Abstract
Summary
The aim of the present study was to determine the action of AsGA laser irradiation on bone repair in the tibia of osteopenic rats. The animals were randomly divided into eight experimental groups according to the presence of ovarian hormone (sham group) or the absence of the hormone (OVX group), as well as being irradiated or non-irradiated. Low-level 904-nm laser (50 mJ/cm2) accelerated the repair process of osteopenic fractures, especially in the initial phase of bone regeneration.
Introduction
The development of new techniques to speed the process of bone repair has provided significant advances in the treatment of fractures. Some attention recently focused on the effects of biostimulation on bone.
Methods
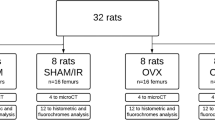
Forty-eight adult rats were randomly divided into eight experimental groups (six animals in each group) according to the presence of ovarian hormone (sham group) or absence of the hormone (ovariectomized (OVX) group) as well as being irradiated or non-irradiated. For the application of low-level laser therapy, the animals were anesthetized with one third of the dose sufficient to immobilize the animal and irradiated with AsGa laser (904 nm, 50 mJ/cm2 for 2 s, point form and in contact). The control animals received the same type of manipulation as the irradiated animals, but with the laser turned off. Half of the animals were killed 7 days following the confection of the bone defect, and the other half were killed 21 days after the surgery. After complete demineralization, the tibias were cut cross-sectionally in the central region of the bone defect and embedded in paraffin blocks. The blocks were then cut in semi-seriated slices and stained with hematoxylin and eosin.
Results
There was new bone formation in the animals in the OVX group with laser treatment killed after 7 days (p < 0.001). The lowest percentage of bone formation was observed in the OVX without laser killed after 7 days (p > 0.05). All animals killed after 21 days exhibited linear closure of the lesion.
Conclusion
Low-level 904-nm laser (50 mJ/cm2) accelerated the repair process of osteopenic fractures, especially in the initial phase of bone regeneration.
Similar content being viewed by others
Introduction
Osteoporosis is a skeletal disease characterized by the reduction in and deterioration of the micro-architecture of bone tissue, with a consequent increase in bone frailty and susceptibility to fractures, especially among the radius, femur, and bones of the spinal column [1, 2]. Osteoporosis has considerable socioeconomic impact and is recognized as a major public health problem stemming from different physiological and pathological conditions, with physiological bone loss occurring mainly at more advanced ages [3].
A large number of treatment options have been developed with the aim of preventing bone loss and stimulating bone formation, including hormonal replacement with estrogen, bisphosphonates, and physical activity programs. The effect of low-level laser therapy (LLLT) on bone regeneration has been the focus of recent studies [4, 5] and has been employed in the repair of fractures or bone perforations with the aim of reducing healing time [6, 7]. LLLT has a stimulating effect on bone tissue, enabling an increase in cell proliferation and accelerating the consolidation of fractures [8].
Currently, attention has been focused on the effects of photo-irradiation on bone tissue. This type of therapy has been reported to have biostimulatory effects on different cells and organelles, demonstrating effectiveness in bone cell stimulation and proliferation, enhancing osteoblast activity at the fracture site, and contributing toward the acceleration of the consolidation of fractures. LLLT also favors angiogenesis and enhances the deposit of collagen fibers [2, 4, 6, 7, 9].
Irradiation at 830 nm and an energy density of 4.8 J/cm2 has demonstrated a biostimulatory effect on the repair of bone defects induced in rats. These effects are reported to be the result of laser irradiation on the cell membrane, mitochondria, synthesis of DNA and RNA, collagen synthesis, prostaglandin levels, neovascularization, cell proliferation, and the production of ATP [10]. A number of studies involving laser therapy have demonstrated positive effects on bone tissue. These studies reflect the idea that undifferentiated mesenchymal cells may be biomodulated by activating osteoblasts [3, 11].
Ovariectomized (OVX) rats are one of the experimental models used in the study of bone loss due to estrogen deficiency which contributes to the development of osteopenia with a consequent reduction in bone mass and increase in bone frailty. The characteristics of bone loss in rats following ovariectomy are very similar to those found in postmenopausal women [12].
