Abstract
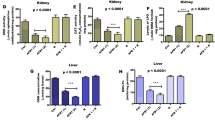
Fumonisin B1 (FB1), a congener of fumonisins produced by Fusarium species, is the most abundant and most toxicologically active fumonisin. FB1 causes severe mycotoxicosis in animals, including nephrotoxicity, hepatotoxicity, and disruption of the intestinal barrier. However, mechanisms associated with FB1 toxicity are still unclear. Preliminary studies have highlighted the role of liver X receptors (LXRs) during FB1 exposure. LXRs belong to the nuclear receptor family and control the expression of genes involved in cholesterol and lipid homeostasis. In this context, the toxicity of FB1 was compared in female wild-type (LXR+/+) and LXRα,β double knockout (LXR−/−) mice in the absence or presence of FB1 (10 mg/kg body weight/day) for 28 days. Exposure to FB1 supplemented in the mice’s drinking water resulted in more pronounced hepatotoxicity in LXR−/− mice compared to LXR+/+ mice, as indicated by hepatic transaminase levels (ALT, AST) and hepatic inflammatory and fibrotic lesions. Next, the effect of FB1 exposure on the liver transcriptome was investigated. FB1 exposure led to a specific transcriptional response in LXR−/− mice that included altered cholesterol and bile acid homeostasis. ELISA showed that these effects were associated with an elevated FB1 concentration in the plasma of LXR−/− mice, suggesting that LXRs participate in intestinal absorption and/or clearance of the toxin. In summary, this study demonstrates an important role of LXRs in protecting the liver against FB1-induced toxicity, suggesting an alternative mechanism not related to the inhibition of sphingolipid synthesis for mycotoxin toxicity.







Similar content being viewed by others
References
Asselah T, Bièche I, Laurendeau I et al (2005) Liver gene expression signature of mild fibrosis in patients with chronic hepatitis C. Gastroenterology 129:2064–2075. https://doi.org/10.1053/j.gastro.2005.09.010
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57:289–300
Benlasher E, Geng X, Xuan Nguyen NT et al (2012) Comparative effects of fumonisins on sphingolipid metabolism and toxicity in ducks and Turkeys. Avian Dis 56:120–127. https://doi.org/10.1637/9853-071911-Reg.1
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Bolstad BM, Irizarry RA, Astrand M, Speed TP (2003) A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19:185–193
Bondy GS, Suzuki CAM, Fernie SM et al (1997) Toxicity of fumonisin B1to B6C3F1mice: A 14-day gavage study. Food Chem Toxicol 35:981–989. https://doi.org/10.1016/S0278-6915(97)87267-5
Bonzón-Kulichenko E, Schwudke D, Gallardo N et al (2009) Central leptin regulates total ceramide content and sterol regulatory element binding protein-1C proteolytic maturation in rat white adipose tissue. Endocrinology 150:169–178. https://doi.org/10.1210/en.2008-0505
Burel C, Tanguy M, Guerre P et al (2013) Effect of low dose of fumonisins on pig health: immune status, intestinal microbiota and sensitivity to Salmonella. Toxins (Basel) 5:841–864. https://doi.org/10.3390/toxins5040841
Devriendt B, Gallois M, Verdonck F et al (2009) The food contaminant fumonisin B(1) reduces the maturation of porcine CD11R1(+) intestinal antigen presenting cells and antigen-specific immune responses, leading to a prolonged intestinal ETEC infection. Vet Res 40:40. https://doi.org/10.1051/vetres/2009023
Ducheix S, Montagner A, Theodorou V et al (2013) The liver X receptor: a master regulator of the gut–liver axis and a target for non alcoholic fatty liver disease. Biochem Pharmacol 86:96–105. https://doi.org/10.1016/j.bcp.2013.03.016
(Ec) No 1126/2007 Commission Regulation (2007) COMMISSION REGULATION (EC) No 1126/2007
(Ec) No 576/2006 Commission Recommendation (2006) COMMISSION RECOMMENDATION (EC) No 576/2006
Edgar R, Domrachev M, Lash AE (2002) Gene expression omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30:207–210
Gentleman RC, Carey VJ, Bates DM et al (2004) Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5:R80. https://doi.org/10.1186/gb-2004-5-10-r80
Grenier B, Bracarense A-PFL, Schwartz HE et al (2012) The low intestinal and hepatic toxicity of hydrolyzed fumonisin B1 correlates with its inability to alter the metabolism of sphingolipids. Biochem Pharmacol 83:1465–1473. https://doi.org/10.1016/j.bcp.2012.02.007
