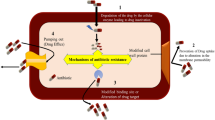
Abstract
Staphylococcus xylosus is a gram-positive bacterium that has attracted much attention due to its increasing clinical appearance, frequently associated with serious multidrug resistance cases. L-lactate dehydrogenase (LDH) has been related to drug resistance in several bacterial species. However, the mechanism of multidrug resistance in S. xylosus remains unclear as well as the involvement of LDH in such resistance. To explore the relationship between multidrug resistance and LDH in S. xylosus, we used tylosin-resistant S. xylosus as the parent strain to construct ldh knockout and complemented strains. Then, we tested their resistance to macrolides, lincosamides, tetracyclines, and aminoglycosides. In addition, the enzyme activity, metabolite content, and transcriptional level of key genes involved in the TCA cycle and thioredoxin system were determined to clarify the mechanism of resistance. We observed that the resistance to multiple antibiotics increased significantly after ldh knockout, especially that to lincomycin, whereas antibiotic sensitivity was partially restored in the complemented strain. The levels of pyruvate, nicotinamide adenine dinucleotide, and reactive oxygen species decreased significantly upon ldh knockout, and the activity of isocitrate dehydrogenase and malate dehydrogenase decreased. These results indicate that the lack of LDH promotes multidrug resistance in S. xylosus by inhibiting the TCA cycle and regulating the thioredoxin system.





Similar content being viewed by others
Availability of data and material
All data in the current study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Acuff NV, LaGatta M, Nagy T et al (2017) Severe Dermatitis Associated with Spontaneous Staphylococcus xylosus Infection in Rag–/–Tpl2–/–Mice. Comp Med 67:344–349
Akhaddar A, Elouennass M, Naama O et al (2010) Staphylococcus xylosus isolated from an otogenic brain abscess in an adolescent. Surg Infect 11:559–561. https://doi.org/10.1089/sur.2010.010
Allison KR, Brynildsen MP, Collins JJ (2011) Metabolite-enabled eradication of bacterial persisters by aminoglycosides. Nature 473:216–220. https://doi.org/10.1038/nature10069
Bernat GA, Roig RP, Sanchéz AJ et al (1992) Acute endocarditis due to Staphylococcus xylosus. Rev Clin Esp 190:213–214
Bochniarz M, Wawron W, Szczubial M (2013) Coagulase-negative staphylococci (CNS) as an aetiological factor of mastitis in cows. Pol J Vet Sci 16:487–492. https://doi.org/10.2478/pjvs-2013-0068
Bradfield J (1993) Epizootic fatal dermatitis in athymic nude mice due to Staphylococcus xylosus. Lab Anim Sci 43:111–113
Brisson-Noël A, Trieu-Cuot P, Courvalin P (1988) Mechanism of action of spiramycin and other macrolides. J Antimicrob Chemoth 22:13–23. https://doi.org/10.1093/jac
Bruckner R (1997) Gene replacement in Staphylococcus carnosus and Staphylococcus xylosus. Fems Microbiol Lett 151:1–8. https://doi.org/10.1111/j.1574-6968.1997.tb10387.x
Cheah SE, Johnson MD, Zhu Y et al (2016) Polymyxin resistance in Acinetobacter baumannii: genetic mutations and transcriptomic changes in response to clinically relevant dosage regimens. Sci Rep 6:1–11. https://doi.org/10.1038/srep26233
De Visscher A, Piepers S, Haesebrouck F et al (2017) Coagulase-negative Staphylococcus species in bulk milk: prevalence, distribution, and associated subgroup-and species-specific risk factors. J Dairy Sci 100:629–642. https://doi.org/10.3168/jds.2016-11476
Dharmaraja AT (2017) Role of reactive oxygen species (ROS) in therapeutics and drug resistance in cancer and bacteria. J Med Chem 60:3221–3240. https://doi.org/10.1021/acs.jmedchem.6b01243
Duarte CM, Freitas PP, Bexiga R (2015) Technological advances in bovine mastitis diagnosis: an overview. J Vet Diagn Invest 27:665–672. https://doi.org/10.1177/1040638715603087
Ferreira MT, Manso AS, Gaspar P et al (2013) Effect of oxygen on glucose metabolism: utilization of lactate in Staphylococcus aureus as revealed by in vivo NMR studies. PLoS ONE 8:e58277. https://doi.org/10.1371/journal.pone.0058277
