Abstract
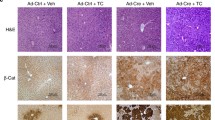
Activation of Wnt/β-catenin signaling is important for human and rodent hepatocarcinogenesis. In mice, the tumor promoter phenobarbital (PB) selects for hepatocellular tumors with activating β-catenin mutations via constitutive androstane receptor activation. PB-dependent tumor promotion was studied in mice with genetic inactivation of Apc, a negative regulator of β-catenin, to circumvent the problem of randomly induced mutations by chemical initiators and to allow monitoring of PB- and Wnt/β-catenin-dependent tumorigenesis in the absence of unknown genomic alterations. Moreover, the study was designed to investigate PB-induced proliferation of liver cells with activated β-catenin. PB treatment provided Apc-deficient hepatocytes with only a minor proliferative advantage, and additional connexin 32 deficiency did not affect the proliferative response. PB significantly promoted the outgrowth of Apc-deficient hepatocellular adenoma (HCA), but simultaneously inhibited the formation of Apc-deficient hepatocellular carcinoma (HCC). The probability of tumor promotion by PB was calculated to be much lower for hepatocytes with loss of Apc, as compared to mutational β-catenin activation. Comprehensive transcriptomic and phosphoproteomic characterization of HCA and HCC revealed molecular details of the two tumor types. HCC were characterized by a loss of differentiated hepatocellular gene expression, enhanced proliferative signaling, and massive over-activation of Wnt/β-catenin signaling. In conclusion, PB exerts a dual role in liver tumor formation by promoting the growth of HCA but inhibiting the growth of HCC. Data demonstrate that one and the same compound can produce opposite effects on hepatocarcinogenesis, depending on context, highlighting the necessity to develop a more differentiated view on the tumorigenicity of this model compound.






Similar content being viewed by others
References
Aydinlik H, Nguyen TD, Moennikes O, Buchmann A, Schwarz M (2001) Selective pressure during tumor promotion by phenobarbital leads to clonal outgrowth of beta-catenin-mutated mouse liver tumors. Oncogene 20:7812–7816
Behrens J (2000) Control of beta-catenin signaling in tumor development. Ann N Y Acad Sci 910:21–33
Braeuning A (2009) Regulation of cytochrome P450 expression by Ras- and beta-catenin-dependent signaling. Curr Drug Metab 10:138–158
Braeuning A (2014) Liver cell proliferation and tumor promotion by phenobarbital: relevance for humans? Arch Toxicol 88:1771–1772
Braeuning A, Sanna R, Huelsken J, Schwarz M (2009) Inducibility of drug-metabolizing enzymes by xenobiotics in mice with liver-specific knockout of Ctnnb1. Drug Metab Dispos 37:1138–1145
Braeuning A, Singh Y, Rignall B et al (2010) Phenotype and growth behavior of residual beta-catenin-positive hepatocytes in livers of beta-catenin-deficient mice. Histochem Cell Biol 134:469–481
Braeuning A, Heubach Y, Knorpp T et al (2011) Gender-specific interplay of signaling through beta-catenin and CAR in the regulation of xenobiotic-induced hepatocyte proliferation. Toxicol Sci 123:113–122
Buchmann A, Karcier Z, Schmid B, Strathmann J, Schwarz M (2008) Differential selection for B-raf and Ha-ras mutated liver tumors in mice with high and low susceptibility to hepatocarcinogenesis. Mutat Res 638:66–74
Cagatay T, Ozturk M (2002) P53 mutation as a source of aberrant beta-catenin accumulation in cancer cells. Oncogene 21:7971–7980
Caldwell CM, Kaplan KB (2009) The role of APC in mitosis and in chromosome instability. Adv Exp Med Biol 656:51–64
Campbell HA, Pitot HC, Potter VR, Laishes BA (1982) Application of quantitative stereology to the evaluation of enzyme-altered foci in rat liver. Cancer Res 42:465–472
Chen J, Bardes EE, Aronow BJ, Jegga AG (2009) ToppGene Suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res 37(Web Server issue):W305–W311
Colnot S, Decaens T, Niwa-Kawakita M et al (2004) Liver-targeted disruption of Apc in mice activates beta-catenin signaling and leads to hepatocellular carcinomas. Proc Natl Acad Sci USA 101:17216–17221
Dong B, Lee JS, Park YY et al (2015) Activating CAR and beta-catenin induces uncontrolled liver growth and tumorigenesis. Nat Commun 6:5944
Elcombe CR, Peffer RC, Wolf DC et al (2014) Mode of action and human relevance analysis for nuclear receptor-mediated liver toxicity: a case study with phenobarbital as a model constitutive androstane receptor (CAR) activator. Crit Rev Toxicol 44:64–82
Ganzenberg K, Singh Y, Braeuning A (2013) The time point of beta-catenin knockout in hepatocytes determines their response to xenobiotic activation of the constitutive androstane receptor. Toxicology 308:113–121