The use of LLLT for biostimulation in bone repair has been on the rise. A number of studies have demonstrated positive results regarding the healing of bone tissue. The use of λ = 904 nm arsenide–gallium laser (AsGa) has increased in the last 10 years, which has a greater penetration power in comparison to other types of laser, thereby making it an indispensable resource in rehabilitation therapy [13]. We therefore hypothesized that 904-nm laser could act on bone regeneration in osteopenic tissue at a low dose following OVX and with no combined medication.
The aim of the present study was to determine the action of AsGA laser irradiation on bone repair in the tibia of osteopenic rats using bone densitometry and histomorphometric analyses.
Materials and methods
Forty-eight adult rats (Rattus norvegicus albinus, Wistar variety) were used. The animals were provided by the Bioterium of the São José dos Campos Dental School/Universidade Estadual de São Paulo; were approximately 90 days of age, with body mass of 300 g; were kept in cages at room temperature; and were offered food (Guabi Nutrilabor feed) and water ad libitum. The study was conducted in compliance with the Ethical Principles on Animal Experimentation adopted by the Brazilian College of Animal Experimentation (COBEA) and received approval from the Research Ethics Committee of the Universidade Vale do Paraíba (process no. A045/2006/CEP).
Surgical procedures
All procedures were performed in the surgery room of the Bioterium of the São José dos Campos Dental School/Universidade Estadual de São Paulo. For general anesthesia, the rats received a solution of 13 mg/kg of 2-(2,6-xylidine)-5,6-dihydro-4H-1,3-thiazine chlorhydrate (Anasedan, Bayer of Brazil)—a substance with sedative, analgesic, and muscle-relaxant properties—and 33 mg/kg of ketamine (Dopalen, Agribands do Brasil Ltda) via intramuscular injection.
Ovariectomy and simulated ovariectomy surgery (sham)
All rats pertaining to the OVX group at 3 months of age were anesthetized, the lateral abdominal region was shaved and underwent antiseptic treatment with iodated alcohol, and a 1-cm longitudinal incision was made in the skin and musculature of the lateral region of the body near the kidney level and below the last rib with a no. 15 scalpel. The ovary was identified and exposed. The ligature was performed using no. 4.0 silk thread (Ethicon/Johnson & Johnson, São José dos Campos, SP, Brazil) just below the ovary for hemostasis; these procedures were performed bilaterally. The ovaries were removed together with the surrounding fat tissue and a small portion of the uterus. At the end of the procedures, the muscle layers were sutured with absorbable no. 4 catgut (Cirumédica, Cotia, SP, Brazil) and the skin was sutured with 4.0 silk thread (Ethicon/Johnson & Johnson). The same procedures, except the hemostasis (ligature) and the removal of the ovaries, were performed on the animals in the sham group (simulated surgery) in order to simulate surgical stress. Thus, after exposing the ovaries, the organs were replaced into the abdominal cavity and the tissues were sutured.
Confection of monocortical bone defect
Thirty days following ovariectomy and sham surgery, all animals were anesthetized to receive the bone defect in the right tibia. After shaving the skin and antiseptic treatment of the site, an incision of approximately 2 cm was made in the skin and muscle of the proximal third of the tibia to the periosteum with a no. 15 scalpel. The soft tissue and periosteum were moved aside with a no. 7 spatula for exposure of the bone. The monocortical defect was made in the region of the greatest lateral medial extension of the bone with the use of a trephine bur (3 mm in diameter) and electric motor (aseptic; AEV-707 Implant Surgery System) at 1,600 rpm under constant irrigation and abundant 0.9% sodium chloride. The subsequent suturing of the muscle layer was performed with absorbable no. 4 catgut (Cirumédica); the skin was sutured with 4.0 silk thread (Ethicon/Johnson & Johnson) and swabbed with iodated alcohol again.
Division of experimental groups
The animals were randomly divided into eight experimental groups (six animals in each group) according to the presence of ovarian hormone (sham group) or absence of the hormone (OVX group) as well as being irradiated or non-irradiated. Half of the animals were killed 7 days following the confection of the bone defect, and the other half were killed 21 days after the surgery (Table 1).
