Abstract
Wastewater monitoring for SARS-CoV-2 has attracted considerable attention worldwide to complement the existing clinical-based surveillance system. In this study, we report our first successful attempt to prove the circulation of SARS-CoV-2 genes in Malaysian urban wastewater. A total of 18 wastewater samples were obtained from a regional sewage treatment plant that received municipal sewage between February 2021 and May 2021. Using the quantitative PCR assay targeting the E and RdRp genes of SARS-CoV-2, we confirmed that both genes were detected in the raw sewage, while no viral RNA was found in the treated sewage. We were also able to show that the trend of COVID-19 cases in Kuala Lumpur and Selangor was related to the changes in SARS-CoV-2 RNA levels in the wastewater samples. Overall, our study highlights that monitoring wastewater for SARS-CoV-2 should help local health professionals to obtain additional information on the rapid and silent circulation of infectious agents in communities at the regional level.
Similar content being viewed by others
Introduction
Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), a causative agent of Coronavirus Disease 2019 (COVID-19), remains to impact public health and the global economy. The virus was originally discovered in Wuhan, China, in late December 2019 (Chen et al. 2020a, b; Zhou et al. 2020) and has now spread rapidly to all continents and all parts of the world. The recent SARS-CoV-2 is genetically distant from the previous coronaviruses, Middle East Respiratory Syndrome Coronavirus (MERS-CoV) and Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) (Song et al. 2019). This newly identified coronavirus is known to be transmitted through direct contact, contaminated fomites and aerosolised respiratory fluids (Peng et al. 2020; Zaid et al. 2020). In addition, SARS-CoV-2 has also been found in the faeces of COVID-19 patients (Wang et al. 2020a, b), indicating possible faecal-oral transmission. This finding is an interesting thought that wastewater could serve as a platform to determine the ongoing circulation of SARS-CoV-2 in the community. Indeed, the occurrence of SARS-CoV-2 RNA in sewage has been described in many countries including Australia (Ahmed et al. 2020), The Netherlands (Medema et al. 2020), United States (Gonzalez et al. 2020; Nemudryi et al. 2020; Sherchan et al. 2020), France (Wurtzer et al. 2020), Germany (Westhaus et al. 2021), Iran (Tanhaei et al. 2021), Japan (Haramoto et al. 2020; Hata et al. 2021), United Arab Emirates (Hasan et al. 2021), Italy (la Rosa et al. 2020a, b), India (Chakraborty et al. 2021; Kumar et al. 2020), Pakistan (Sharif et al. 2021) and Singapore (Lee et al. 2021)However, at the time of writing this manuscript, there was no published study informing the identification of SARS-CoV-2 RNA in municipal wastewater in Malaysia.
Malaysia reported the first confirmed COVID-19 cases in January 2020 (Ahmad et al. 2020). Since then, the country reported a steady increase in cumulative COVID-19 cases, with more than 31,000 deaths by 5 January 2021 (World Health Organization 2021). In response to increasing concerns about new strains of the SARS-CoV-2 virus, stringent preventive actions have been taken to limit the spread of COVID -19 infections, including the introduction of Movement Control Orders (MCOs). Given the vagaries of the current pandemic and the effectiveness of the vaccination programme in reducing the infection rate in Malaysia, there is a need to predict the subsequent wave of COVID-19 infections in the population. Thus, wastewater surveillance can provide anonymous, non-invasive and large-scale population-level coverage within a given community, including asymptomatic individuals. This information can be used by public health officials to track the spread of this virus in the community and could serve as an early signal for future epidemics.
In this study, we aimed to find out whether SARS-CoV-2 RNA can be detected in Malaysian urban wastewater using quantitative polymerase chain reaction (qPCR), as reported by other developed countries. Thus, the objectives of our study are (1) to confirm the presence of SARS-CoV-2 RNA in influent (untreated) and effluent (treated) urban wastewater in Malaysia, (2) to quantify the total amount of SARS-CoV-2 genes found in wastewater and (3) to determine whether the number of daily COVID-19 cases correlates with the concentration of SARS-CoV-2 RNA in wastewater.
Materials and methods
Wastewater sampling
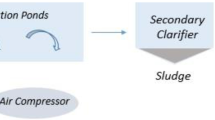
Three wastewater samples including influent (untreated), effluent (treated) and returned activated sludge were collected from municipal wastewater treatment plant with capacity of 1.432 million inhabitants (Fig. 1). A total of 500 mL of wastewater samples were obtained between 14 h February 2021 until 20th May 2021. Necessary precautions and personal protective equipment were used during sample collection. The samples were transported to the lab in a cold chain with information, indicating sampling sites, temperature, pH and sampling date. The sewage samples were processed immediately or kept – 20 ºC for short-term storage.
