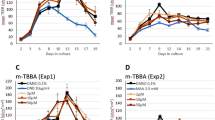
Abstract
A number of para-substituted benzoic acids (p-BA) and chemicals metabolized to p-BA have been found to confer adverse effects in male rats on sperm viability, motility, and morphology. These effects are putatively associated with the metabolism of p-BA to toxic intermediates. We had shown that p-BA lead to accumulation of high levels of p-alkyl-benzoyl-CoA conjugates in plated primary rat hepatocytes. Here we further investigated the relevance of this metabolic pathway for the reprotoxic effects in rats and rabbits. We extended the structure–activity relationship to a set of 19 chemicals (nine reprotoxic and ten non-reprotoxic) and confirmed a very strong correlation between p-alkyl-benzoyl-CoA accumulation in rat hepatocytes and the toxic outcome. Species specificity was probed by comparing rat, rabbit and human hepatocytes, and p-benzoyl-CoA accumulation was found to be specific to the rat hepatocytes, not occurring in human hepatocytes. There was also very limited accumulation in hepatocytes from rabbits that are a non-responder species in in vivo studies. Tissues of rats treated with 3-(4-isopropylphenyl)-2-methylpropanal were analysed and p-isopropyl-benzoyl-CoA conjugates were detected in the liver and in the testes in animals at toxic doses indicating that the metabolism observed in vitro is relevant to the in vivo situation and the critical metabolite does also occur in the reproductive tissue. These multiple lines of evidence further support benzoyl-CoA accumulation as a key initiating event for a specific group of male reproductive toxicants, and indicate a species-specific effect in the rat.




Similar content being viewed by others
References
Badenhorst CP, Erasmus E, van der Sluis R, Nortje C, van Dijk AA (2014) A new perspective on the importance of glycine conjugation in the metabolism of aromatic acids. Drug Metab Rev 46(3):343–361. https://doi.org/10.3109/03602532.2014.908903
Begriche K, Massart J, Robin MA, Borgne-Sanchez A, Fromenty B (2011) Drug-induced toxicity on mitochondria and lipid metabolism: mechanistic diversity and deleterious consequences for the liver. J Hepatol 54(4):773–794. https://doi.org/10.1016/j.jhep.2010.11.006
Bradshaw PR, Athersuch TJ, Stachulski AV, Wilson ID (2020) Acyl glucuronide reactivity in perspective. Drug Discov Today. https://doi.org/10.1016/j.drudis.2020.07.009
Browne GS, Nelson C, Nguyen T, Ellis BA, Day RO, Williams KM (1999) Stereoselective and substrate-dependent inhibition of hepatic mitochondrial β-oxidation and oxidative phosphorylation by the non-steroidal anti-inflammatory drugs ibuprofen, flurbiprofen, and ketorolac. Biochem Pharmacol 57(7):837–844. https://doi.org/10.1016/S0006-2952(98)00342-6
Cagen SZ, Patterson DR, Wimberly HC, Lu CC, Gardiner TH (1989) Toxicity induced by subchronic dermal exposure to paratertiary butyl benzoic acid (ptBBA) in Fischer 344 rats. J Am Coll Toxicol 8(5):1027–1038
Darnell M, Weidolf L (2013) Metabolism of xenobiotic carboxylic acids: focus on coenzyme A conjugation, reactivity, and interference with lipid metabolism. Chem Res Toxicol 26(8):1139–1155. https://doi.org/10.1021/tx400183y
Darnell M, Breitholtz K, Isin EM, Jurva U, Weidolf L (2015) Significantly different covalent binding of oxidative metabolites, acyl glucuronides, and S-acyl CoA conjugates formed from xenobiotic carboxylic acids in human liver microsomes. Chem Res Toxicol 28(5):886–896. https://doi.org/10.1021/tx500514z
ECHA (2016a) European Chemicals Agency: REACH registration dossier, 3-(3-isopropylphenyl)-3-methylpropanal. https://echa.europa.eu/registration-dossier/-/registered-dossier/9059. Accessed 10 Nov 2016
ECHA (2016b) European Chemicals Agency: REACH registration dossier, 3-(3-tert-butylphenyl)-2-methylpropanal. https://echa.europa.eu/registration-dossier/-/registered-dossier/10357. Accessed 21 Nov 2016