Fijalkowski K, Struk M, Karakulska J et al (2014) Comparative analysis of superantigen genes in Staphylococcus xylosus and Staphylococcus aureus isolates collected from a single mammary quarter of cows with mastitis. J Microbiol 52:366–372. https://doi.org/10.1007/s12275-014-3436-2
Frey Y, Rodriguez JP, Thomann A et al (2013) Genetic characterization of antimicrobial resistance in coagulase-negative staphylococci from bovine mastitis milk. J Dairy Sci 96:2247–2257. https://doi.org/10.3168/jds.2019-17028
Fthenakis G, Marples R, Richardson J et al (1994) Some properties of coagulase–negative Staphylococci isolated from cases of ovine mastitis. Epidemiol Infect 112:171–176. https://doi.org/10.1017/s0950268800057538
Gales AC, Jones RN (2002) Respiratory tract pathogens isolated from patients hospitalized with suspected pneumonia in Latin America: frequency of occurrence and antimicrobial susceptibility profile: results from the SENTRY Antimicrobial Surveillance Program (1997–2000). Diagn Micr Infec Dis 44:301–311. https://doi.org/10.1016/s0732-8893(02)00499-6
Griffiths EJ, Rutter GA (2009) Mitochondrial calcium as a key regulator of mitochondrial ATP production in mammalian cells. Biochim Biophys Acta 1787:1324–1333. https://doi.org/10.1016/j.bbabio.2009.01.019
Han J, Shi W, Xu X et al (2015) Conditions and mutations affecting Staphylococcus aureus L-form formation. Microbiology 161:57–66. https://doi.org/10.1099/mic.0.082354-0
Hricak V, Kovacik J, Marks P et al (1999) Aetiology and outcome in 53 cases of native valve staphylococcal endocarditis. Postgrad Med J 75:540–543. https://doi.org/10.1136/pgmj.75.887.540
Jeong OM, Kang MS, Blackall PJ et al (2017) Isolation and characterization of Avibacterium paragallinarum with different nicotinamide adenine dinucleotide requirements. Vet Microbiol 205:62–65. https://doi.org/10.1016/j.vetmic.2017.05.005
Klibi A, Maaroufi A, Torres C et al (2018) Detection and characterization of methicillin-resistant and susceptible coagulase-negative staphylococci in milk from cows with clinical mastitis in Tunisia. Int J Antimicrob Ag 52:930–935. https://doi.org/10.1016/j.ijantimicag.2018.07.026
Kohanski MA, Dwyer DJ, Hayete B et al (2007) A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130:797–810. https://doi.org/10.1016/j.cell.2007.06.049
Kohanski MA, DePristo MA, Collins JJ (2010) Sublethal antibiotic treatment leads to multidrug resistance via radical-induced mutagenesis. Mol Cell 37:311–320. https://doi.org/10.1016/j.molcel.2010.01.003
Koksal F, Yasar H, Samasti M (2009) Antibiotic resistance patterns of coagulase-negative staphylococcus strains isolated from blood cultures of septicemic patients in Turkey. Microbiol Res 164:404–410. https://doi.org/10.1016/j.micres.2007.03.004
Lee D-S, Park J-S, Kim Y et al (2014) Corynebacterium glutamicum sdhA encoding succinate dehydrogenase subunit A plays a role in cysR-mediated sulfur metabolism. Appl Microbiol Biot 98:6751–6759. https://doi.org/10.1007/s00253-014-5823-3
Liu X, Wang J, Chen M et al (2019) Comparative proteomic analysis reveals drug resistance of Staphylococcus xylosus ATCC700404 under tylosin stress. BMC Vet Res 15:224. https://doi.org/10.1186/s12917-019-1959-9
Ma L, Mi H, Xue Y et al (2016) The mechanism of ROS regulation of antibiotic resistance and antimicrobial lethality. Yi Chuan 38:902–909. https://doi.org/10.16288/j.yczz.16-157
Mai B, Wang X, Liu Q et al (2016) The antibacterial effect of sinoporphyrin sodium photodynamic therapy on Staphylococcus aureus planktonic and biofilm cultures. Laser Surg Med 48:400–408. https://doi.org/10.1002/lsm.22468
Mai B, Gao Y, Li M et al (2017) Photodynamic antimicrobial chemotherapy for Staphylococcus aureus and multidrug-resistant bacterial burn infection in vitro and in vivo. Int J Nanomed 12:5915. https://doi.org/10.2147/IJN.S138185