SARS-CoV-2 concentration using polyethylene glycol (PEG) precipitation approach
To minimize the risk of infection during sample processing, the wastewater samples were heat-inactivated at 60 ºC for 90 min prior to virus concentration. Previous reports indicated that heat pasteurisation at 60 ºC for 30 min was sufficient to inactivate SARS-CoV-1 (Rabenau et al. 2005) and SARS-CoV-2 (Saththasivam et al. 2021; Wu et al. 2020a, b) in wastewater samples without affecting viral RNA integrity. The inactivated water samples were centrifuged at 5,000xg for 15 min to discard any debris. The viral RNA in the wastewater was concentrated using the PEG precipitation method as previously described (Hata et al. 2021). The PEG precipitation method was selected in this study as it has been demonstrated to be highly efficient in the recovery of indigenous F-phage (Hata et al. 2021) and RNA virus (Amdiouni et al. 2012) in wastewater. In comparison to the ultrafiltration-based approach, the PEG precipitation method is more cost-effective for recovering SARS-CoV-2 from low-volume wastewater samples, especially due to the limited research funding. Briefly, 75 ml of wastewater was mixed with 25 ml of sodium chloride (NaCl) and PEG 8000 at a final concentration of 0.6 M and 10% (w/v), respectively. Subsequently, the PEG-treated wastewater samples were incubated overnight at 4 ºC on a shaker followed by centrifugation at 10,000×g for 2 h. The viral precipitate was resuspended in 200 µl of phosphate-buffered saline (PBS) (Calbiochem, USA) and used as virus concentrate for RNA extraction.
RNA isolation and reverse transcription quantitative PCR (RT-qPCR)
Viral RNA was extracted from 140 µl of the virus concentrates using the QIAmp Viral extraction kit (Qiagen) according to the manufacturer’s protocol. Sixty microliters of elution buffer were used to recover the RNA. Two primers and probes targeting RNA dependent RNA polymerase (RdRp) and envelope (E) were employed to detect the presence of SARS-CoV-2 RNA in wastewater as previously described (Corman et al. 2020). RT-qPCR was carried out for both targets using the StepOnePlus™ qPCR machine (Applied Biosystems) in a total reaction volume of 10 µl containing 2 µl of 5X RapiDxFire™ qPCR Master Mix (LGC Bioresearch Technologies, Lucigen), 0.25 µl of EpiScript™ RNase H- Reverse Transcriptase (LGC Bioresearch Technologies, Lucigen), 400 nM of E_Sarbeco primers (forward; 5′-ACAGGTACGTTAATAGTTAATAGCGT-3′ and reverse: 5′-ATATTGCAGCAGTACGCACACA-3′), 200 nM of E-gene probe (5′-FAM-ACACTAGCCATCCTTACTGCGCTTCG-BBQ-3′) or 600 nM of forward (5′-GTGARATGGTCATGTGTGGCGG-3′) and 800 nM reverse RdRp_SARSr primers (5′-CARATGTTAAASACACTATTAGCATA-3′) and 100 nM of RdRp probe specific to SARS-CoV-2 (5′-FAM-CAGGTGGAACCTCATCAGGAGATGC-BBQ-3′), 5 µl of RNA template and 1.25 of µl molecular grade H20. Thermocycler parameters were conducted at 55 ºC for 10 min for reverse transcription reaction, followed by 94 ºC for 3 min and 45 amplification cycles of 15 s at 94 ºC and 30 s at 58 ºC. Standard curves for E and RdRP were generated by serial dilutions of the positive control plasmid (PC), 2019-nCoV Positive Control v2 (MBS-4101, 1st BASE), at concentrations ranging from 100 to 100,000 copies per reaction to calculate the presence of E- and RdRp genes in wastewater samples (Nemudryi et al. 2020). Three technical replicates were performed, and molecular grade water was added as a negative control.
SARS-CoV-2 RNA quantification
The mean of at least two technical replicates was used to create the data in the figure and table in this study. The error bar represents the standard error of the mean (SEM). For each technical replicate, the predicted copy numbers in the qPCR reaction were used to calculate the copies per litre of wastewater (Nemudryi et al. 2020).