Gautier L, Cope L, Bolstad BM, Irizarry RA (2004) affy—analysis of Affymetrix GeneChip data at the probe level. Bioinformatics 20:307–315
Groll N, Kollotzek F, Goepfert J, Joos TO, Schwarz M, Braeuning A (2016) Phenobarbital inhibits calpain activity and expression in mouse hepatoma cells. Biol Chem 397:91–96
Kauffmann A, Gentleman R, Huber W (2009) arrayQualityMetrics—a bioconductor package for quality assessment of microarray data. Bioinformatics 25:415–416
Koike C, Moore R, Negishi M (2007) Extracellular signal-regulated kinase is an endogenous signal retaining the nuclear constitutive active/androstane receptor (CAR) in the cytoplasm of mouse primary hepatocytes. Mol Pharmacol 71:1217–1221
Lee GH (2000) Paradoxical effects of phenobarbital on mouse hepatocarcinogenesis. Toxicol Pathol 28:215–225
Lee GH, Ooasa T, Osanai M (1998) Mechanism of the paradoxical, inhibitory effect of phenobarbital on hepatocarcinogenesis initiated in infant B6C3F1 mice with diethylnitrosamine. Cancer Res 58:1665–1669
Marx-Stoelting P, Mahr J, Knorpp T et al (2008) Tumor promotion in liver of mice with a conditional Cx26 knockout. Toxicol Sci 103:260–267
Moennikes O, Buchmann A, Romualdi A et al (2000) Lack of phenobarbital-mediated promotion of hepatocarcinogenesis in connexin32-null mice. Cancer Res 60:5087–5091
Nault JC, Zucman-Rossi J (2011) Genetics of hepatobiliary carcinogenesis. Semin Liver Dis 31:173–187
Oda H, Imai Y, Nakatsuru Y, Hata J, Ishikawa T (1996) Somatic mutations of the APC gene in sporadic hepatoblastomas. Cancer Res 56:3320–3323
Pirnia F, Pawlak M, Thallinger GG et al (2009) Novel functional profiling approach combining reverse phase protein microarrays and human 3-D ex vivo tissue cultures: expression of apoptosis-related proteins in human colon cancer. Proteomics 9:3535–3548
Saeed AI, Bhagabati NK, Braisted JC et al (2006) TM4 microarray software suite. Methods Enzymol 411:134–193
Schneikert J, Behrens J (2007) The canonical Wnt signalling pathway and its APC partner in colon cancer development. Gut 56:417–425
Schreiber S, Rignall B, Braeuning A et al (2011) Phenotype of single hepatocytes expressing an activated version of beta-catenin in liver of transgenic mice. J Mol Histol 42:393–400
Singh Y, Braeuning A, Schmid A, Pichler BJ, Schwarz M (2013) Selective poisoning of Ctnnb1-mutated hepatoma cells in mouse liver tumors by a single application of acetaminophen. Arch Toxicol 87:1595–1607
Singh Y, Port J, Schwarz M, Braeuning A (2014) Genetic ablation of beta-catenin inhibits the proliferative phenotype of mouse liver adenomas. Br J Cancer 111:132–138
Smyth GK (2005) Limma: linear models for microarray data. In: Gentleman V, Carey S, Dudoit R (eds) Bioinformatics and computational biology solutions using R and bioconductor. Springer, New York, pp 397–420
Strathmann J, Schwarz M, Tharappel JC et al (2006) PCB 153, a non-dioxin-like Tumor Promoter, Selects for {beta}-Catenin (Catnb) Mutated Mouse Liver Tumors. Toxicol Sci 93:34–40
Tannour-Louet M, Porteu A, Vaulont S, Kahn A, Vasseur-Cognet M (2002) A tamoxifen-inducible chimeric Cre recombinase specifically effective in the fetal and adult mouse liver. Hepatology 35:1072–1081
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98:5116–5121
Unterberger EB, Eichner J, Wrzodek C et al (2014) Ha-ras and beta-catenin oncoproteins orchestrate metabolic programs in mouse liver tumors. Int J Cancer 135:1574–1585
Wachstein M, Meisel E (1957) Histochemistry of hepatic phosphatases of a physiologic pH; with special reference to the demonstration of bile canaliculi. Am J Clin Pathol 27:13–23
Wolff GL, Morrissey RL, Chen JJ (1986) Amplified response to phenobarbital promotion of hepatotumorigenesis in obese yellow Avy/A (C3H × VY) F-1 hybrid mice. Carcinogenesis 7:1895–1898
Zeller E, Hammer K, Kirschnick M, Braeuning A (2013) Mechanisms of RAS/beta-catenin interactions. Arch Toxicol 87:611–632
Acknowledgments
The authors thank Johanna Mahr, Silvia Vetter, and Elke Zabinsky for excellent technical assistance. The help of Janine Brettschneider with bioinformatic analyses is also greatly acknowledged. This work was supported by the European Union (Grant IMI-MARCAR).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Braeuning, A., Gavrilov, A., Geissler, M. et al. Tumor promotion and inhibition by phenobarbital in livers of conditional Apc-deficient mice. Arch Toxicol 90, 1481–1494 (2016). https://doi.org/10.1007/s00204-016-1667-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1667-1