Abstract
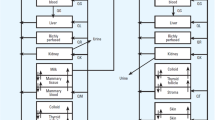
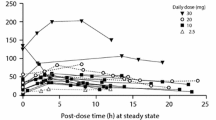
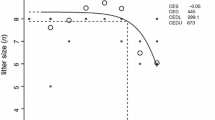
Chemical exposure via breast milk is one of the great concerns in public health. Previously, we demonstrated that most body burden of PCB 153 can be transferred from the mother to the pups in mice during lactational period. Here we present a physiologically based pharmacokinetic (PBPK) model to describe the lactational transfer of PCB 153 with or without PCB 126 in mice. The model incorporated physiological changes on the volume and the blood flow into mammary tissues, and considered mechanistic information on the movement of PCB 153 from adipose tissue to the mammary gland during lactational period. The mechanistic consideration includes fat volume changes, binding of PCB 153 to very low density lipoprotein (VLDL) and increased uptake of VLDL in mammary tissues. Model parameters depicting physiological changes were obtained from research articles dealing with chemical transfer during lactational period in rodents. Chemical-specific parameters were derived from previous PBPK models focusing on the PCB disposition in rodents. The developed model adequately described the lactational transfer of PCB 153 with or without PCB 126 in mice. Our model will provide a useful mechanistic tool to estimate the disposition of PCBs in diverse experimental designs regarding PCB effects during developmental period and to improve quantitative risk assessment of PCBs in the developing organisms.





Similar content being viewed by others
References
Anderson HA, Falk C, Hanrahan L, Olson J, Burse VW, Needham L, Paschal D, Patterson D Jr, Hill RH Jr (1998) Profiles of Great Lakes critical pollutants: a sentinel analysis of human blood and urine. The Great Lakes Consortium. Environ Health Perspect 106:279–289
Birnbaum LS, Weber H, Harris MW, Lamb JC, McKinney JD (1985) Toxic interaction of specific polychlorinated biphenyls and 2,3,7,8-tetrachlorodibenzo-p-dioxin: increased incidence of cleft palate in mice. Toxicol Appl Pharmacol 77:292–302
Borlakoglu JT, Clarke S, Huang SW, Dils RR, Haegele KD, Gibson GG (1992) Lactational transfer of 3,3′,4,4′-tetrachloro- and 2,2′,4,4′,5,5′-hexachlorobiphenyl induces cytochrome P450IVA1 in neonates. Evidence for a potential synergistic mechanism. Biochem Pharmacol 43:153–157
Brown RP, Delp MD, Lindstedt SL, Rhomberg LR, Beliles RP (1997) Physiological parameter values for physiologically based pharmacokinetic models. Toxicol Ind Health 13:407–484
Buck GM (1996) Epidemiologic perspective of the developmental neurotoxicity of PCBs in humans. Neurotoxicol Teratol 18:239–241; discussion 271–236
Carpenter DO (1998) Polychlorinated biphenyls and human health. Int J Occup Med Environ Health 11:291–303
Clewell RA, Gearhart JM (2002) Pharmacokinetics of toxic chemicals in breast milk: use of PBPK models to predict infant exposure. Environ Health Perspect 110:A333–A337
Corley RA, Mast TJ, Carney EW, Rogers JM, Daston GP (2003) Evaluation of physiologically based models of pregnancy and lactation for their application in children’s health risk assessment. CRC Crit Rev Toxicol 33:137–211
De Jongh J, Verhaar HJ, Hermens JL (1997) A quantitative property–property relationship (QPPR) approach to estimate in vitro tissue–blood partition coefficients of organic chemicals in rats and humans. Arch Toxicol 72:17–25