Gronemeyer H, Gustafsson J-Å, Laudet V (2004) Principles for modulation of the nuclear receptor superfamily. Nat Rev Drug Discov 3:950–964. https://doi.org/10.1038/nrd1551
Haschek WM, Gumprecht LA, Smith G et al (2001) Fumonisin toxicosis in swine: an overview of porcine pulmonary edema and current perspectives. Environ Health Perspect 109 Suppl:251–257
Holland WL, Bikman BT, Wang L-P et al (2011) Lipid-induced insulin resistance mediated by the proinflammatory receptor TLR4 requires saturated fatty acid–induced ceramide biosynthesis in mice. J Clin Invest 121:1858–1870. https://doi.org/10.1172/JCI43378
Howard PC, Eppley RM, Stack ME et al (2001) Fumonisin b1 carcinogenicity in a two-year feeding study using F344 rats and B6C3F1 mice. Environ Health Perspect 109:277–282. https://doi.org/10.1289/ehp.01109s2277
Howard PC, Couch LH, Patton RE et al (2002) Comparison of the toxicity of several fumonisin derivatives in a 28-day feeding study with female B6C3F(1) mice. Toxicol Appl Pharmacol 185:153–165
Humphreys SH, Carrington C, Bolger M (2001) A quantitative risk assessment for fumonisins B1 and B2 in US corn. Food Addit Contam 18:211–220. https://doi.org/10.1080/02652030010021486
JECFA (2001) Safety Evaluation of Certain Mycotoxins in Food (WHO Food Additives Series No. 47:150–161). 56th Meeting of the JECFA, Geneva, International Programme on Chemical Safety, WHO
Johnson V, Sharma R (2001) Gender-dependent immunosuppression following subacute exposure to fumonisin B1. Int Immunopharmacol 1:2023–2034. https://doi.org/10.1016/S1567-5769(01)00131-X
Loiseau N, Debrauwer L, Sambou T et al (2007) Fumonisin B1 exposure and its selective effect on porcine jejunal segment: Sphingolipids, glycolipids and trans-epithelial passage disturbance. Biochem Pharmacol. https://doi.org/10.1016/j.bcp.2007.03.031
Loiseau N, Polizzi A, Dupuy A et al (2015) New insights into the organ-specific adverse effects of fumonisin B1: comparison between lung and liver. Arch Toxicol 89:1619–1629. https://doi.org/10.1007/s00204-014-1323-6
Marasas WFO, Riley RT, Hendricks KA et al (2004) Fumonisins disrupt sphingolipid metabolism, folate transport, and neural tube development in embryo culture and in vivo: a potential risk factor for human neural tube defects among populations consuming fumonisin-contaminated maize. J Nutr 134:711–716. https://doi.org/10.1093/jn/134.4.711
Marin DE, Taranu I, Pascale F et al (2006) Sex-related differences in the immune response of weanling piglets exposed to low doses of fumonisin extract. Br J Nutr 95:1185–1192
Masching S, Naehrer K, Schwartz-Zimmermann H-E et al (2016) Gastrointestinal degradation of fumonisin b1 by carboxylesterase FumD prevents fumonisin induced alteration of sphingolipid metabolism in Turkey and swine. Toxins (Basel) 8:84. https://doi.org/10.3390/toxins8030084
Merrill AH, Wang E, Vales TR et al (1996) Fumonisin toxicity and sphingolipid biosynthesis. Adv Exp Med Biol 392:297–306
National Toxicology Program (2001) Toxicology and carcinogenesis studies of fumonisin B1 (cas no. 116355-83-0) in F344/N rats and B6C3F1 mice (feed studies). Natl Toxicol Program Tech Rep Ser (496):1–352
Ohno Y, Suto S, Yamanaka M et al (2010) ELOVL1 production of C24 acyl-CoAs is linked to C24 sphingolipid synthesis. Proc Natl Acad Sci USA 107:18439–18444. https://doi.org/10.1073/pnas.1005572107
Peet DJ, Turley SD, Ma W et al (1998) Cholesterol and bile acid metabolism are impaired in mice lacking the nuclear oxysterol receptor LXR alpha. Cell 93:693–704
R Core Team (2008) R: a language and environment for statistical computing
Raichur S, Wang ST, Chan PW et al (2014) CerS2 Haploinsufficiency Inhibits β-Oxidation and Confers Susceptibility to Diet-Induced Steatohepatitis and Insulin Resistance. Cell Metab 20:687–695. https://doi.org/10.1016/j.cmet.2014.09.015
Régnier M, Gourbeyre P, Pinton P et al (2017a) Identification of signaling pathways targeted by the food contaminant FB1: transcriptome and kinome analysis of samples from pig liver and intestine. Mol Nutr Food Res 61:1700433. https://doi.org/10.1002/mnfr.201700433
Régnier M, Polizzi A, Lippi Y et al (2017b) Insights into the role of hepatocyte PPARα activity in response to fasting. Mol Cell Endocrinol. https://doi.org/10.1016/j.mce.2017.07.035
