Abstract
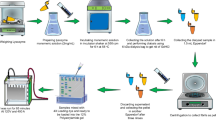
Aggregation of α-synuclein into toxic oligomeric structures has been implicated in the pathogenesis of Parkinson’s disease via several key stages of fibrillation, oligomerization, and aggregation. Disaggregation or prevention of aggregation has garnered a lot of attention as a therapeutic strategy to prevent or delay the progression of Parkinson’s disease. It has been recently established that certain polyphenolic compounds and catechins present in plants and tea extracts exhibit the potential to inhibit the α-synuclein aggregation. However, their copious supply for therapeutic development is still unsolved. Herein, we report for the first time the disaggregation potential of α-synuclein by an endophytic fungus residing in tea leaves (Camellia sinensis). Briefly, a recombinant yeast expressing α-synuclein was used for pre-screening of 53 endophytic fungi isolated from tea using anti-oxidant activity as a marker for the disaggregation of the protein. One isolate #59CSLEAS exhibited 92.4% reduction in production of the superoxide ions, which were similar to the already established α-synuclein disaggregator, Piceatannol exhibiting 92.8% reduction. Thioflavin T assay further established that #59CSLEAS decreased the oligomerization of α-synuclein by 1.63-fold. Subsequently Dichloro-dihydro-fluorescein diacetate-based fluorescence assay exhibited a reduction in total oxidative stress in the recombinant yeast in the presence of fungal extract, thereby indicating the prevention of oligomerization. Oligomer disaggregation potential of the selected fungal extract was found to be 56.5% as assessed by sandwich ELISA assay. Using morphological as well as molecular methods, the endophytic isolate #59CSLEAS was identified as Fusarium sp. The sequence was submitted in the Genbank with accession number ON226971.1.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable and justifiable request.
References
Alam P, Bousset L, Melki R, Otzen DE (2019) α-synuclein oligomers and fibrils: a spectrum of species, a spectrum of toxicities. J Neurochem 150:522–534. https://doi.org/10.1111/jnc.14808
Albani D, Polito L, Batelli S, De Mauro S, Fracasso C, Martelli G, Colombo L, Manzoni C, Salmona M, Caccia S, Negro A, Forloni G (2009) The SIRT1 activator resveratrol protects SK-N-BE cells from oxidative stress and against toxicity caused by α-synuclein or amyloid-β (1–42) peptide. J Neurochem 110(5):1445–1446. https://doi.org/10.1111/j.1471-4159.2009.06228.x
Bernal-Conde LD, Ramos-Acevedo R, Reyes-Hernandez MA, Balbuena-Olvera AJ, Morales-Moreno ID, Arguero- Sanchez R, Schule B, Guerra-Crespo M (2020) Alpha-synuclein physiology and pathology: a perspective on cellular structures and organelles. Front Neurosci 13:1399. https://doi.org/10.3389/fnins.2019.01399
Berrocal R, Vasudevaraju P, Indi SS, Sambasiva Rao KR, Rao KS (2014) In vitro evidence that an aqueous extract of Centella asiatica modulates α-synuclein aggregation dynamics. J Alzheimer’s Dis 39(2):457–465. https://doi.org/10.3233/JAD-131187
Bielecka M, Pencakowski B, Nicoletti R (2022) Using next-generation sequencing technology to explore genetic pathways in endophytic fungi in the syntheses of plant bioactive metabolites. Agriculture 12(2):187. https://doi.org/10.3390/agriculture12020187
Brundin P, Dave KD, Kordower JH (2017) Therapeutic approaches to target alpha-synuclein pathology. Exp Neurol 298(Pt B):225–235. https://doi.org/10.1016/j.expneurol.2017.10.003
Caruana M, Cauchi R, Vassallo N (2016) Putative role of red wine polyphenols against brain pathology in Alzheimer’s and Parkinson’s disease. Front Nutr 3:31. https://doi.org/10.3389/fnut.2016.00031