Results
Detection of SARS-CoV-2 RNA in influent wastewater samples
In this present study, we investigated the presence of two genes of SARS-CoV-2, envelope (E) and RNA dependent RNA polymerase (RdRp) in influent, return activated sludge (RAS), and effluent from municipal wastewater collected between February 2021 until May 2021. Preliminary screening was carried out using Envelope (E) gene specific to Sarbeco sub-genus, followed by confirmatory test using RNA dependent RNA polymerase (RdRp) gene which specific to SARS-CoV-2 (Corman et al. 2020). In this study, the wastewater sample was considered positive of SARS-CoV-2 if both genes were detected with Ct-value less than 40. Due to the travel restrictions and operational constraints during Movement Control Order (MCO), only a total of 18 wastewater samples were obtained in this study. Among 6 influent samples analysed in this study, only 4 (66%) were tested positive for both target genes except for sample collected on 6th May 2021. Crucially, none of RAS and effluent samples were found positive for any SARS-CoV-2 target genes (Table 1). This result may suggest that the current wastewater treatment process in Malaysia is efficient in removing this viral pathogen. However, the possibility of false positive results should also be considered. Therefore, both positive and negative template controls were integrated in the same qPCR run. For positive control, the Ct-values of E and RdRp gene were ranging from 23 to 35 while these genes were not identified in our negative template control, confirming the presence of SARS-CoV-2 in untreated municipal wastewater samples.
Based on our observation, we noticed that only the E gene was detected in the influent sample collected on 6th May 2021 but not RdRp gene following the same procedure (Table 1). This inconclusive result could be due to the relatively low sensitivity of the RdRp-based assay (Alagarasu et al. 2020). Indeed, it has been reported that the reverse primer of RdRp contains a degenerate base at position 12 which reduces the sensitivity of RdRp-based RT-qPCR assay in detecting viral genomics (Vogels et al. 2020). In addition, previous research has shown that the same RdRp assay has overlooked 35% of positive SARS-CoV-2 cases in comparison to another RdRp/helicase-based approach (Chan et al. 2020). Based on our observation, it is worth considering other SARS-CoV-2 genes such as nucleocapsid (N), open reading frame (ORF1ab) and spike (S) to complement the existing RdRp-based assay.
The monthly trend of SARS-CoV-2 concentrations in municipal sewage
The occurrence of SARS-CoV-2 in influent has been reported in many countries including the neighbouring country, Singapore (Wong et al. 2021). The quantification of SARS-CoV-2 genes in influent wastewater may have potential to facilitate the surveillance of unreported COVID-19 cases of asymptomatic and pre-symptomatic individuals. This will help to inform public health practitioners of possible outbreak in the given location and propose immediate action to contain the spread of virus in the community.
In this study, we showed that the estimated concentration of E gene in urban wastewater is ranging between 1.7 × 105—1.0 × 107 copies/L (Fig. 2a). To our knowledge, this study is the first available evidence uncovering the presence of SARS-CoV-2 genes in urban wastewater in Malaysia. The same E and RdRp-based assay have been employed for SARS-CoV-2 detection in France (Wurtzer et al. 2020). Although our protocol for virus concentration is different, we noticed that the estimated concentration of the SARS-CoV-2 E gene in our study is comparable with the study reported by Wurtzer and colleagues (Wurtzer et al. 2020). We noticed in our study that wastewater samples taken on 18th March 2021 and 20th May 2021 showed no presence of SARS-CoV-2 genes in all types of wastewaters (Table 1). The absence of SARS-CoV-2 RNA in wastewater might not necessarily imply the non-existence of viral RNA. A decrease in RNA concentration is predicted to occur when virus laden-human faeces are released into the sewage plant because of the dilution effect and other environmental factors such as temperature, pH and salinity of the wastewater as well as the presence of disinfectants and detergents (la Rosa et al. 2020a, b; Foladori et al. 2020; Giacobbo et al. 2021). Certainly, disinfectants are applied in large quantities beyond normal levels to reduce the number of COVID-19 infections, which may ultimately lead to possible negative impacts on the environment.
Detection of SARS-CoV-2 RNA in municipal wastewater in relation to COVID-19 cases reported in Kuala Lumpur (KL). a The concentration of SARS-CoV-2 RNA based on E gene-based RT-qPCR assay using influent samples. Error bars indicate standard error of the mean for at least two technical replicates. b The number of daily COVID-19 cases reported from the Ministry of Health Malaysia (Kementerian Kesihatan Malaysia 2022)
Quantification of viral RNA in municipal wastewater to mirror infections in the community.