Low-level laser therapy
LLLT was performed beginning 24 h after the confection of the bone defect at 48-h intervals. The group killed on the seventh day received a total of four applications, and the group killed on the 21st day received a total of 11 applications. For the application of LLLT, the animals were anesthetized with one third of the dose sufficient to immobilize the animal and irradiated with AsGa laser (904 nm, Endophoton, model LLTO 0107 KLD brand, Biosistemas Equipamentos Eletrônicos Ltda, Brasil), repetition rate of 10 KHz, output power of 50 mW, irradiated at 50 mJ/cm2 for 2 s in point form, and contact at four points on the skin over the bone defect region. The energy density used in the present study was based on studies carried out by Oliveira et al. [14] and Pires Oliveira et al. [15] in which doses of 6 J/cm2 and 50 mJ/cm2 were used on cell cultures, with a better response obtained at 50 mJ/cm2. Although these studies involved cell cultures, the 904-nm laser at a dose of 50 mJ/cm2 proved to have a greater effect on cell growth rates.
The control animals received the same type of manipulation as the irradiated animals, but with the laser turned off. A mechanical support was used so that all animals received the same contact with the laser pen during application.
Euthanasia
For killing, the animals were anesthetized through an intramuscle injection of pre-anesthetic medication. After 15 min, the anesthesia was administered with an intraperitoneal injection of Zoletil (40 mg/kg, IM; Zolazepam tiletamine, La. Virbac S.A), and the animals were killed with an intra-heart injection of potassium chloride. Following euthanasia, the tibia was removed, the soft tissue was eliminated, and the bone was fixed in 10% buffered formaldehyde solution for a minimum of 48 h.
Histomorphometric analysis
After taking radiograms, the right tibias were submitted to decalcification in an aqueous solution with 100 g of EDTA and 10 g of PA sodium hydroxide diluted in 1,000 ml of distilled water for approximately 120 days. After complete demineralization, the tibias were cut cross-sectionally in the central region of the bone defect and embedded in paraffin blocks. The blocks were then cut in semi-seriated slices, stained with hematoxylin and eosin, and examined under an optical microscope. For the morphometric analysis of the neoformed bone tissue, a grid was used with 80 points resulting from the intersections between the ten vertical and eight horizontal lines, which was superimposed on the histology image. The histomorphometrical analysis involved a stereological method based on that proposed by Taga and Stipp [16] and Junqueira et al. [17]. Volume density evaluation (Vvi) was expressed in percentages using a square reticule and ×20 objective. The image of the reticule was laid over the histological field, and a count was made of the number of intersection points between the reticule lines over a particular structure and the total number of points over the bone defect. The fraction of volume density of the bone defect occupied by this structure was calculated using the formula below:
Statistical analysis
Results are expressed as mean values ± SEM. Comparisons between groups were performed with one-way analysis of variance. Post hoc Tukey honestly significant difference analysis was used to determine significant differences between groups (95% confidence interval). Values of p ≤ 0.05 were considered statistically significant. Data were analyzed using the GraphPad Prism 4.0 statistical software (GraphPad Software, San Diego, CA, USA).
Results
The histomorphometric analysis of the irradiated and non-irradiated groups killed at 7 and 21 days revealed the bone defect region (easily located and distinguished from the rest of the cortical bone) with well-defined margins and intensive neoformation of bone tissue of a spongy appearance originating from the extremities toward the center of the lesion. The histological pattern was similar in the experimental groups at 7 days, although differences were found when comparing the groups killed at 7 and 21 days (Fig. 1).
In the OVX group without laser killed at 7 days, there were numerous, disorganized, immature trabeculae that differed from the trabeculae located on the margin, which demonstrated a more mature pattern, were less defined, less numerous, and hypodense (Fig. 1a) when compared to the OVX group with laser killed at 7 days (Fig. 1b).
In the animals killed at 21 days, the most striking characteristic was the presence of a band of bone tissue uniting the margins of the defect, which was more mature and organized, with a cortical appearance (Fig. 1c, d). All animals exhibited linear closure of the lesion. The most striking difference between the irradiated and non-irradiated OVX animals at 21 days was the greater thickness of the bone bridge in the irradiated group (Fig. 1d).
