Abstract
Summary
We aimed to study the mechanisms involved in bone-related iron impairment by using the osteoblast-like MG-63 cell line. Our results indicate that iron impact the S1P/S1PR signalizing axis and suggest that iron can affect the S1P process and favor the occurrence of osteoporosis during chronic iron overload.
Introduction
Systemic iron excess favors the development of osteoporosis, especially during genetic hemochromatosis. The cellular mechanisms involved are still unclear despite numerous data supporting a direct effect of iron on bone biology. Therefore, the aim of this study was to characterize mechanisms involved in the iron-related osteoblast impairment.
Methods
We studied, by using the MG-63 cell lines, the effect of iron excess on SPNS2 gene expression which was previously identified by us as potentially iron-regulated. Cell-type specificity was investigated with hepatoma HepG2 and enterocyte-like Caco-2 cell lines as well as in iron-overloaded mouse liver. The SPNS2-associated function was also investigated in MG-63 cells by fluxomic strategy which led us to determinate the S1P efflux in iron excess condition.
Results
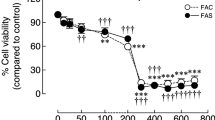
We showed in MG-63 cells that iron exposure strongly increased the mRNA level of the SPNS2 gene. This was not observed in HepG2, in Caco-2 cells, and in mouse livers. Fluxomic study performed concomitantly on MG-63 cells revealed an unexpected decrease in the cellular capacity to export S1P. Iron excess did not modulate SPHK1, SPHK2, SGPL1, or SGPP1 gene expression, but decreased COL1A1 and S1PR1 mRNA levels, suggesting a functional implication of low extracellular S1P concentration on the S1P/S1PR signalizing axis.
Conclusions
Our results indicate that iron impacts the S1P/S1PR signalizing axis in the MG-63 cell line and suggest that iron can affect the bone-associated S1P pathway and favor the occurrence of osteoporosis during chronic iron overload.






Similar content being viewed by others
Abbreviations
- C16d3Carn:
-
Deuterated C16d3 palmitoyl–carnitine
- DFO:
-
Desferrioxamine
- FAC:
-
Ferric ammonium citrate
- FCS:
-
Fetal calf serum
- GH:
-
Genetic hemochromatosis
- IREs:
-
Iron responsive elements
- MEM:
-
Minimum essential medium
- S1P:
-
Sphingosine-1-phosphate
- S1Pd7:
-
Deuterated sphingosine-1-phosphate d7
- S1PR:
-
Sphingosine-1-phosphate receptor
- Sphd7:
-
Deuterated sphingosine d7
References
(2000) Osteoporosis prevention, diagnosis, and therapy. NIH Consensus Statement 17:1–45
Zaidi M (2007) Skeletal remodeling in health and disease. Nat Med 13:791–801. https://doi.org/10.1038/nm1593
Delbarre F (1960) Osteoporosis in hemochromatosis. Sem Hop 36:3279–3294
Diamond T, Stiel D, Posen S (1989) Osteoporosis in hemochromatosis: Iron excess, gonadal deficiency, or other factors? Ann Intern Med 110:430–436. https://doi.org/10.7326/0003-4819-110-6-430
Bergeron C, Kovacs K (1978) Pituitary siderosis. A histologic, immunocytologic, and ultrastructural study. Am J Pathol 93:295–309
Guggenbuhl P, Deugnier Y, Boisdet JF, Rolland Y, Perdriger A, Pawlotsky Y, Chalès G (2005) Bone mineral density in men with genetic hemochromatosis and HFE gene mutation. Osteoporos Int 16:1809–1814. https://doi.org/10.1007/s00198-005-1934-0
Sinigaglia L, Fargion S, Fracanzani AL, Binelli L, Battafarano N, Varenna M, Piperno A, Fiorelli G (1997) Bone and joint involvement in genetic hemochromatosis: role of cirrhosis and iron overload. J Rheumatol 24:1809–1813
