Abstract
Summary
Osteoporosis is a major public health problem worldwide. Its significance in the fields of traumatology and implantology of the maxillofacial area requires investigation. A large animal model was used to assess bone loss in the lumbar spine and mandible.
Introduction
Osteoporosis is a prevalent disease characterized by low bone mass and microarchitectural deterioration of bone tissue. Osteoporosis-related fractures represent a major public health burden. The presence and relevance of osteoporosis in the maxillofacial area remain controversial. Research in humans is limited by difficulties in finding large homogenous study groups and, due to ethical considerations, numerous animal models have been used in osteoporosis research. The aim of this study was to assess a sheep model of generalized osteopenia for changes in the maxillofacial area.
Methods
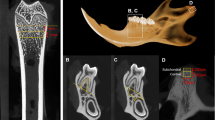
Bone loss was induced in ten Merino sheep by ovariectomy, intramuscular administration of glucocorticoids, and a calcium-reduced diet. Five untreated animals served as controls. Bone mineral density (BMD) was assessed by dual-energy X-ray absorptiometry at the lumbar spine at baseline and after 1, 3, 6, 9, and 12 months. Lumbar and mandibular bone biopsies were obtained and analyzed with microcomputed tomography.
Results
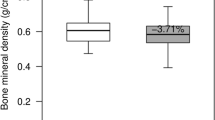
Lumbar BMD decreased progressively in the intervention group and was most significantly low after 6 months (p < 0.001). Lumbar trabecular bone showed a significant decrease in bone volume (BV)/tissue volume (TV; p < 0.05) in the inducted group. Significant changes were found in both analyzed mandibular regions for BV/TV (p < 0.05). Regional variations were found for other parameters in the mandible. The cortical width was substantially reduced in the intervention group (p < 0.001).
Conclusions
Microstructural changes occurring in sheep as a result of induction seem to have a generalized nature. This sheep model meets the criteria for further investigation in the maxillofacial area.


Similar content being viewed by others
References
WHO Scientific Group on the Prevention and Management of Osteoporosis (2003) Prevention and management of Osteoporosis: report of a WHO scientific group. WHO technical report series: pp 1–7, 45, 53–65
Norris RJ (1992) Medical costs of osteoporosis. Bone 13(2):S11–S16
MacLeay JM, Olson JD, Emms RM, Les CM, Toth CA, Wheeler DL, Turner AS (2004) Dietary-induced metabolic acidosis decreases bone mineral density in mature ovariectomized ewes. Calcif Tissue Int 75:431–437
Egermann M, Goldhahn J, Schneider E (2005) Animal models for fracture treatment in osteoporosis. Osteoporos Int 16(2):S129–S138
Turner AS (2001) Animal models of osteoporosis—necessity and limitations. Eur Cell Mater 22(1):66–81
Lill CA, Fluegel AK, Schneider E (2002) Effect of ovariectomy, malnutrition and glucocorticoid application on bone properties in sheep: a pilot study. Osteoporos Int 13(6):480–486
Kimmel DB (1996) Animal models for in vivo experimentation in osteoporosis research. In: Marcus R, Feldman D, Kelsey J (eds) Osteoporosis, 1st edn. Academic, London, pp 671–681
Hohlweg-Majert B, Schmelzeisen R, Pfeiffer BM, Schneider E (2006) Significance of osteoporosis in craniomaxillofacial surgery: a review of the literature. Osteoporos Int 17(2):167–179
Bodic F, Hamel L, Lerouxel E, Baslé MF, Chappard D (2005) Bone loss and teeth. Joint Bone Spine 72(3):215–221
von Wowern N (1988) Bone mineral content of mandibles: normal reference values—rate of age-related bone loss. Calcif Tissue Int 43(4):193–198