ECHA (2016c) European Chemicals Agency: REACH registration dossier, 3-(4-isopropylphenyl)-2-methylpropanal. https://echa.europa.eu/registration-dossier/-/registered-dossier/5681. Accessed 15 Nov 2016
ECHA (2016d) European Chemicals Agency: REACH registration dossier, 4-tert-butylbenzoic acid. https://echa.europa.eu/registration-dossier/-/registered-dossier/12153. Accessed 11 Nov 2016
ECHA (2016e) European Chemicals Agency: REACH registration dossier, 4-tert-butyltoluene. https://echa.europa.eu/registration-dossier/-/registered-dossier/1614. Accessed 11 Nov 2016
ECHA (2016f) European Chemicals Agency: REACH registration dossier, 3-(4-tert-butylphenyl)propionaldehyde. https://echa.europa.eu/registration-dossier/-/registered-dossier/12753. Accessed 10 Nov 2016
ECHA (2016g) European Chemicals Agency: summary of classification and labeling, 3-(p-cumenyl)propionaldehyde. https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/84780. Accessed 15 Nov 2016
ECHA (2017a) Decision on compliance check: 3-p-cumenyl-2-methylpropionaldehyde.https://echa.europa.eu/documents/10162/3c90abf1-6aec-ad9e-b5c4-483b58b642fb. Accessed 02 Nov 2019
ECHA (2017b) European Chemicals Agency: REACH registration dossier, 3-(4-isobutyl-2-methylphenyl)propanal. https://echa.europa.eu/registration-dossier/-/registered-dossier/16715. Accessed 16 Oct 2017
ECHA (2017c) European Chemicals Agency: REACH registration dossier, 4-hydroxybenzoic acid. https://echa.europa.eu/registration-dossier/-/registered-dossier/15944/7/9/2. Accessed 16 Oct 2017
ECHA (2017d) European Chemicals Agency: REACH registration dossier, benzoic acid. https://echa.europa.eu/registration-dossier/-/registered-dossier/13124/7/9/2. Accessed 16 Oct 2017
ECHA (2017e) European Chemicals Agency: REACH registration dossier, ethyl 4-hydroxybenzoate. https://echa.europa.eu/registration-dossier/-/registered-dossier/13843/7/9/2. Accessed 16 Oct 2017
ECHA (2018a) 2-(4-tert-butylbenzyl)propionaldehyde. In: CLH report: proposal for harmonised classification and labelling. European Chemicals Agency. Accessed 30 Oct 2018
ECHA (2018b) European Chemicals Agency: REACH registration dossier, 2-(4-tert-butylbenzyl)propionaldehyde. https://echa.europa.eu/registration-dossier/-/registered-dossier/13572. Accessed 01 Oct 2018
ECHA (2019) Committee for Risk Assessment—Annex 2. Response to comments document (RCOM) to the Opinion proposing harmonised classification and labelling at EU level of 2-(4-tert-butylbenzyl)propionaldehyde. https://echa.europa.eu/documents/10162/23665416/clh_rcom_2-%284-tert-butylbenzyl%29propionaldehyde_11896_enpdf/1f6d5f80-d8bc-ae6c-eed2-1a8e7c813ebb. Adopted 28 Jan 2019
ECHA (2020a) European Chemicals Agency: REACH registration dossier, 3-(p-cumenyl)propionaldehyde. https://echa.europa.eu/de/registration-dossier/-/registered-dossier/22566/7/9/1. Accessed 04 Jun 2020
ECHA (2020b) European Chemicals Agency: REACH registration dossier, 3-(p-methoxyphenyl)-2-methylpropionaldehyde. https://echa.europa.eu/de/registration-dossier/-/registered-dossier/19498. Accessed 04 Jun 2020
ECHA (2020c) European Chemicals Agency: REACH registration dossier, α-methyl-1,3-benzodioxole-5-propionaldehyde. https://echa.europa.eu/de/registration-dossier/-/registered-dossier/20444. Accessed 04 Jun 2020
EU (2009) European Union risk assessment report: 4-tert-butylbenzoic acid. https://echa.europa.eu/documents/10162/15c7dba3-848b-463b-ae60-c6f187b7b5d4. Accessed 14 Nov 2016
Geoffroy-Siraudin C, Perrard MH, Chaspoul F et al (2010) Validation of a rat seminiferous tubule culture model as a suitable system for studying toxicant impact on meiosis effect of hexavalent chromium. Toxicol Sci 116(1):286–296. https://doi.org/10.1093/toxsci/kfq099