Domrongkitchaiporn S, Sirikulchayanonta V, Angchaisuksiri P, Stitchantrakul W, Kanokkantapong C, Rajatanavin R (2003) Abnormalities in bone mineral density and bone histology in thalassemia. J Bone Miner Res 18:1682–1688. https://doi.org/10.1359/jbmr.2003.18.9.1682
Sarrai M, Duroseau H, D’Augustine J, Moktan S, Bellevue R (2007) Bone mass density in adults with sickle cell disease. Br J Haematol 136:666–672. https://doi.org/10.1111/j.1365-2141.2006.06487.x
Kim B-J, Ahn SH, Bae SJ, Kim EH, Lee S-H, Kim H-K, Choe JW, Koh J-M, Kim GS (2012) Iron overload accelerates bone loss in healthy postmenopausal women and middle-aged men: a 3-year retrospective longitudinal study. J Bone Miner Res 27:2279–2290. https://doi.org/10.1002/jbmr.1692
Guggenbuhl P, Fergelot P, Doyard M, Libouban H, Roth M-P, Gallois Y, Chalès G, Loréal O, Chappard D (2011) Bone status in a mouse model of genetic hemochromatosis. Osteoporos Int 22:2313–2319. https://doi.org/10.1007/s00198-010-1456-2
Doyard M, Chappard D, Leroyer P, Roth M-P, Loréal O, Guggenbuhl P (2016) Decreased bone formation explains osteoporosis in a genetic mouse model of Hemochromatosiss. PLoS One 11:e0148292. https://doi.org/10.1371/journal.pone.0148292
Tsay J, Yang Z, Ross FP, Cunningham-Rundles S, Lin H, Coleman R, Mayer-Kuckuk P, Doty SB, Grady RW, Giardina PJ, Boskey AL, Vogiatzi MG (2010) Bone loss caused by iron overload in a murine model: importance of oxidative stress. Blood 116:2582–2589. https://doi.org/10.1182/blood-2009-12-260083
Matsushima S, Hoshimoto M, Torii M, Ozaki K, Narama I (2001) Iron lactate-induced osteopenia in male Sprague-Dawley rats. Toxicol Pathol 29:623–629
de Vernejoul MC, Pointillart A, Golenzer CC, Morieux C, Bielakoff J, Modrowski D, Miravet L (1984) Effects of iron overload on bone remodeling in pigs. Am J Pathol 116:377–384
Jiang Y, Yan Y, Wang X, Zhu G, Xu Y-J (2016) Hepcidin inhibition on the effect of osteogenesis in zebrafish. Biochem Biophys Res Commun 476:1–6. https://doi.org/10.1016/j.bbrc.2016.05.118
Messer JG, Kilbarger AK, Erikson KM, Kipp DE (2009) Iron overload alters iron-regulatory genes and proteins, down-regulates osteoblastic phenotype, and is associated with apoptosis in fetal rat calvaria cultures. Bone 45:972–979. https://doi.org/10.1016/j.bone.2009.07.073
Yamasaki K, Hagiwara H (2009) Excess iron inhibits osteoblast metabolism. Toxicol Lett 191:211–215. https://doi.org/10.1016/j.toxlet.2009.08.023
Doyard M, Fatih N, Monnier A, Island ML, Aubry M, Leroyer P, Bouvet R, Chalès G, Mosser J, Loréal O, Guggenbuhl P (2012) Iron excess limits HHIPL-2 gene expression and decreases osteoblastic activity in human MG-63 cells. Osteoporos Int 23:2435–2445. https://doi.org/10.1007/s00198-011-1871-z
Jia P, Xu YJ, Zhang ZL, Li K, Li B, Zhang W, Yang H (2012) Ferric ion could facilitate osteoclast differentiation and bone resorption through the production of reactive oxygen species. J Orthop Res 30:1843–1852. https://doi.org/10.1002/jor.22133
Ishii K, Fumoto T, Iwai K, Takeshita S, Ito M, Shimohata N, Aburatani H, Taketani S, Lelliott CJ, Vidal-Puig A, Ikeda K (2009) Coordination of PGC-1beta and iron uptake in mitochondrial biogenesis and osteoclast activation. Nat Med 15:259–266. https://doi.org/10.1038/nm.1910