Fisher JW, Whittaker TA, Taylor DH, Clewell HJ 3rd, Andersen ME (1990) Physiologically based pharmacokinetic modeling of the lactating rat and nursing pup: a multiroute exposure model for trichloroethylene and its metabolite, trichloroacetic acid. Toxicol Appl Pharmacol 102:497–513
Gallenberg LA, Ring BJ, Vodicnik MJ (1990) The influence of time of maternal exposure to 2,2′,4,4′,5,5′-hexachlorobiphenyl on its accumulation in their nursing offspring. Toxicol Appl Pharmacol 104:1–8
Goldey ES, Kehn LS, Lau C, Rehnberg GL, Crofton KM (1995) Developmental exposure to polychlorinated biphenyls (Aroclor 1254) reduces circulating thyroid hormone concentrations and causes hearing deficits in rats. Toxicol Appl Pharmacol 135:77–88
Hansen LG (1998) Stepping backward to improve assessment of PCB congener toxicities. Environ Health Perspect 106(Suppl 1):171–189
Hsu ST, Ma CI, Hsu SK, Wu SS, Hsu NH, Yeh CC, Wu SB (1985) Discovery and epidemiology of PCB poisoning in Taiwan: a four-year followup. Environ Health Perspect 59:5–10
Huisman M, Koopman-Esseboom C, Fidler V, Hadders-Algra M, van der Paauw CG, Tuinstra LG, Weisglas-Kuperus N, Sauer PJ, Touwen BC, Boersma ER (1995) Perinatal exposure to polychlorinated biphenyls and dioxins and its effect on neonatal neurological development. Early Hum Dev 41:111–277
Jacobson JL, Jacobson SW, Humphrey HE (1990) Effects of in utero exposure to polychlorinated biphenyls and related contaminants on cognitive functioning in young children. J Pediatr 116:38–455
Kafafi SA, Afeefy HY, Ali AH, Said HK, Kafafi AG (1993) Binding of polychlorinated biphenyls to the aryl hydrocarbon receptor. Environ Health Perspect 101:422–428
Kedderis LB, Mills JJ, Andersen ME, Birnbaum LS (1993) A physiologically based pharmacokinetic model for TBDD in the rat: tissue distribution and CYP1A induction. Toxicol Appl Pharmacol 121:87–98
Koga N, Beppu M, Yoshimura H (1990) Metabolism in vivo of 3,4,5,3′,4′-pentachlorobiphenyl and toxicological assessment of the metabolite in rats. J Pharmacobiodyn 13:497–506
Kuratsune M, Yoshimura T, Matsuzaka J, Yamaguchi A (1971) Yusho, a poisoning caused by rice oil contaminated with polychlorinated biphenyls. HSMHA Health Rep 86:1083–1091
Landers JP, Bunce NJ (1991) The Ah receptor and the mechanism of dioxin toxicity. Biochem J 276:273–287
Lee SK (2005) Perinatal pharmacokinetics. In: Reddy MB, Yang RSH, Clewell HJ, Andersen ME (eds) Physiologically based pharmacokinetic modeling. Wiley, Hoboken, pp. 321–347
Lee SK, Ou YC, Yang RSH (2002) Comparison of pharmacokinetic interactions and physiologically based pharmacokinetic modeling of PCB 153 and PCB 126 in nonpregnant mice, lactating mice, and suckling pups. Toxicol Sci 65:26–34
Lutz RJ, Dedrick RL, Matthews HB, Eling TE, Anderson MW (1977) A preliminary pharmacokinetic model for several chlorinated biphenyls in the rat. Drug Metab Dispos 5:386–396
Lutz RJ, Dedrick RL, Tuey D, Sipes IG, Anderson MW, Matthews HB (1984) Comparison of the pharmacokinetics of several polychlorinated biphenyls in mouse, rat, dog, and monkey by means of a physiological pharmacokinetic model. Drug Metab Dispos 12:527–535
Matsumura F (1995) Mechanism of action of dioxin-type chemicals, pesticides, and other xenobiotics affecting nutritional indexes. Am J Clin Nutr 61(Suppl 3):695S–711S
Matthews HB, Dedrick RL (1984) Pharmacokinetics of PCBs. Annu Rev Pharmacol Toxicol 24:85–103
Mimura J, Fujii-Kuriyama Y (2003) Functional role of AhR in the expression of toxic effects by TCDD. Biochim Biophys Acta 1619:263–268
Noren K, Weistrand C, Karpe F (1999) Distribution of PCB congeners, DDE, hexachlorobenzene, and methylsulfonyl metabolites of PCB and DDE among various fractions of human blood plasma. Arch Environ Contam Toxicol 37:408–414