Riedel S, Abel S, Burger H-M et al (2016) Differential modulation of the lipid metabolism as a model for cellular resistance to fumonisin B1—induced cytotoxic effects in vitro. Prostaglandins Leukot Essent Fat Acids 109:39–51. https://doi.org/10.1016/j.plefa.2016.04.006
Riley RT, Enongene E, Voss KA et al (2001) Sphingolipid perturbations as mechanisms for fumonisin carcinogenesis. Environ Health Perspect 109(Suppl 2):301–308
Rong X, Albert CJ, Hong C et al (2013) LXRs regulate ER stress and inflammation through dynamic modulation of membrane phospholipid composition. Cell Metab 18:685–697. https://doi.org/10.1016/j.cmet.2013.10.002
Rosenthal EA, Ronald J, Rothstein J et al (2011) Linkage and association of phospholipid transfer protein activity to LASS4. J Lipid Res 52:1837–1846. https://doi.org/10.1194/jlr.P016576
Scheek S, Brown MS, Goldstein JL (1997) Sphingomyelin depletion in cultured cells blocks proteolysis of sterol regulatory element binding proteins at site 1. Proc Natl Acad Sci USA 94:11179–11183
Scheig R (1996) Evaluation of tests used to screen patients with liver disorders. Prim Care 23:551–560
Schmidt E, Schmidt FW (1993) Enzyme diagnosis of liver diseases. Clin Biochem 26:241–251
Smyth GK (2004) Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3:1–25. https://doi.org/10.2202/1544-6115.1027
Stratford S, DeWald DB, Summers SA (2001) Ceramide dissociates 3′-phosphoinositide production from pleckstrin homology domain translocation. Biochem J 354:359–368
Szklarczyk D, Franceschini A, Wyder S et al (2015) STRING v10: protein–protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–D452. https://doi.org/10.1093/nar/gku1003
Teboul M, Enmark E, Li Q et al (1995) OR-1, a member of the nuclear receptor superfamily that interacts with the 9-cis-retinoic acid receptor. Proc Natl Acad Sci USA 92:2096–2100
Turpin SM, Nicholls HT, Willmes DM et al (2014) Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab 20:678–686. https://doi.org/10.1016/j.cmet.2014.08.002
Voss KA, Chamberlain WJ, Bacon CW et al (1995) Subchronic feeding study of the mycotoxin fumonisin B1 in B6C3F1 mice and Fischer 344 rats. Fundam Appl Toxicol 24:102–110. https://doi.org/10.1006/FAAT.1995.1012
Wan Norhas WM, Abdulamir AS, Abu Bakar F et al (2009) The health and toxic adverse effects of fusarium fungal mycotoxin, fumonisins, on human population. Am J Infect Dis 5:273–281. https://doi.org/10.3844/ajidsp.2009.273.281
Wang E, Norred WP, Bacon CW et al (1991) Inhibition of sphingolipid biosynthesis by fumonisins. Implications for diseases associated with Fusarium moniliforme. J Biol Chem 266:14486–14490
Wang B, Rong X, Duerr MA et al (2016) Intestinal phospholipid remodeling is required for dietary-lipid uptake and survival on a high-fat diet. Cell Metab 23:492–504. https://doi.org/10.1016/j.cmet.2016.01.001
Yang M, Wang C, Li S et al (2017) Annexin A2 promotes liver fibrosis by mediating von Willebrand factor secretion. Dig Liver Dis 49:780–788. https://doi.org/10.1016/j.dld.2017.02.013
Yu L, York J, von Bergmann K et al (2003) Stimulation of cholesterol excretion by the liver X receptor agonist requires ATP-binding cassette transporters G5 and G8. J Biol Chem 278:15565–15570. https://doi.org/10.1074/jbc.M301311200
Yu L, Gupta S, Xu F et al (2005) Expression of ABCG5 and ABCG8 Is required for regulation of biliary cholesterol secretion. J Biol Chem 280:8742–8747. https://doi.org/10.1074/jbc.M411080200
Acknowledgements
MR was supported by a Fellowship from the Ministère de l’Education Nationale, de la Recherche et de la Technologie. This study was supported by the ANR Fumolip (ANR-16-CE21-0003) and ANR LipoReg (ANR-15-Carn0016), France. We thank Dr. David J. Mangelsdorf (Howard Hughes Medical Institute, Dallas, TX) for providing us with the LXR-deficient mice and for constructive discussions. We thank all members of the EZOP staff. We thank Aurore Laurent Monbrun for his excellent work on plasma biochemistry. We also thank the staff from the Genotoul: Anexplo, GeT-TriX, and Metatoul-Lipidomic facilities.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Régnier, M., Polizzi, A., Lukowicz, C. et al. The protective role of liver X receptor (LXR) during fumonisin B1-induced hepatotoxicity. Arch Toxicol 93, 505–517 (2019). https://doi.org/10.1007/s00204-018-2345-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2345-2