-throughput micronucleus assay for in vitro genotoxicity assessment. J Toxicol Environ Health A 83(21–22):702–717. https://doi.org/10.1080/15287394.2020.1822972
ICH (2013) ICH guidance S2(R1) on genotoxicity testing and data interpretation for pharmaceuticals intened for human use. https://www.ema.europa.eu/en/ich-s2-r1-genotoxicity-testing-data-interpretation-pharmaceuticals-intended-human-use. Accessed 11/28/2022
Ivanov DP, Parker TL, Walker DA et al (2014) Multiplexing spheroid volume, resazurin and acid phosphatase viability assays for high-throughput screening of tumour spheroids and stem cell neurospheres. PLoS ONE 9(8):e103817. https://doi.org/10.1371/journal.pone.0103817
Josse R, Rogue A, Lorge E, Guillouzo A (2012) An adaptation of the human HepaRG cells to the in vitro micronucleus assay. Mutagenesis 27(3):295–304. https://doi.org/10.1093/mutage/ger076
Kavlock RJ, Bahadori T, Barton-Maclaren TS, Gwinn MR, Rasenberg M, Thomas RS (2018) Accelerating the pace of chemical risk assessment. Chem Res Toxicol 31(5):287–290. https://doi.org/10.1021/acs.chemrestox.7b00339
Kirkland D, Aardema M, Henderson L, Muller L (2005) Evaluation of the ability of a battery of three in vitro genotoxicity tests to discriminate rodent carcinogens and non-carcinogens I. Sensitivity, specificity and relative predictivity. Mutat Res 584(1–2):1–256. https://doi.org/10.1016/j.mrgentox.2005.02.004
Le Hegarat L, Mourot A, Huet S et al (2014) Performance of comet and micronucleus assays in metabolic competent HepaRG cells to predict in vivo genotoxicity. Toxicol Sci 138(2):300–309. https://doi.org/10.1093/toxsci/kfu004
Luca AC, Mersch S, Deenen R et al (2013) Impact of the 3D microenvironment on phenotype, gene expression, and EGFR inhibition of colorectal cancer cell lines. PLoS ONE 8(3):e59689. https://doi.org/10.1371/journal.pone.0059689
OECD (2015) Guidance document on revisions to OECD genetic toxicology test guidelines. OECD Workgroup of National Coordinators for Test 42 Guidelines (WNT) https://www.oecd.org/chemicalsafety/testing/Genetic%20Toxicology%20Guidance%20Document%20Aug%2031%202015.pdf. Accessed 11/28/2022
OECD (2016) In vitro mammalian cell micronucleus test, OECD Guidelines for the Testing of Chemicals, No. 487. Organisation for Economic Co-operation and Development (Paris, France) https://doi.org/10.1787/9789264264861-en. Assessed 11/28/2022
Pacifici GM, Warholm M, Guthenberg C, Mannervik B, Rane A (1987) Detoxification of styrene oxide by human-liver glutathione transferase. Hum Toxicol 6(6):483–489. https://doi.org/10.1177/096032718700600606
Pfuhler S, Fellows M, van Benthem J et al (2011) In vitro genotoxicity test approaches with better predictivity: summary of an IWGT workshop. Mutat Res 723(2):101–107. https://doi.org/10.1016/j.mrgentox.2011.03.013
Pfuhler S, van Benthem J, Curren R et al (2020) Use of in vitro 3D tissue models in genotoxicity testing: Strategic fit, validation status and way forward. Report of the working group from the 7(th) International Workshop on Genotoxicity Testing (IWGT). Mutat Res 850–851:503135. https://doi.org/10.1016/j.mrgentox.2020.503135
Pfuhler S, Downs TR, Hewitt NJ et al (2021) Validation of the 3D reconstructed human skin micronucleus (RSMN) assay: an animal-free alternative for following-up positive results from standard in vitro genotoxicity assays. Mutagenesis 36(1):1–17. https://doi.org/10.1093/mutage/geaa035
Pridgeon CS, Schlott C, Wong MW et al (2018) Innovative organotypic in vitro models for safety assessment: aligning with regulatory requirements and understanding models of the heart, skin, and liver as paradigms. Arch Toxicol 92(2):557–569. https://doi.org/10.1007/s00204-018-2152-9
Rose S, Ezan F, Cuvellier M et al (2021) Generation of proliferating human adult hepatocytes using optimized 3D culture conditions. Sci Rep 11(1):515. https://doi.org/10.1038/s41598-020-80019-4
Rose S, Cuvellier M, Ezan F et al (2022) DMSO-free highly differentiated HepaRG spheroids for chronic toxicity, liver functions and genotoxicity studies. Arch Toxicol 96(1):243–258. https://doi.org/10.1007/s00204-021-03178-x
Seo JE, Wu Q, Bryant M et al (2020) Performance of high-throughput CometChip assay using primary human hepatocytes: a comparison of DNA damage responses with in vitro human hepatoma cell lines. Arch Toxicol 94(6):2207–2224. https://doi.org/10.1007/s00204-020-02736-z
Seo JE, He X, Muskhelishvili L et al (2022) Evaluation of an in vitro three-dimensional HepaRG spheroid model for genotoxicity testing using the high-throughput CometChip platform. Altex 39(4):583–604. https://doi.org/10.14573/altex.2201121
Shah UK, Mallia JO, Singh N, Chapman KE, Doak SH, Jenkins GJS (2018) A three-dimensional in vitro HepG2 cells liver spheroid model for genotoxicity studies. Mutat Res 825:51–58. https://doi.org/10.1016/j.mrgentox.2017.12.005
Tascher G, Burban A, Camus S et al (2019) In-depth proteome analysis highlights HepaRG cells as a versatile cell system surrogate for primary human hepatocytes. Cells 8(2):192. https://doi.org/10.3390/cells8020192
Turesky RJ, Le Marchand L (2011) Metabolism and biomarkers of heterocyclic aromatic amines in molecular epidemiology studies: lessons learned from aromatic amines. Chem Res Toxicol 24(8):1169–1214. https://doi.org/10.1021/tx200135s
Vernon AR, Pemberton RM, Morse HR (2022) A novel in vitro 3D model of the human bone marrow to bridge the gap between in vitro and in vivo genotoxicity testing. Mutagenesis 37(2):112–129. https://doi.org/10.1093/mutage/geac009
VICH (2014) Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing, VICH GL23(R). International Cooperation on Harmonization of Technical Requirements for Registration of Veterinary Medicinal Products (VICH) https://vichsec.org/en/guidelines/pharmaceuticals/pharma-safety/toxicology.html. Accessed 11/28/2022
Wills JW, Johnson GE, Doak SH, Soeteman-Hernandez LG, Slob W, White PA (2016) Empirical analysis of BMD metrics in genetic toxicology part I: in vitro analyses to provide robust potency rankings and support MOA determinations. Mutagenesis 31(3):255–263. https://doi.org/10.1093/mutage/gev085
Acknowledgements
This study was supported by the U.S. Food and Drug Administration (FDA), National Center for Toxicological Research. We greatly appreciate Drs. Robert Heflich, Mugimane Manjanatha, and Lei Guo for their critical review of this article.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
This manuscript reflects the views of the authors and does not necessarily reflect those of the U.S. Food and Drug Administration. Any mention of commercial products is for clarification only and is not intended as approval, endorsement, or recommendation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Seo, JE., Li, X., Le, Y. et al. High-throughput micronucleus assay using three-dimensional HepaRG spheroids for in vitro genotoxicity testing. Arch Toxicol 97, 1163–1175 (2023). https://doi.org/10.1007/s00204-023-03461-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03461-z