Olsen H, Enan E, Matsumura F (1998) TCDD mechanism of action to reduce lipoprotein lipase activity in the 3T3-L1 preadipocyte cell line. J Biochem Mol Toxicol 12:29–39
Oskarsson A, Palminger HI, Sundberg J, Petersson GK (1998) Risk assessment in relation to neonatal metal exposure. Analyst 123:19–23
Poulin P, Krishnan K (1995a) An algorithm for predicting tissue: blood partition coefficients of organic chemicals from n-octanol:water partition coefficient data. J Toxicol Environ Health 46:117–129
Poulin P, Krishnan K (1995b) A biologically-based algorithm for predicting human tissue: blood partition coefficients of organic chemicals. Hum Exp Toxicol 14:273–280
Prabhakaran S, Devi KS (1993) Impact of protein deficiency and exposure to hexachlorocyclohexane or malathion on lipid metabolism in pregnant rats. Indian J Biochem Biophys 30:234–238
Ring BJ, Seitz KR, Gallenberg LA, Vodicnik MJ (1990) The effect of diet and litter size on the elimination of 2,2′,4,4′,5,5′-hexachlorobiphenyl from lactating mice. Toxicol Appl Pharmacol 104:9–16
Ross G (2004) The public health implications of polychlorinated biphenyls (PCBs) in the environment. Ecotoxicol Environ Saf 59:275–291
Safe SH (1994) Polychlorinated biphenyls (PCBs): environmental impact, biochemical and toxic responses, and implications for risk assessment. Crit Rev Toxicol 24:87–149
Seegal RF, Brosch KO, Okoniewski RJ (1997) Effects of in utero and lactational exposure of the laboratory rat to 2,4,2′,4′- and 3,4,3′,4′-tetrachlorobiphenyl on dopamine function. Toxicol Appl Pharmacol 146:95–103
Spindler-Vomachka M, Vodicnik MJ (1984) Distribution of 2,4,5,2′,4′,5′-hexachlorobiphenyl among lipoproteins during pregnancy and lactation in the rat. J Pharmacol Exp Ther 230:263–268
Tang C, Lin JH, Lu AYH (2005) Metabolism-based drug–drug interactions: what determines individual variability in cytochrome P450 induction? Drug Metab Dispos 33:603–613
Tilson HA, Jacobson JL, Rogan WJ (1990) Polychlorinated biphenyls and the developing nervous system: cross-species comparisons. Neurotoxicol Teratol 12:239–488
Tuey DB, Matthews HB (1980) Use of a physiological compartmental model for the rat to describe the pharmacokinetics of several chlorinated biphenyls in the mouse. Drug Metab Dispos 8:397–403
Vodicnik MJ, Lech JJ (1980) The transfer of 2,4,5,2′,4′,5′-hexachlorobiphenyl to fetuses and nursing offspring. I. Disposition in pregnant and lactating mice and accumulation in young. Toxicol Appl Pharmacol 54:293–300
Weistrand C, Noren K (1998) Polychlorinated naphthalenes and other organochlorine contaminants in human adipose and liver tissue. J Toxicol Environ Health 53:293–311
Wolfle D (1997) Interactions between 2,3,7,8-TCDD and PCBs as tumor promoters: limitations of TEFs. Teratog Carcinog Mutagen 17:217–224
Acknowledgments
This study was supported in part by the NIEHS research grant (R03 ES10116-01), ATSDR Cooperative Agreement (U61/ATU 881475), and NIEHS Superfund Basic Research Program (P42 ES 05949). The studies were conducted in accordance with the National Institutes of Health (NIH) guidelines for the care and use of laboratory animals.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
The mathematical expressions of the model which described the mass balance on different compartments are formulated as outlined in Ramsey and Andersen (1984). Equation 1 represents a tissue mass balance equation for a flow-limited transport process.