The presence of viral RNA in sewage is widely used recently as a disease surveillance since viral genomics were discovered earlier before the first documented medical cases (Medema et al. 2020). In addition, because of sustained periods of viral shedding in faeces after negative indication of SARS-CoV-2 RNA in patient’s respiratory samples (Chen et al. 2020a, b; Wu et al. 2020a, b), the quantification of SARS-CoV-2 RNA in wastewater has been widely proposed as a good additional tool for monitoring virus circulation in a given population (Haramoto et al. 2020; Medema et al. 2020). In accordance with previous studies, we assume that the concentration of SARS-CoV-2 RNA in municipal sewage in Malaysia should correlate with the number of reported COVID-19 cases. Based on our quantitative analysis of SARS-CoV-2, a threefold decrease in estimated gene copy number was noticed between 4 and 18 February 2021, 5.4 × 105 copies/L and 1.8 × 105 copies/L which corresponds to a threefold less than the number of COVID-19 individuals on 4 and 18 February, respectively (Fig. 2b). However, between 18 February 2021 and 15 April 2021, we observed an increase in the estimated number of SARS-CoV-2 RNA in wastewater but a decrease in the number of reported COVID-19 cases for both areas in Kuala Lumpur and Selangor on the same dates (Fig. 2b). A similar observation has been reported in Massachusetts, USA by Wu and colleagues where they showed that the level of SARS-CoV-2 in wastewater is higher than the medically documented COVID-19 incidents (Wu et al. 2020a, b). Hence, this result may have suggested that the data from wastewater testing could be a good complement to the existing COVID-19 clinical diagnosis especially when the testing is unavailable or limited. Overall, our study further validates the potential use of wastewater as a reliable approach to track and diagnose viral circulation in a population.
Discussion
In this report, the existence of SARS-CoV-2 genes in municipal sewage in Malaysia was explored. Our results presented here contributed to the proof-of-concept study suggesting that SARS-CoV-2 RNA can be identified in influent wastewater and in agreement with previous studies demonstrating the presence of SARS-CoV-2 RNA in raw sewage (Kitajima et al. 2020). Whether SARS-CoV-2 RNA originating from sewage is viable and could facilitate faecal-oral transmission of COVID-19 is still not clear. However, several attempts to culture SARS-CoV-2-positive samples from domestic and hospital wastewater have been unsuccessful (Rimoldi et al. 2020; Wang et al. 2020a, b), suggesting that the risk of infection is insignificant. Furthermore, SARS-CoV-2 RNA was more persistent than infectious SARS-CoV-2 in wastewater (Bivins et al. 2020), indicating that environmental surveillance of SARS-CoV-2 RNA is unlikely to pose a threat to public health, although extra safety measures are recommended to limit the spread of COVID-19 infections.
Due to the economic reasons and practical limits of clinical screening in areas with inadequate infrastructures, public health professionals in many countries began to implement wastewater-based epidemiology (WBE) as wastewater constantly captures traces of faeces and sputum that contain SARS-CoV-2 virus shed by both asymptomatic and symptomatic individuals (Chen et al. 2020a, b; Gupta et al. 2020; Wang et al. 2020a, b). To continue living in the pandemic era, the Malaysian government gradually eases COVID-19 restrictions in all economic sectors to allow national economic recovery regardless of the continued risk of resurgence of COVID-19 cases. Given the new emergence of more virulent COVID-19 variants, it is crucial to continuously trace the presence of new and existing variants in the community as WBE should cover symptomatic as well as asymptomatic individuals who may be underestimated by medical surveillance. Therefore, the establishment of this study can help The Ministry of Health and other stakeholders to identify specific locations that require special attention and targeted interventions. Indeed, WBE has been successfully applied for identification of SARS-CoV-2 a week earlier before the first announcement of the COVID-19 cases (Medema et al. 2020; Randazzo et al. 2020). Furthermore, WBE has been successfully implemented to monitor the success of the vaccination programme for poliovirus (Mat Rabi et al. 2019). The similar approach can be applied to monitor the progress of ongoing national vaccination programmes.