Goldstein KM, Seyler DE, Durand P, Perrard MH, Baker TK (2016) Use of a rat ex-vivo testis culture method to assess toxicity of select known male reproductive toxicants. Reprod Toxicol 60:92–103. https://doi.org/10.1016/j.reprotox.2016.01.003
Harshbarger KE (1942) Report of a study on the toxicity of several food preserving agents. J Dairy Sci 25(2):169–174. https://doi.org/10.3168/jds.S0022-0302(42)95278-0
Hunter CG, Chambers PL, Stevenson DE (1965) Studies on the oral toxicity of p-tert-butyl benzoic acid in rats. Food Cosmet Toxicol 3(2):289–298
Kasuya F, Oti Y, Tatsuki T, Igarashi K (2004) Analysis of medium-chain acyl-coenzyme A esters in mouse tissues by liquid chromatography-electrospray ionization mass spectrometry. Anal Biochem 325(2):196–205. https://doi.org/10.1016/j.ab.2003.10.043
Lassila T, Hokkanen J, Aatsinki SM, Mattila S, Turpeinen M, Tolonen A (2015) Toxicity of carboxylic acid-containing drugs: the role of acyl migration and CoA conjugation investigated. Chem Res Toxicol 28(12):2292–2303. https://doi.org/10.1021/acs.chemrestox.5b00315
Laue H, Kern S, Badertscher RP, Ellis G, Natsch A (2017) p-Alkyl-benzoyl-CoA conjugates as relevant metabolites of aromatic aldehydes with rat testicular toxicity—studies leading to the design of a safer new fragrance chemical. Toxicol Sci 160(2):244–255. https://doi.org/10.1093/toxsci/kfx178
Lauer B, Tuschl G, Kling M, Mueller SO (2009) Species-specific toxicity of diclofenac and troglitazone in primary human and rat hepatocytes. Chem-Biol Interact 179(1):17–24. https://doi.org/10.1016/j.cbi.2008.10.031
Mangino MJ, Zografakis J, Murphy MK, Anderson CB (1992) Improved and simplified tissue extraction method for quantitating long-chain acyl-coenzyme A thioesters with picomolar detection using high-performance liquid chromatography. J Chromatogr 577(1):157–162
RIFM (2010) Preliminary findings for the 14-day reproductive toxicity studies. http://www.rifm.org/doc/Table1%20Prelim%20Findings14-Day%20ReproTox%20Studies.pdf. Accessed 14 Nov 2016
Scherer M, Koch HM, Schutze A et al (2017) Human metabolism and excretion kinetics of the fragrance lysmeral after a single oral dosage. Int J Hyg Environ Health 220(2, Part A):123–129
Skonberg C, Olsen J, Madsen KG, Hansen SH, Grillo MP (2008) Metabolic activation of carboxylic acids. Expert Opin Drug Metab Toxicol 4(4):425–438. https://doi.org/10.1517/17425255.4.4.425
Smith DA, Hammond T, Baillie TA (2018) Safety assessment of acyl glucuronides—a simplified paradigm. Drug Metab Dispos 46(6):908–912. https://doi.org/10.1124/dmd.118.080515
Walles M, Brown AP, Zimmerlin A, End P (2020) New perspectives on drug-induced liver injury risk assessment of acyl glucuronides. Chem Res Toxicol 33(7):1551–1560. https://doi.org/10.1021/acs.chemrestox.0c00131
Yan Z, Li J, Huebert N, Caldwell GW, Du Y, Zhong H (2005) Detection of a novel reactive metabolite of diclofenac: Evidence for CYP2C9-mediated bioactivation via arene oxides. Drug Metab Disposition 33(6):706–713. https://doi.org/10.1124/dmd.104.003095
Acknowledgements
The authors thank Felix Flachsmann for preparing the p-tBBA-CoA conjugate and Thierry Granier for the synthesis of the glucuronides as reference. Furthermore, the authors are grateful for the very detailed comments and suggestions of the two anonymous reviewers.
Funding
This work was entirely funded by Givaudan, no external funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors are employees of Givaudan.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Laue, H., Badertscher, R.P., Hostettler, L. et al. Benzoyl-CoA conjugate accumulation as an initiating event for male reprotoxic effects in the rat? Structure–activity analysis, species specificity, and in vivo relevance. Arch Toxicol 94, 4115–4129 (2020). https://doi.org/10.1007/s00204-020-02918-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-020-02918-9