Caruana M, Högen T, Levin J, Hillmer A, Giese A, Vassallo N (2011) Inhibition and disaggregation of alpha-synuclein oligomers by natural polyphenolic compounds. FEBS Lett 585:1113–1120. https://doi.org/10.1016/j.febslet.2011.03.046
Caruana M, Neuner J, Högen T, Schmidt F, Kamp F, Scerri C, Giese A, Vassallo N (2012) Polyphenolic compounds are novel protective agents against lipid membrane damage by alpha-synuclein aggregates in vitro. Biochim Biophys Acta 1818:2502–2510. https://doi.org/10.1016/j.bbamem.2012.05.019
Caruana M, Vassallo N (2015) Tea polyphenols in Parkinson’s disease. Adv Exp Med Biol 863:117–137. https://doi.org/10.1007/978-3-319-18365-7_6
Celej MS, Sarroukh R, Goormaghtigh E, Fidelio GD, Ruysschaert JM, Raussens V (2012) Toxic prefibrillar alpha-synuclein amyloid oligomers adopt a distinctive antiparallel beta-sheet structure. Biochem J 443(3):719–726. https://doi.org/10.1042/BJ20111924
Chau E, Kim H, Shin J, Martinez A, Kim JR (2021) Inhibition of alpha-synuclein aggregation by AM17, a synthetic resveratrol derivative. Biochem Biophys Res Commun 574:85–90. https://doi.org/10.1016/j.bbrc.2021.08.049
Chen SW, Drakulic S, Deas E, Ouberai M, Aprile FA, Arranz R, Ness S, Roodveldt C, Guilliams T, De-Genst EJ, Klenerman D, Wood NW, Knowles TPJ, Alfonso C, Rivas G, Abramov AY, Valpeusta JM, Dobson CM, Cremades N (2015) Structural characterization of toxic oligomers that are kinetically trapped during alpha-synuclein fibril formation. Proc Natl Acad Sci USA 112(16):E1994–E2003. https://doi.org/10.1073/pnas.1421204112
da Costa IM, Cavalcanti JRLP, de Queiroz DB, de Azevedo EP, Do Rêgo ACM, Filho AI, Parente P, Botelho MA, Guzen FP (2017) Supplementation with herbal extracts to promote behavioural and neuroprotective effects in experimental models of Parkinson’s disease: a systematic review. Phytother Res 31:959–970. https://doi.org/10.1002/ptr.5813
Dorsey ER, Sherer T, Okun MS, Bloem BR (2018) The emerging evidence of the Parkinson’s disease pandemic. J Parkinsons Dis 8(S1):S3–S8. https://doi.org/10.3233/JPD-181474
Esteves AR, Arduino DM, Swerdlow RH, Oliveira CR, Cardoso SM (2009) Oxidative stress involvement in α-synuclein oligomerization in Parkinson’s disease cybrids. Antioxid Redox Signal 11(3):439–448. https://doi.org/10.1089/ars.2008.2247
Estuan-Panzano J, Arotcarena MA, Bezard E (2023) Monitoring alpha-synuclein aggregation. Exp Neurol 298(Pt B):225–235. https://doi.org/10.1016/j.nbd.2022.105966
Fields CR, Bengoa-Vergniory N, Wade-Martins R (2019) Targeting alpha-synuclein as a therapy for Parkinson’s disease. Front Mol Neurosci 12:299. https://doi.org/10.3389/fnmol.2019.00299
Fink AL (2006) The aggregation and fibrillation of alpha-synuclein. Acc Chem Res 39:628–634. https://doi.org/10.1021/ar050073t
El Gaamouch F, Liu K, Lin HY, Wu C, Wang J (2021) Development of grape polyphenols as multi- targeting strategies for Alzheimer’s disease. Neurochem Int 147:105046. https://doi.org/10.1016/j.neuint.2021.105046
Goedert M, Masuda-Suzukake M, Falcon B (2017) Like prions: the propagation of aggregated tau and α-synuclein in neurodegeneration. Brain 140(2):266–278. https://doi.org/10.1093/brain/aww230
Goswami A, Dikshit P, Mishra A, Mulherkar S, Nukina N, Jana NR (2006) Oxidative stress promotes mutant huntingtin aggregation and mutant huntingtin-dependent cell death by mimicking proteasomal malfunction. Biochem Biophys Res Commun 342(1):184–190. https://doi.org/10.1016/j.bbrc.2006.01.136
Griendling KK, Touyz RM, Zweier JL, Dikalov S, Chilian W, Chen YR, Harrison DG, Bhatnagar A (2016) Measurement of reactive oxygen species, reactive nitrogen species, and redox-dependent signalling in the cardiovascular system. Circ Res 119(5):e39–e75. https://doi.org/10.1161/RES.0000000000000110