Figures 2 and 3 display the histomorphometric data on the irradiated and non-irradiated animals (sham and OVX) killed at 7 and 21 days. The mean values for the OVX group with laser irradiation killed at 7 days (p < 0.001) were higher than the other groups (Fig. 2). The lowest percentage of bone formation was found in the OVX group without laser irradiation killed at 7 days (p > 0.05). Regarding the animals killed at 21 days (Fig. 3), the values for the sham group without laser irradiation were higher than the other groups (p < 0.001), and there were statistically significant differences between the OVX groups treated with and without laser irradiation (p < 0.01).
Discussion
The use of laser for the biostimulation of bone repair has been on the rise, and a number of studies report positive results regarding the healing of bone tissue. However, osteopenic bone tissue has been under-investigated. From the standpoint of physical therapy, this is an important issue especially when considering the safety and effectiveness of LLLT in elderly individuals with osteopenia and some types of fracture [6, 12, 18].
The radiation emitted in the near-infrared region (904 nm) has a low absorption coefficient and, consequently, greater penetration potential into the tissue, increasing the resistance and volume and promoting bone mineralization. This is a clear indication for the use of the 904-nm infrared laser on bone tissue, making it an indispensable resource for bone rehabilitation therapy [3, 11, 13].
The histomorphometric analysis of the bone revealed that the laser (904 nm, 50 mJ/cm2) had an inductive effect in both the sham and OVX groups killed at 7 days, as demonstrated by the presence of immature, disorganized trabeculae differing from the non-irradiated groups. This finding is in agreement with that described by Dörtbudak et al. [19] who report that laser has a significant effect on the production of bone matrix.
Pinheiro and Gerbi [3] report that the stimulatory effect of laser irradiation on bone tissue occurs during the initial proliferation phase in fibroblastic and osteoblastic cells as well as the initial differentiation of mesenchymal cells. According to the authors, bone repair begins immediately following an injury or damage in vascularized regions in tissue anoxia, and the acceleration of the repair may be the result of LLLT on the synthesis of the bone matrix due to the increase in vascularization and early onset of the inflammatory response. These data corroborate the findings of the present study in which a better response to LLLT was found in the irradiated OVX group killed at 7 days in comparison to the sham group which exhibited granulation tissue, thereby suggesting the inducing effect on bone repair in tissue with bone loss.
Irradiation in the early stage of bone repair and repeated irradiation over a short period of time may be effective. LLLT has been used in the repair of bone fractures or perforations with the aim of reducing the repair time [20]. Laser irradiation in the first 3 days is of considerable efficacy, and irradiation in the initial repair stage for a short period may be effective [21]. In the present study, the irradiated animals in the OVX group had a more positive response at 7 days (p < 0.001) when compared to 21 days.
Laser irradiation stimulates cell proliferation and differentiation of the osteoblast cell line, resulting in an increase in the differentiation of osteoblasts and bone formation [22]. The action of LLLT has been investigated in bone tissue in different fields of medicine and dentistry, with wavelengths ranging from 670 to 1,064 nm. However, there is a scarcity of studies on the use of 904-nm laser on bone tissue and in the field of physical therapy. The most often employed lasers are 670, 690, 780, 830, and 1,064 nm [2, 6, 8, 11–13, 19].
Biomodulation triggered by laser irradiation on cells depends on a combination of parameters, such as wavelength, energy density, and power density, which contribute effectiveness to the stimulation and proliferation of bone cells, increasing osteoblastic activity at the fracture site and contributing toward the acceleration of the consolidation of the fracture. LLLT also favors angiogenesis and increases the depositing of collagen fibers [4, 6–9, 11, 14, 18, 23–25].
The negative results found in some papers on the use of laser irradiation on bone regeneration may be attributed to very low levels and an inappropriate wavelength [12]. The energy density employed in the present study (50–200 mJ/cm2, 7 days) had a biostimulatory effect, suggesting that the 904-nm wavelength was adequate, which is an important factor in relation to energy density.
Studies comparing low-level laser and low-level ultrasound found that the effect of 904-nm laser on fibroblast cultures was greater than ultrasound, and the laser irradiation achieved better results regarding cell growth when at 50 mJ/cm2 than at 6 J/cm2 [14].