Ulm CW, Solar P, Ulm MR, Matejka M (1994) Sex-related changes in the bone mineral content of atrophic mandibles. Calcif Tissue Int 54(3):203–207
Choël L, Duboeuf F, Bourgeois D, Briguet A, Lissac M (2003) Trabecular alveolar bone in the human mandible: a dual-energy X-ray absorptiometry study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 95(3):364–370
Werning JW, Downey NM, Brinker RA, Khuder SA, Davis WJ, Rubin AM, Elsamaloty HM (2004) The impact of osteoporosis on patients with maxillofacial trauma. Arch Otolaryngol Head Neck Surg 130(3):353–356
Iida S, Reuther T, Kogo M, Matsuya T, Mühling J (2002) Retrospective analysis of facial fractures related to falls in 260 Japanese patients. Mund Kiefer Gesichtschir 6(6):421–426
Sundstol F, Owen E (1984) Straw and other fibrous by-products as feed. Elsevier Science Publishers B.V, Amsterdam
Zarrinkalam MR, Beard H, Schultz CG, Moore RJ (2009) Validation of the sheep as a large animal model for the study of vertebral osteoporosis. Eur Spine J 18(2):244–253
Newton BI, Cooper RC, Gilbert JA, Johnson RB, Zardiackas LD (2004) The ovariectomized sheep as a model for human bone loss. J Comp Pathol 130(4):323–326
Aldini NN, Fini M, Giavaresi G, Giardino R, Greggi T, Parisini P (2002) Pedicular fixation in the osteoporotic spine: a pilot in vivo study on long-term ovariectomized sheep. J Orthop Res 20(6):1217–1224
Giavaresi G, Fini M, Torricelli P, Martini L, Giardino R (2001) The ovariectomized ewe model in the evaluation of biomaterials for prosthetic devices in spinal fixation. Int J Artif Organs 24(11):814–820
Tsugeno H, Tsugeno H, Fujita T, Goto B, Sugishita T, Hosaki Y, Ashida K, Mitsunobu F, Tanizaki Y, Shiratori Y (2002) Vertebral fracture and cortical bone changes in corticosteroid-induced osteoporosis. Osteoporosis Int 13(8):650–656
Schorlemmer S, Gohl C, Iwabu S, Ignatius A, Claes L, Augat P (2003) Glucocorticoid treatment of ovariectomized sheep affects mineral density, structure, and mechanical properties of cancellous bone. J Bone Miner Res 18(11):2010–2015
Lill CA, Fluegel AK, Schneider E (2000) Sheep model for fracture treatment in osteoporotic bone: a pilot study about different induction regimens. J Orthop Trauma 14(8):559–565
Goldhahn J, Jenet A, Schneider E, Lill CA (2005) Slow rebound of cancellous bone after mainly steroid-induced osteoporosis in ovariectomized sheep. J Orthop Trauma 19(1):23–28
Lund B, Storm TL, Lund B, Melsen F, Mosekilde L, Andersen RB, Egmose C, Sørensen OH (1985) Bone mineral loss, bone histomorphometry and vitamin D metabolism in patients with rheumatoid arthritis on long-term glucocorticoid treatment. Clin Rheumatol 4(2):143–149
Lill CA, Gerlach UV, Eckhardt C, Goldhahn J, Schneider E (2002) Bone changes due to glucocorticoid application in an ovariectomized animal model for fracture treatment in osteoporosis. Osteoporos Int 13(5):407–414
Acknowledgments
Financial support for this study was provided by a Research Fund from the AO Foundation (Project S-07-31V) and the German Research Foundation (DFG) (Project SCHM 769/10-1). The authors would like to specially thank Prof. Dr. Benjamin Martínez (Universidad Mayor, Santiago de Chile) for the outstanding scientific cooperation and Adelaide Microscopy for excellent technical assistance.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Veigel, E., Moore, R.J., Zarrinkalam, M.R. et al. Osteopenia in the maxillofacial area: a study in sheep. Osteoporos Int 22, 1115–1121 (2011). https://doi.org/10.1007/s00198-010-1289-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1289-z