where A i is amount in ith tissue; Q i is blood flow to ith tissue. C B is arterial blood concentration; C Vi is venous blood concentration leaving ith tissue. C Vi = C i /P i , where P i is tissue/blood partition coefficient, C i is concentration in ith tissue.
C i = A i /V i ; where V i is the volume of ith tissue.
Equations 2 and 3 are tissue mass balance equations for a diffusion-limited transport process; under this assumption, C i /P i does not equal to C Vi .
where A iB is amount in the blood compartment of ith tissue; Q i is blood flow to ith tissue; P Ai is the diffusion permeation constant of ith tissue.
For the liver compartment, additional terms were added to describe a first-order oral absorption of PCB 153 and to account for a first-order metabolism of PCB 153. The following equation described above-mentioned mass transfer.
where K AS is the first-order absorption rate constant from the stomach; K AD is the first-order absorption rate constant from duodenum; A ST is the amount in stomach; A DU is the amount in duodenum; K FC is the first-order metabolism constant in the liver.
For model development involving co-exposure with PCB 126, the partition coefficient in the liver (PL) was modified to a time-dependent equation, PL = 10 + KFL × T where KFL is the coefficient for time-dependent increase of partition coefficient and T is the hours after PCB 153 exposure, where 0 ≤ T ≤ 340 h (14 days).
Oral absorption of PCB 153 was described as follows.
where K DT is the transfer rate of PCB 153 from stomach to duodenum.
The rate change in the amount of PCB 153 in the milk compartment of lactating dams comprises two terms; one related to the maternal blood circulation and partitioning to the milk and the other to pup suckling. The equation for the mammary tissue/milk is
where A MA is the amount in mammary/milk compartment; Q MA is blood flow to mammary/milk; C B is arterial blood concentration. N is the number of pups. C VMA = C MA/P MA, where P MA is mammary/milk tissue:blood partition coefficient. C VMA is venous blood concentration leaving mammary/milk tissue. C MA is concentration in mammary/milk tissue. The second term (dA suck/dt) is identical to the pup litter suckling rate.
where K MILK is the milk transfer rate to the pups and was assumed to equal the milk production rate.
The amount of PCB 153 transferred to the pup’s liver is modeled via Eq. 11:
where A LP is the amount in pup’s liver; Q LP is blood flow to pup’s liver; C BP is arterial blood concentration in the pups.
where P LP is partition coefficient and C VLP is venous blood concentration leaving pup’s liver. For model development involving co-exposure with PCB 126, P LP was modified to a time-dependent equation, P LP = 20 + KFLP × T where KFLP is the coefficient for time-dependent increase of partition coefficient and T is the hours after PCB 153 exposure, where 0 ≤ T ≤ 340 h (14 days).
Physiological changes during lactational period were described using GRAPH function. For example, body weight changes of the dam and the pup were described as follows.
where T i is the first measured time point; BW i is the first measured body weight; T f is the last measured time point; BW f is the last measured body weight. For the body weight of pup, BW p is used to discriminate from the dam. The changes of cardiac output (QCC), blood flow in mammary glands (QMAC), volume in mammary glands (VMAC), and fat volume (VFC) were also described in a same way.
Rights and permissions
About this article
Cite this article
Lee, S.K., Ou, Y.C., Andersen, M.E. et al. A physiologically based pharmacokinetic model for lactational transfer of PCB 153 with or without PCB 126 in mice. Arch Toxicol 81, 101–111 (2007). https://doi.org/10.1007/s00204-006-0130-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-006-0130-0