Conclusion
While many countries have demonstrated the proof of WBE concept, Malaysia also can participate in proofing the concept of WBE and its applicability. The scope of this study was limited by the use of influent and effluent from a single treatment plant. For future work, several important features can be explored such as the presence of silent variant of concern (VOC) and the new emergence of virus strains circulating in the community. In addition, WBE can also be employed to study the association between the emergence of antibiotic resistance microorganisms and the COVID-19 crisis. Consequently, continuous monitoring of wastewater will ensure the safety of communities and populations, confirming the effectiveness of disinfection and mitigation measures of any harmful microorganisms in our water sources.
Data availability
Not applicable.
References
Ahmad NA, Lin CZ, Abd Rahman S, bin Ghazali MH, Nadzari EE, Zakiman Z, Redzuan S, Md Taib S, Kassim MSA, Wan Mohamed Noor WN (2020) First local transmission cluster of COVID-19 in Malaysia: public health response. Int J Travel Med Global Health 8(3):124–130. https://doi.org/10.34172/ijtmgh.2020.21
Ahmed W, Angel N, Edson J, Bibby K, Bivins A, O’Brien JW, Choi PM, Kitajima M, Simpson SL, Li J, Tscharke B, Verhagen R, Smith WJM, Zaugg J, Dierens L, Hugenholtz P, Thomas KV, Mueller JF (2020) First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: a proof of concept for the wastewater surveillance of COVID-19 in the community. Sci Total Environ 728:138764. https://doi.org/10.1016/j.scitotenv.2020.138764
Alagarasu K, Choudhary ML, Lole KS, Abraham P, Potdar V, Team, N (2020) Evaluation of RdRp & ORF-1b-nsp14-based real-time RT-PCR assays for confirmation of SARS-CoV-2 infection: an observational study. Indian J Med Res 151(5):483. https://doi.org/10.4103/IJMR.IJMR_1256_20
Amdiouni H, Maunula L, Hajjami K, Faouzi A, Soukri A, Nourlil J (2012) Recovery comparison of two virus concentration methods from wastewater using cell culture and real-time PCR. Curr Microbiol 65(4):432–437. https://doi.org/10.1007/S00284-012-0174-8/METRICS
Bivins A, Greaves J, Fischer R, Yinda KC, Ahmed W, Kitajima M, Munster VJ, Bibby K (2020) Persistence of SARS-CoV-2 in water and wastewater. Environ Sci Technol Lett. https://doi.org/10.1021/acs.estlett.0c00730
Chakraborty P, Pasupuleti M, JaiShankar MR, Bharat GK, Krishnasamy S, Dasgupta SC, Sarkar SK, Jones KC (2021) First surveillance of SARS-CoV-2 and organic tracers in community wastewater during post lockdown in Chennai, South India: Methods, occurrence and concurrence. Sci Total Environ 778:146252. https://doi.org/10.1016/J.SCITOTENV.2021.146252
Chan JFW, Yip CCY, To KKW, Tang THC, Wong SCY, Leung KH, Fung AYF, Ng ACK, Zou Z, Tsoi HW, Choi GKY, Tam AR, Cheng VCC, Chan KH, Tsang OTY, Yuen KY (2020) Improved molecular diagnosis of COVID-19 by the novel, highly sensitive and specific COVID-19-RdRp/Hel real-time reverse transcription-PCR assay validated in vitro and with clinical specimens. J Clin Microbiol. https://doi.org/10.1128/JCM.00310-20
Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L (2020a) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395(10223):507–513. https://doi.org/10.1016/S0140-6736(20)30211-7
Chen Y, Chen L, Deng Q, Zhang G, Wu K, Ni L, Yang Y, Liu B, Wang W, Wei C, Yang J, Ye G, Cheng Z (2020b) The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol 92(7):833–840. https://doi.org/10.1002/jmv.25825
Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DKW, Bleicker T, Brünink S, Schneider J, Schmidt ML, Mulders DGJC, Haagmans BL, Van Der Veer B, Van Den Brink S, Wijsman L, Goderski G, Romette JL, Ellis J, Zambon M et al (2020) Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance. https://doi.org/10.2807/1560-7917.ES.2020.25.3.2000045