Hansen CA, Miller DR, Annarumma S, Rusch CT, Ramirez-Zamora A, Khoshbouei H (2022) Levodopa-induced dyskinesia: a historical review of Parkinson’s disease, dopamine, and modern advancements in research and treatment. J Neurol 269:2892–2909. https://doi.org/10.1007/s00415-022-10963-w
Henrich CJ, Beutler JA (2013) Matching the power of high throughput screening to the chemical diversity of natural products. Nat Prod Rep 30:1284–1298. https://doi.org/10.1007/s00415-022-10963-w
Hu X, Dong D, Xia M, Yang Y, Wang J, Su J, Sun L, Yu H (2020) Oxidative stress and antioxidant capacity: development and prospects. New J Chem 44:11405–11419. https://doi.org/10.1039/D0NJ02041A
Inoue E, Shimizu Y, Masui R, Hayakawa T, Tsubonoya T, Hori S et al (2018) Effects of saffron and its constituents, crocin-1, crocin-2, and crocetin on α-synuclein fibrils. J Nat Med 72:274–279. https://doi.org/10.1007/s11418-017-1150-1
Jadiya P, Khan A, Sammi SR, Kaur S, Mir SS, Nazira A (2011) Anti-Parkinsonian effects of Bacopa monnieri: insights from transgenic and pharmacological Caenorhabditis elegans models of Parkinson’s disease. Biochem Biophys Res Commun 413:605–610. https://doi.org/10.1016/j.bbrc.2011.09.010
Javed H, Meeran MFN, Azhimullah S, Adem A, Sadek B, Ojha SK (2019) Plant extracts and phytochemicals targeting α-synuclein aggregation in Parkinson’s disease models. Front Pharmacol 9:1555. https://doi.org/10.3389/fphar.2018.01555
Javvaji PK, Dhali A, Francis JR, Kolte AP, Mech A, Roy SC, Mishra A, Bhatta R (2020) An efficient nitroblue tetrazolium staining and bright-field microscopy based method for detecting and quantifying intracellular reactive oxygen species in oocytes, cumulus cells and embryos. Front Cell Dev Biol 8:764. https://doi.org/10.3389/fcell.2020.00764
Kapoor N, Saxena S (2014) Potential xanthine oxidase inhibitory activity of endophytic Lasiodiplodia pseudotheobromae. Appl Biochem Biotechnol 173:1360–1374. https://doi.org/10.1007/s12010-014-0927-x
Kapoor N, Saxena S (2016) Xanthine oxidase inhibitory and antioxidant potential of Indian Muscodor species. 3Biotech 6:248. https://doi.org/10.1007/s13205-016-0569-5
Keston AS, Brandt R (1965) The fluorometric analysis of ultra-microquantities of hydrogen peroxide. Anal Biochem 11(1):1–5. https://doi.org/10.1016/0003-2697(65)90034-5
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/bf01731581
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis a cross computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Kõljalg U, Larsson KH, Abarenkov K, Nilsson RH, Alexander IJ, Eberhardt U, Erland S, Høiland K, Kjøller R, Larsson E, Pennanen T, Sen R, Taylor AFS, Tedersoo L, Vrålsta T (2005) UNITE: a database providing web-based methods for the molecular identification of ectomycorrhizal fungi. New Phytol 166:1063–1068. https://doi.org/10.1111/j.1469-8137.2005.01376.x
Langston JW, Schüle B, Rees L, Nichols RJ, Barlow C (2015) Multisystem Lewy body disease and the other parkinsonian disorders. Nat Genet 47(12):1378–1384. https://doi.org/10.1038/ng.3454
Lassen LB, Gregersen E, Isager AK, Betzer C, Kofoed RH, Jensen PH (2018) ELISA method to detect α-synuclein oligomers in cell and animal models. PLoS One 13(4):e0196056. https://doi.org/10.1371/journal.pone.0196056
LeBel CP, Ischiropoulos H, Bondy SC (1992) Evaluation of the probe 2’,7’-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem Res Toxicol 5(2):227–231. https://doi.org/10.1021/tx00026a012