Kawahara A, Nishi T, Hisano Y, Fukui H, Yamaguchi A, Mochizuki N (2009) The sphingolipid transporter Spns2 functions in migration of zebrafish myocardial precursors. Science 323:524–527. https://doi.org/10.1126/science.1167449
Strub GM, Maceyka M, Hait NC, Milstien S, Spiegel S (2010) Extracellular and intracellular actions of Sphingosine-1-phosphate. Adv Exp Med Biol 688:141–155
Matsuzaki E, Hiratsuka S, Hamachi T, Takahashi-Yanaga F, Hashimoto Y, Higashi K, Kobayashi M, Hirofuji T, Hirata M, Maeda K (2013) Sphingosine-1-phosphate promotes the nuclear translocation of β-catenin and thereby induces osteoprotegerin gene expression in osteoblast-like cell lines. Bone 55:315–324. https://doi.org/10.1016/j.bone.2013.04.008
Higashi K, Matsuzaki E, Hashimoto Y, Takahashi-Yanaga F, Takano A, Anan H, Hirata M, Nishimura F (2016) Sphingosine-1-phosphate/S1PR2-mediated signaling triggers Smad1/5/8 phosphorylation and thereby induces Runx2 expression in osteoblasts. Bone 93:1–11. https://doi.org/10.1016/j.bone.2016.09.003
Brizuela L, Martin C, Jeannot P, Ader I, Gstalder C, Andrieu G, Bocquet M, Laffosse J-M, Gomez-Brouchet A, Malavaud B, Sabbadini RA, Cuvillier O (2014) Osteoblast-derived sphingosine 1-phosphate to induce proliferation and confer resistance to therapeutics to bone metastasis-derived prostate cancer cells. Mol Oncol 8:1181–1195. https://doi.org/10.1016/j.molonc.2014.04.001
Ishii M, Egen JG, Klauschen F, Meier-Schellersheim M, Saeki Y, Vacher J, Proia RL, Germain RN (2009) Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 458:524–528. https://doi.org/10.1038/nature07713
Ishii M, Kikuta J, Shimazu Y, Meier-Schellersheim M, Germain RN (2010) Chemorepulsion by blood S1P regulates osteoclast precursor mobilization and bone remodeling in vivo. J Exp Med 207:2793–2798. https://doi.org/10.1084/jem.20101474
Pederson L, Ruan M, Westendorf JJ, Khosla S, Oursler MJ (2008) Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. PNAS 105:20764–20769. https://doi.org/10.1073/pnas.0805133106
Keller J, Catala-Lehnen P, Huebner AK, Jeschke A, Heckt T, Lueth A, Krause M, Koehne T, Albers J, Schulze J, Schilling S, Haberland M, Denninger H, Neven M, Hermans-Borgmeyer I, Streichert T, Breer S, Barvencik F, Levkau B, Rathkolb B, Wolf E, Calzada-Wack J, Neff F, Gailus-Durner V, Fuchs H, de Angelis MH, Klutmann S, Tsourdi E, Hofbauer LC, Kleuser B, Chun J, Schinke T, Amling M (2014) Calcitonin controls bone formation by inhibiting the release of sphingosine 1-phosphate from osteoclasts. Nat Commun 5:5215. https://doi.org/10.1038/ncomms6215
Lescoat G, Gouffier L, Cannie I, Lowe O, Morel I, Lepage S, Ropert M, Loréal O, Brissot P, Gaboriau F (2013) Involvement of polyamines in iron(III) transport in human intestinal Caco-2 cell lines. Mol Cell Biochem 378:205–215. https://doi.org/10.1007/s11010-013-1611-0
Cavey T, Ropert M, de Tayrac M, Bardou-Jacquet E, Island M-L, Leroyer P, Bendavid C, Brissot P, Loréal O (2015) Mouse genetic background impacts both on iron and non-iron metals parameters and on their relationships. Biometals 28:733–743. https://doi.org/10.1007/s10534-015-9862-8
Bielawski J, Szulc ZM, Hannun YA, Bielawska A (2006) Simultaneous quantitative analysis of bioactive sphingolipids by high-performance liquid chromatography-tandem mass spectrometry. Methods 39:82–91. https://doi.org/10.1016/j.ymeth.2006.05.004