Foladori P, Cutrupi F, Segata N, Manara S, Pinto F, Malpei F, Bruni L, la Rosa G (2020) SARS-CoV-2 from faeces to wastewater treatment: what do we know? A review. Sci Total Environ 743:140444. https://doi.org/10.1016/J.SCITOTENV.2020.140444
Giacobbo A, Rodrigues MAS, Zoppas Ferreira J, Bernardes AM, de Pinho MN (2021) A critical review on SARS-CoV-2 infectivity in water and wastewater. What do we know? Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2021.145721
Gonzalez R, Curtis K, Bivins A, Bibby K, Weir MH, Yetka K, Thompson H, Keeling D, Mitchell J, Gonzalez D (2020) COVID-19 surveillance in Southeastern Virginia using wastewater-based epidemiology. Water Research 186:116296. https://doi.org/10.1016/J.WATRES.2020.116296
Gupta S, Parker J, Smits S, Underwood J, Dolwani S (2020) Persistent viral shedding of SARS-CoV-2 in faeces—a rapid review. Colorectal Dis 22(6):611–620. https://doi.org/10.1111/codi.15138
Haramoto E, Malla B, Thakali O, Kitajima M (2020) First environmental surveillance for the presence of SARS-CoV-2 RNA in wastewater and river water in Japan. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.140405
Hasan SW, Ibrahim Y, Daou M, Kannout H, Jan N, Lopes A, Alsafar H, Yousef AF (2021) Detection and quantification of SARS-CoV-2 RNA in wastewater and treated effluents: surveillance of COVID-19 epidemic in the United Arab Emirates. Sci Total Environ 764:142929. https://doi.org/10.1016/j.scitotenv.2020.142929
Hata A, Hara-Yamamura H, Meuchi Y, Imai S, Honda R (2021) Detection of SARS-CoV-2 in wastewater in Japan during a COVID-19 outbreak. Sci Total Environ 758:143578. https://doi.org/10.1016/J.SCITOTENV.2020.143578
Kementerian Kesihatan Malaysia (2022) Home | COVID-19 MALAYSIA. http://covid-19.moh.gov.my/
Kitajima M, Ahmed W, Bibby K, Carducci A, Gerba CP, Hamilton KA, Haramoto E, Rose JB (2020) SARS-CoV-2 in wastewater: state of the knowledge and research needs. Sci Total Environ 739:139076. https://doi.org/10.1016/j.scitotenv.2020.139076
Kumar M, Patel AK, Shah AV, Raval J, Rajpara N, Joshi M, Joshi CG (2020) First proof of the capability of wastewater surveillance for COVID-19 in India through detection of genetic material of SARS-CoV-2. Sci Total Environ 746:141326. https://doi.org/10.1016/J.SCITOTENV.2020.141326
La Rosa G, Bonadonna L, Lucentini L, Kenmoe S, Suffredini E (2020) Coronavirus in water environments: occurrence, persistence and concentration methods—a scoping review. Water Res 179:115899. https://doi.org/10.1016/J.WATRES.2020.115899
La Rosa G, Iaconelli M, Mancini P, Ferraro GB, Veneri C, Bonadonna L, Lucentini L, Suffredini E (2020) First detection of SARS-CoV-2 in untreated wastewaters in Italy. Sci Total Environ 736:139652. https://doi.org/10.1016/j.scitotenv.2020.139652
Lee WL, Imakaev M, Armas F, McElroy KA, Gu X, Duvallet C, Chandra F, Chen H, Leifels M, Mendola S, Floyd-O’Sullivan R, Powell MM, Wilson ST, Berge KLJ, Lim CYJ, Wu F, Xiao A, Moniz K, Ghaeli N et al (2021) Quantitative SARS-CoV-2 alpha variant B.1.1.7 tracking in wastewater by Allele-Specific RT-qPCR. Environ Sci Technol Lett 8(8):675–682. https://doi.org/10.1021/ACS.ESTLETT.1C00375
Mat Rabi KH, Ismail AK, Samsuddin MS, Abdul Wahid MZ, Mohd Zamawi Z, Thayan R (2019) Milestone of polio environmental surveillance in Malaysia conjoining with the polio global eradication initiative. MyJurnal 15(6):78–78. https://myjurnal.mohe.gov.my/public/article-view.php?id=147831
Medema G, Heijnen L, Elsinga G, Italiaander R, Brouwer A (2020) Presence of SARS-coronavirus-2 RNA in sewage and correlation with reported COVID-19 prevalence in the early stage of the epidemic in The Netherlands. Environ Sci Technol Lett. https://doi.org/10.1021/acs.estlett.0c00357