Lee YJ, Burlet E, Wang S, Xu B, Huang S, Galiano FJ, Witt SN (2011) Triclabendazole protects yeast and mammalian cells from oxidative stress: identification of a potential neuroprotective compound. Biochem Biophys Res Commun 414(1):205–208. https://doi.org/10.1016/j.bbrc.2011.09.057
Lees AJ, Hardy J, Revesz T (2009) Parkinson’s disease. Lancet 373:2055–2066. https://doi.org/10.1016/S0140-6736(09)60492-X
Lundvig D, Lindersson E, Jensen PH (2005) Pathogenic effects of alpha-synuclein aggregation. Brain Res Mol Brain Res 134:3–17. https://doi.org/10.1016/j.molbrainres.2004.09.001
Magalhães P, Lashuel HA (2022) Opportunities and challenges of alpha-synuclein as a potential biomarker for Parkinson’s disease and other synucleinopathies. Npj Parkinsons Dis 8:93. https://doi.org/10.1038/s41531-022-00357-0
Mazo NA, Echeverria V, Cabezas R, Avila-Rodriguez M, Tarasov VV, Yarla NS, Aliev G, Barreto EG (2017) Medicinal plants as protective strategies against Parkinson’s disease. Curr Pharm Des 23:4180–4188. https://doi.org/10.2174/1381612823666170316142803
Musavi SF, Balakrishnan RM (2013) Biodiversity, antimicrobial potential, and phylogenetic placement of an endophytic Fusarium oxysporum NFX 06 isolated from Nothapodytes foetida. J Mycol. https://doi.org/10.1155/2013/172056
Najmi A, Javed SA, Al Bratty M, Alhazmi HA (2022) Modern approaches in the discovery and development of plant-based natural products and their analogues as potential therapeutic agents. Molecules 27(2):349. https://doi.org/10.3390/molecules27020349
Nam D, Lee JY, Lee M, Kim J, Seol W, Son I, Ho DH (2020) Detection and assessment of α-synuclein oligomers in the urine of Parkinson’s disease patients. J Parkinsons Dis 10(3):981–991
Ono K, Tsuji M, Yamasaki TR, Pasinetti GM (2020) Anti-aggregation effects of phenolic compounds on alpha-synuclein. Molecules 25(10):2444. https://doi.org/10.3390/molecules25102444
Pagan FL, Hebron ML, Wilmarth B, Torres-Yaghi Y, Lawler A, Mundel EE, Yusuf N, Starr NJ, Arellano J, Howard HH, Peyton M, Matar S, Liu X, Fowler AJ, Schwartz SL, Ahn J, Moussa C (2019) Pharmacokinetics and pharmacodynamics of a single dose Nilotinib in individuals with Parkinson’s disease. Pharmacol Res Perspect 7(2):e00470. https://doi.org/10.1002/prp2.470
Pezzoli G, Zini M (2010) Levodopa in Parkinson’s disease: from the past to the future. Expert Opin Pharmacother 11(4):627–635. https://doi.org/10.1517/14656561003598919
Rho T, Choi SK, Jung M, Kil HW, Hong YD, Yoon KD (2019) Identification of fermented tea (Camellia sinensis) polyphenols and their inhibitory activities against amyloid-beta aggregation. Phytochemistry 160:11–18. https://doi.org/10.1016/j.phytochem.2018.12.013
Schoch CL, Seifert KA, Hundorf S, Robert V, Spouge JL, Levesque CA, Chen W, Fungal Barcoding Consortium (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci USA 109:6241–6246. https://doi.org/10.1073/pnas.1117018109
Schulz B, Boyle C (2005) The endophytic continuum. Mycol Res 109(6):661–686. https://doi.org/10.1017/s095375620500273x
Shaltiel-Karyo R, Davidi D, Frenkel-Pinter M, Ovadia M, Segal D, Gazit E (2012) Differential inhibition of α-synuclein oligomeric and fibrillar assembly in Parkinson’s disease model by cinnamon extract. Biochim Biophys Acta 1820:1628–1635. https://doi.org/10.1016/j.bbagen.2012.04.021
Siddique YH, Mujtaba SF, Jyoti S, Naz F (2013) GC-MS analysis of Eucalyptus citriodora leaf extract and its role on the dietary supplementation in transgenic Drosophila model of Parkinson’s disease. Food Chem Toxicol 55:29–35. https://doi.org/10.1016/j.fct.2012.12.028
Strobel GA (2003) Endophytes as sources of bioactive products. Microbes Infect 5(6):535–544. https://doi.org/10.1016/S1286-4579(03)00073-X