Miedzobrodzki J, Naidu A, Watts J et al (1989) Effect of milk on fibronectin and collagen type I binding to Staphylococcus aureus and coagulase-negative staphylococci isolated from bovine mastitis. J Clin Microbiol 27:540–544. https://doi.org/10.1128/jcm.27.3.540-544.1989
Milewski WM, Boyle-Vavra S, Moreira B et al (1996) Overproduction of a 37-kilodalton cytoplasmic protein homologous to NAD+-linked D-lactate dehydrogenase associated with vancomycin resistance in Staphylococcus aureus. Antimicrob Agents Ch 40:166–172. https://doi.org/10.1128/AAC.40.1.166
Mizumachi T, Suzuki S, Naito A et al (2008) Increased mitochondrial DNA induces acquired docetaxel resistance in head and neck cancer cells. Oncogene 27:831–838. https://doi.org/10.1038/sj.onc.1210681
Mohammadi H, Babaie F, Hemmatzadeh M et al (2018) Evaluation of ERAP1 gene single nucleotide polymorphism in impressing the inflammatory cytokine profile of ankylosing spondylitis patients. Iran J Allergy Asthm 17:464–474
Muralikrishnan G, Amanullah S, Basha MI et al (2010) Modulating effect of Withania somnifera on TCA cycle enzymes and electron transport chain in azoxymethane-induced colon cancer in mice. Immunopharm Immunot 32:523–527. https://doi.org/10.3109/08923970903581540
Pan J-S, Hong M-Z, Ren J-L (2009) Reactive oxygen species: a double-edged sword in oncogenesis. Word J Gastroenterol 15:1702. https://doi.org/10.3748/wjg.15.1702
Pinna A, Zanetti S, Sotgiu M et al (1999) Identification and antibiotic susceptibility of coagulase negative staphylococci isolated in corneal/external infections. Brit J Ophthalmol 83:771–773. https://doi.org/10.1136/bjo.83.7.771
Rezende C, Souza L, Almeida M et al (2012) Optimisation and validation of a quantitative and confirmatory method for residues of macrolide antibiotics and lincomycin in kidney by liquid chromatography coupled to mass spectrometry. Food Addit Contam A 29:587–595. https://doi.org/10.1080/19440049.2011.652196
Rissi DR, Elsmo EJ, Sanchez S (2015) Cystitis and peritonitis caused by Staphylococcus xylosus infection in a calf. Braz J of Vet Pathol 8:99–101
Ruegg PL (2018) Making antibiotic treatment decisions for clinical mastitis. Vet Clin N Am-Food A 34:413–425. https://doi.org/10.1016/j.cvfa.2018.06.002
Sampimon O, Lam T, Mevius D et al (2011) Antimicrobial susceptibility of coagulase-negative staphylococci isolated from bovine milk samples. Vet Microbiol 50:173–179. https://doi.org/10.1016/j.vetmic.2011.01.017
Siqueira J Jr, Lima K (2002) Staphylococcus epidermidis and Staphylococcus xylosus in a secondary root canal infection with persistent symptoms: a case report. Aust Endod J 28:61–63. https://doi.org/10.1111/j.1747-4477.2002.tb00382.x
Slaughter DM, Patton TG, Sievert G et al (2001) Antibiotic resistance in coagulase-negative staphylococci isolated from Cope’s gray treefrogs (Hyla chrysoscelis). Fems Microbiol Lett 205:265–270. https://doi.org/10.1111/j.1574-6968.2001.tb10959.x
Solomon H, Dixon D, Pouch W (1990) A survey of staphylococci isolated from the laboratory gerbil. Lab Anim Sci 40:316–318
Somerville GA, Proctor RA (2009) At the crossroads of bacterial metabolism and virulence factor synthesis in Staphylococci. Microbiol Mol Biol R 73:233–248. https://doi.org/10.1126/scitranslmed.aaf7444
Stafford WC, Peng X, Olofsson MH et al (2018) Irreversible inhibition of cytosolic thioredoxin reductase 1 as a mechanistic basis for anticancer therapy. Sci Transl Med 10:eaaf7444. https://doi.org/10.1126/scitranslmed.aaf7444
Su YB, Peng B, Li H et al (2018) Pyruvate cycle increases aminoglycoside efficacy and provides respiratory energy in bacteria. P Natl Acad Sci USA 115:E1578–E1587. https://doi.org/10.1073/pnas.1714645115
Sun L, Chen H, Lin W et al (2017) Quantitative proteomic analysis of Edwardsiella tarda in response to oxytetracycline stress in biofilm. J Proteomics 150:141–148. https://doi.org/10.1016/j.jprot.2016.09.006
Thomas VC, Kinkead LC, Janssen A et al (2013) A dysfunctional tricarboxylic acid cycle enhances fitness of Staphylococcus epidermidis during β-lactam stress. Mbio 4:e00437-e1413. https://doi.org/10.1128/mBio.00437-13