Nemudryi A, Nemudraia A, Wiegand T, Surya K, Buyukyoruk M, Cicha C, Vanderwood KK, Wilkinson R, Wiedenheft B (2020) Temporal detection and phylogenetic assessment of SARS-CoV-2 in municipal wastewater. Cell Rep Med 1(6):100098. https://doi.org/10.1016/j.xcrm.2020.100098
Peng X, Xu X, Li Y, Cheng L, Zhou X, Ren B (2020) Transmission routes of 2019-nCoV and controls in dental practice. Int J Oral Sci 12(1):9. https://doi.org/10.1038/s41368-020-0075-9
Rabenau HF, Cinatl J, Morgenstern B, Bauer G, Preiser W, Doerr HW (2005) Stability and inactivation of SARS coronavirus. Med Microbiol Immunol 194(1–2):1–6. https://doi.org/10.1007/s00430-004-0219-0
Randazzo W, Truchado P, Cuevas-Ferrando E, Simón P, Allende A, Sánchez G (2020) SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res 181:115942. https://doi.org/10.1016/j.watres.2020.115942
Rimoldi SG, Stefani F, Gigantiello A, Polesello S, Comandatore F, Mileto D, Maresca M, Longobardi C, Mancon A, Romeri F, Pagani C, Moja L, Gismondo MR, Salerno F (2020) Presence and vitality of SARS-CoV-2 virus in wastewaters and rivers. MedRxiv. https://doi.org/10.1101/2020.05.01.20086009
Saththasivam J, El-Malah SS, Gomez TA, Jabbar KA, Remanan R, Krishnankutty AK, Ogunbiyi O, Rasool K, Ashhab S, Rashkeev S, Bensaad M, Ahmed AA, Mohamoud YA, Malek JA, Abu Raddad LJ, Jeremijenko A, Abu Halaweh HA, Lawler J, Mahmoud KA (2021) COVID-19 (SARS-CoV-2) outbreak monitoring using wastewater-based epidemiology in Qatar. Sci Total Environ 774:145608. https://doi.org/10.1016/J.SCITOTENV.2021.145608
Sharif S, Ikram A, Khurshid A, Salman M, Mehmood N, Arshad Y, Ahmed J, Safdar RM, Rehman L, Mujtaba G, Hussain J, Ali J, Angez M, Alam MM, Akthar R, Malik MW, Baig MZI, Rana MS, Usman M et al (2021) Detection of SARs-CoV-2 in wastewater using the existing environmental surveillance network: a potential supplementary system for monitoring COVID-19 transmission. PLOS One 16(6):e0249568. https://doi.org/10.1371/JOURNAL.PONE.0249568
Sherchan SP, Shahin S, Ward LM, Tandukar S, Aw TG, Schmitz B, Ahmed W, Kitajima M (2020) First detection of SARS-CoV-2 RNA in wastewater in North America: a study in Louisiana, USA. Sci Total Environ 743:140621. https://doi.org/10.1016/J.SCITOTENV.2020.140621
Song Z, Xu Y, Bao L, Zhang L, Yu P, Qu Y, Zhu H, Zhao W, Han Y, Qin C (2019) From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses. https://doi.org/10.3390/v11010059
Tanhaei M, Mohebbi SR, Hosseini SM, Rafieepoor M, Kazemian S, Ghaemi A, Shamloei S, Mirjalali H, Asadzadeh Aghdaei H, Zali MR (2021) The first detection of SARS-CoV-2 RNA in the wastewater of Tehran, Iran. Environ Sci Pollut Res 28(29):38629–38636. https://doi.org/10.1007/s11356-021-13393-9
Vogels CBF, Brito AF, Wyllie AL, Fauver JR, Ott IM, Kalinich CC, Petrone ME, Casanovas-Massana A, Catherine Muenker M, Moore AJ, Klein J, Lu P, Lu-Culligan A, Jiang X, Kim DJ, Kudo E, Mao T, Moriyama M, Oh JE, Grubaugh ND (2020) Analytical sensitivity and efficiency comparisons of SARS-CoV-2 RT–qPCR primer–probe sets. Nat Microbiol 5(10):1299–1305. https://doi.org/10.1038/s41564-020-0761-6
Wang J, Feng H, Zhang S, Ni Z, Ni L, Chen Y, Zhuo L, Zhong Z, Qu T (2020a) SARS-CoV-2 RNA detection of hospital isolation wards hygiene monitoring during the Coronavirus Disease 2019 outbreak in a Chinese hospital. Int J Infect Dis 94:103–106. https://doi.org/10.1016/J.IJID.2020.04.024
Wang W, Xu Y, Gao R, Lu R, Han K, Wu G, Tan W (2020) Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. https://doi.org/10.1001/jama.2020.3786