Takahashi R, Ono K, Takamura Y, Mizuguchi M, Ikeda T, Nishijo H, Yamada M (2015) Phenolic compounds prevent the oligomerization of a-synuclein and reduce synaptic toxicity. J Neurochem 134:943–955. https://doi.org/10.1111/jnc.13180
Vats S, Kondabala R, Saxena S (2022) Identification of alpha-synuclein disaggregator from Camellia sp. insight of molecular docking and molecular dynamics simulations. ChemistrySelect 7:e202104131. https://doi.org/10.1002/slct.202104131
Venugopalan A, Srivastava S (2015) Endophytes as in vitro production platforms of high value plant secondary metabolites. Biotechnol Adv 33(6):873–887. https://doi.org/10.1016/biotechadv.2015.07.004
Visentin I, Tamietti G, Valentino D, Portis E, Karlovsky P, Moretti A, Cardinale F (2009) The ITS region as a taxonomic discriminator between Fusarium verticillioides and Fusarium proliferatum. Mycol Res 113(10):1137–1145. https://doi.org/10.1016/j.mycres.2009.07.011
Vivacqua G, Latorre A, Suppa A, Nardi M, Pietracupa S, Mancinelli R, Fabbrini G, Colosimo C, Gaudio E, Berardelli A (2016) Abnormal salivary total and oligomeric alpha-synuclein in Parkinson’s disease. PLoS One 11(3):e0151156. https://doi.org/10.1371/journal.pone.0151156
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27(5–6):612–616. https://doi.org/10.1016/s0891-5849(99)00107-0
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ White TJ, Eds., PCR Protocols: A Guide to Methods and Applications Academic Press New York 315-322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Wilson D (1995) Endophyte: the evolution of a term and clarification of its use and definition. Oikos 73:274–276. https://doi.org/10.2307/3545919
Winner BR, Jappelli SK, Maji PA, Boyer DL, Aigner S, Hetzer C, Loher T, Vilar CS, Tzitzilonis C, Soragni A, Jessberger A, Mira H, Consiglio A, Pham E, Masliah E, Gage FH, Riek R (2011) In vivo demonstration that alpha-synuclein oligomers are toxic. Proc Natl Acad Sci USA 108(10):4194–4199. https://doi.org/10.1073/pnas.1100976108
Witt SN, Flower TR (2006) α-Synuclein, oxidative stress and apoptosis from the perspective of a yeast model of Parkinson’s disease. FEMS Yeast Res 6(8):1107–1116. https://doi.org/10.1111/j.1567-1364.2006.00135.x
Wordehoff MM, Hoyer W (2018) α-Synuclein aggregation monitored by thioflavin T fluorescence assay. BioProtocol J. https://doi.org/10.21769/BioProtoc.2941
Xie H, Feng X, Wang M, Wang Y, Awasthi MK, Xu P (2020) Implications of endophytic microbiota in Camellia sinensis: a review on current understanding and future insights. Bioengineered 11(1):1001–1015. https://doi.org/10.1080/21655979.2020.1816788
Zhao J, Liang Q, Sun Q, Chen C, Xu L, Ding Y, Zhou P (2017) (−)-Epigallocatechin-3-gallate (EGCG) inhibits fibrillation, disaggregates amyloid fibrils of α-synuclein, and protects PC12 cells against α-synuclein-induced toxicity. RSC Adv 7:32508–32517. https://doi.org/10.1039/C7RA03752J
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
SS conceived the research. SV performed the research and acquired the data. Both authors analyzed and interpreted the data and were involved in drafting the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or non-financial interest to disclose.
Ethical approval
Not applicable.
Consent to participate
All authors agree on what is described about their participation in this work.
Consent to publication
All authors agree on the submission of the manuscript.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vats, S., Saxena, S. Endophytic Fusarium species, a unique bioresource for disaggregator of misfolded alpha-synuclein. Arch Microbiol 205, 224 (2023). https://doi.org/10.1007/s00203-023-03575-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03575-z