Tselenis-Kotsowilis AD, Koliomichalis MP, Papavassiliou JT (1982) Acute pyelonephritis caused by Staphylococcus xylosus. J Clin Microbiol 16:593–594. https://doi.org/10.1128/jcm.16.3.593-594.1982
Wang Y, Bojer MS, George SE et al (2018) Inactivation of TCA cycle enhances Staphylococcus aureus persister cell formation in stationary phase. Sci Rep 8:1–13. https://doi.org/10.1038/s41598-018-29123-0
Won YS, Kwon HJ, Oh GT et al (2002) Identification of Staphylococcus xylosus Isolated from C57BL/6J-Nos2tm1Lau Mice with Dermatitis. Microbiol Immunol 46:629–632. https://doi.org/10.1111/j.1348-0421.2002.tb02744.x
Xu CG, Yang YB, Zhou YH et al (2017) Comparative proteomic analysis provides insight into the key proteins as possible targets involved in aspirin inhibiting biofilm formation of Staphylococcus xylosus. Front Microbiol 8:543. https://doi.org/10.3389/fphar.2017.00543
Yang YB, Chen JQ, Zhao YL et al (2016) Sub-MICs of azithromycin decrease biofilm formation of Streptococcus suis and increase capsular polysaccharide content of S. suis. Front Microbiol 7:1659. https://doi.org/10.3389/fmicb.2016.01659
Ye S-f, Yang Y, Wu L et al (2017) Ethaselen: a novel organoselenium anticancer agent targeting thioredoxin reductase 1 reverses cisplatin resistance in drug-resistant K562 cells by inducing apoptosis. J Zhejiang Univ Sci B 18:373–382. https://doi.org/10.1631/jzus.B1600073
Yeswanth S, Kumar YN, Prasad UV et al (2013) Cloning and characterization of L-lactate dehydrogenase gene of Staphylococcus aureus. Anaerobe 24:43–48. https://doi.org/10.1016/j.anaerobe.2013.09.003
Yuan Z, Ouyang P, Gu K et al (2019) The antibacterial mechanism of oridonin against methicillin-resistant Staphylococcus aureus (MRSA). Pharm Biol 57:710–716. https://doi.org/10.1080/13880209.2019.1674342
Yuan Z, Dai Y, Ouyang P et al (2020) Thymol inhibits biofilm formation, eliminates pre-existing biofilms, and enhances clearance of methicillin-resistant Staphylococcus aureus (MRSA) in a mouse peritoneal implant infection model. Microorganisms 8:99. https://doi.org/10.3390/microorganisms8010099
Zalis EA, Nuxoll AS, Manuse S et al (2019) Stochastic variation in expression of the tricarboxylic acid cycle produces persister cells. Mbio 10:e01930-e11919. https://doi.org/10.1128/mBio.01930-19
Zheng Y, Liu W, Qin Z et al (2018) Mercaptopyrimidine-conjugated gold nanoclusters as nanoantibiotics for combating multidrug-resistant superbugs. Bioconjugate Chem 29:3094–3103. https://doi.org/10.1021/acs.bioconjchem.8b00452
Zhou YH, Xu CG, Yang YB et al (2018) Histidine metabolism and IGPD play a key role in cefquinome inhibiting biofilm formation of Staphylococcus xylosus. Front Microbiol 9:665. https://doi.org/10.3389/fmicb.2018.00665
Zhu Y, Xiong YQ, Sadykov MR et al (2009) Tricarboxylic acid cycle-dependent attenuation of Staphylococcus aureus in vivo virulence by selective inhibition of amino acid transport. Infect Immun 77:4256–4264. https://doi.org/10.1128/IAI.00195-09
Acknowledgements
We thank Prof. Rui Zhou (Huazhong Agricultural University) for the generous donation of the pBT2 vector used in the course of this study. We would like to thank Editage (www.editage.cn) for English language editing.
Funding
The study was funded by the National Natural Science Foundation of China (Grant numbers. 31772787 and 3190190314) and the “Young Talents” Project of Northeast Agricultural University (19QC24).
Author information
Authors and Affiliations
Contributions
YL and XL conceived and designed the experiments; JW, YZ, CD, LL, and MC performed the experiments; ZY analyzed the data and wrote the manuscript; GB-O, RC, EN, and ZY revised the discussion and proofread the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, Z., Wang, J., Che, R. et al. Relationship between L-lactate dehydrogenase and multidrug resistance in Staphylococcus xylosus. Arch Microbiol 204, 91 (2022). https://doi.org/10.1007/s00203-021-02625-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02625-8