Westhaus S, Weber FA, Schiwy S, Linnemann V, Brinkmann M, Widera M, Greve C, Janke A, Hollert H, Wintgens T, Ciesek S (2021) Detection of SARS-CoV-2 in raw and treated wastewater in Germany—suitability for COVID-19 surveillance and potential transmission risks. Sci Total Environ 751:141750. https://doi.org/10.1016/J.SCITOTENV.2020.141750
Wong JCC, Tan J, Lim YX, Arivalan S, Hapuarachchi HC, Mailepessov D, Griffiths J, Jayarajah P, Setoh YX, Tien WP, Low SL, Koo C, Yenamandra SP, Kong M, Lee VJM, Ng LC (2021) Non-intrusive wastewater surveillance for monitoring of a residential building for COVID-19 cases. Sci Total Environ 786:147419. https://doi.org/10.1016/J.SCITOTENV.2021.147419
World Health Organization (2021) Malaysia: WHO Coronavirus Disease (COVID-19) Dashboard With Vaccination Data | WHO Coronavirus (COVID-19) Dashboard With Vaccination Data. World Health Organization. https://covid19.who.int/%0Ahttps://covid19.who.int/%0Ahttps://covid19.who.int/%0Ahttps://covid19.who.int/region/searo/country/bd
Wu F, Zhang J, Xiao A, Gu X, Lee WL, Armas F, Kauffman K, Hanage W, Matus M, Ghaeli N, Endo N, Duvallet C, Poyet M, Moniz K, Washburne AD, Erickson TB, Chai PR, Thompson J, Alm EJ (2020) SARS-CoV-2 titers in wastewater are higher than expected from clinically confirmed cases. MSystems. https://doi.org/10.1128/MSYSTEMS.00614-20
Wu Y, Guo C, Tang L, Hong Z, Zhou J, Dong X, Yin H, Xiao Q, Tang Y, Qu X, Kuang L, Fang X, Mishra N, Lu J, Shan H, Jiang G, Huang X (2020b) Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol 5(5):434–435. https://doi.org/10.1016/S2468-1253(20)30083-2
Wurtzer S, Marechal V, Mouchel JM, Maday Y, Teyssou R, Richard E, Almayrac JL, Moulin L (2020) Evaluation of lockdown effect on SARS-CoV-2 dynamics through viral genome quantification in wastewater. Euro Surveill. https://doi.org/10.2807/1560-7917
Zaid S, Afiq Aziz M, Sulaiman R, Simarani K, Suyoto YT, Lou E, Esfandiari M (2020) Review of Coronavirus transmission in urban clusters: survival in water and wastewater systems. J Design Built Environ 20(3):85–102
Zhou P, Yang X-L, Wang X-G, Hu B, Zhang L, Zhang W, Si H-R, Zhu Y, Li B, Huang C-L, Chen H-D, Chen J, Luo Y, Guo H, Jiang R-D, Liu M-Q, Chen Y, Shen X-R, Wang X et al (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579(7798):270–273. https://doi.org/10.1038/s41586-020-2012-7
Acknowledgements
The authors would like to thank Mohd Ikmal Shafiq Bin Rosli for his assistance in figure preparation, Institute for Medical Research, and Indah Water Konsortium for the technical assistance. This project was funded by COVID-19 Special Research Grant (CSRG007-2020ST), Universiti Malaya.
Funding
This work was supported by COVID-19 Special Research Grant (CSRG007-2020ST), Universiti Malaya.
Author information
Authors and Affiliations
Contributions
MAA: formal analysis, writing—original draft, writing—review and editing. SN: formal analysis, writing—review and editing. KS: writing—review & editing. RS: writing—review and editing. AMA: writing—review and editing. KBC: writing—review and editing. RMZ: writing—review and editing. SMZ: writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no known competing personal relationships or financial interests that could have influenced the work in this manuscript.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have read and permitted this manuscript.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aziz, M.A., Norman, S., Mohamed Zaid, S. et al. Environmental surveillance of SARS-CoV-2 in municipal wastewater to monitor COVID-19 status in urban clusters in Malaysia. Arch Microbiol 205, 76 (2023). https://doi.org/10.1007/s00203-023-03417-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03417-y