Campillos M, Cases I, Hentze MW, Sanchez M (2010) SIREs: searching for iron-responsive elements. Nucleic Acids Res 38:W360–W367. https://doi.org/10.1093/nar/gkq371
Spiegel S, Milstien S (2003) Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat Rev Mol Cell Biol 4:397–407. https://doi.org/10.1038/nrm1103
Bradley E, Dasgupta S, Jiang X, Zhao X, Zhu G, He Q, Dinkins M, Bieberich E, Wang G (2014) Critical role of Spns2, a sphingosine-1-phosphate transporter, in lung cancer cell survival and migration. PLoS One 9:e110119. https://doi.org/10.1371/journal.pone.0110119
During A, Penel G, Hardouin P (2015) Understanding the local actions of lipids in bone physiology. Prog Lipid Res 59:126–146. https://doi.org/10.1016/j.plipres.2015.06.002
Zhang J-N, Zhao Y, Liu C, Han ES, Yu X, Lidington D, Bolz S-S, You L (2015) The role of the sphingosine-1-phosphate signaling pathway in osteocyte mechanotransduction. Bone 79:71–78. https://doi.org/10.1016/j.bone.2015.05.017
Hannun YA, Obeid LM (2008) Principles of bioactive lipid signalling: lessons from sphingolipids. Nat Rev Mol Cell Biol 9:139–150. https://doi.org/10.1038/nrm2329
Takabe K, Spiegel S (2014) Export of sphingosine-1-phosphate and cancer progression. J Lipid Res 55:1839–1846. https://doi.org/10.1194/jlr.R046656
Hisano Y, Kobayashi N, Kawahara A, Yamaguchi A, Nishi T (2011) The sphingosine 1-phosphate transporter, SPNS2, functions as a transporter of the phosphorylated form of the Immunomodulating agent FTY720. J Biol Chem 286:1758–1766. https://doi.org/10.1074/jbc.M110.171116
Nagahashi M, Kim EY, Yamada A, Ramachandran S, Allegood JC, Hait NC, Maceyka M, Milstien S, Takabe K, Spiegel S (2013) Spns2, a transporter of phosphorylated sphingoid bases, regulates their blood and lymph levels, and the lymphatic network. FASEB J 27:1001–1011. https://doi.org/10.1096/fj.12-219618
Lee Y-J, Huang X, Kropat J, Henras A, Merchant SS, Dickson RC, Chanfreau GF (2012) Sphingolipid signaling mediates iron toxicity. Cell Metab 16:90–96. https://doi.org/10.1016/j.cmet.2012.06.004
Lester RL, Withers BR, Schultz MA, Dickson RC (2013) Iron, glucose and intrinsic factors alter sphingolipid composition as yeast cells enter stationary phase. Biochim Biophys Acta 1831:726–736. https://doi.org/10.1016/j.bbalip.2012.12.012
Acknowledgements
The authors would like to thank Dr. François Gaboriau and Dr. Lenaick Detivaud for helpful advice and assistance, Claudine Rauch and Brigitte Frenot for technical assistance with cell culture and mass spectrometry, Pr. Véronique David and Dr. Nathalie Le Floc’h for helpful discussions, and Dr. Caroline Moreau for global assistance and support in this project.
Funding
This work was supported by the French Society of Rheumatology (SFR) [grant numbers 2320, 2013].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Lucas Peltier, Claude Bendavid, Thibault Cavey, Marie-Laure Island, Mathilde Doyard, Patricia Leroyer, Coralie Allain, Marie De Tayrac, Martine Ropert, Olivier Loréal and Pascal Guggenbuhl declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 719 kb)
Rights and permissions
About this article
Cite this article
Peltier, L., Bendavid, C., Cavey, T. et al. Iron excess upregulates SPNS2 mRNA levels but reduces sphingosine-1-phosphate export in human osteoblastic MG-63 cells. Osteoporos Int 29, 1905–1915 (2018). https://doi.org/10.1007/s00198-018-4531-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-018-4531-8