The therapeutic progress resulting from technological advances in recent years has led to a significant increase in life expectancy, with a subsequent increase in the population over the age of 65 years. This has created a need for research on physiopathologies as well as the prevention and treatment of health conditions related to aging [18].
Conclusion
The 904-nm laser at a low dose (50 mJ/cm2) accelerated the repair process in osteopenic fractures, especially in the initial phase of bone regeneration. Further investigations are needed to evaluate the possible response mechanisms that may explain the contrasting results obtained in the analysis of laser irradiation on osteopenic tissue. Such studies will contribute toward a better understanding of the safety and efficacy of laser irradiation in rehabilitation therapy.
References
Genant HK et al (2007) Severity of vertebral fracture reflects deterioration of bone microarchitecture. Osteoporos Int 18:69–76
Renno ACM et al (2006) Effects of 830 nm laser used in two doses, on biomechanical properties of osteopenic rat femora. Photomed Laser Surg 24:202–206
Pinheiro ALB, Gerbi MEMM (2006) Photoengineering of bone repair processes. Photomed Laser Surg 24(2):169–178
Khadra M et al (2005) Effect of laser therapy on attachment, proliferation and differentiation of human osteoblast-like cells cultured on titanium implant material. Biomaterials 26:3503–3509
Shimizu N et al (2007) Low-intensity laser irradiation stimulates bone nodule formation via insulin-like factor-I expression in rat calvarial cells. Lasers Surg Med 39:551–559
Diniz JS et al (2009) Effect of low-power gallium-aluminum-arsenium laser therapy (830 nm) in combination with bisphosphonate treatment on osteopenic bone structure: an experimental animal study. Lasers Med Sci 24(3):347–352
Obradovié RR, Kesié LG, Pesevska S (2009) Influence of low-level laser therapy on biomaterial osseointegration: a mini-review. Lasers Med Sci 24:447–451
Renno ACM et al (2007) The effects of laser irradiation on the osteoblast and osteosarcoma cell proliferation and differentiation in vitro. Photomed Laser Surg 25:275–280
Barber A et al (2001) Advances in laser therapy for bone repair. Laser Ther 13:80–85
Pinheiro ALB et al (2003) Effect of 830 nm laser light on the repair of bone defects grafted with inorganic bovine bone and descalcified cortical osseous membrane. J Clin Laser Med Surg 21(6):383–388
Ninomiya T et al (2007) Increase of bone volume by a nanosecond pulsed laser irradiation is caused by a decreased osteoclast number and an activated osteoblasts. Bone 40:140–148
Renno ACM et al (2006) Effects of 830 nm laser, light on preventing bone loss after ovariectomy. Photomed Laser Surg 24:642–645
Nissan J et al (2006) Effect of low intensity laser irradiation on surgically created bony defects in rats. J Oral Rehabil 33:619–624
Oliveira RF et al (2008) Comparison between the effect of low level laser therapy and low intensity pulsed ultrasonic irradiation in vitro. Photomed Laser Surg 26:6–9
Pires Oliveira DAA et al. (2010) Laser biomodulation on L 929 cell cultures. Photomed Laser Surg (in press)
Taga R, Stipp ACM (1994) Morphometry practical manual of the optical microscopy. Planning and developing an experiment. Bauru School of Dentistry, pp 1–35
Junqueira JC et al (2002) Effects of simvastatin on bone regeneration in the mandibles of ovariectomized rats and on blood cholesterol levels. J Oral Sci 44(3/4):117–124
Pires-Oliveira DAA et al (2008) Evaluation of low-level laser therapy of osteoblastic cells. Photomed Laser Surg 26(4):401–404
Dörtbudak O, Haas R, Mailath-Pokorny G (2000) Biostimulation of bone marrow cells with a diode soft laser. Clin Oral Implants Res 11:540–545
Merli LAS et al (2005) Effect of low-intensity laser irradiation on process of bone repair. Photomed Laser Surg 23:212–215
Saito S, Shimizu N (1997) Stimulatory effects of low power irradiation on bone regeneration in midpalatal suture during expansion in the rat. Am J Orthod Dentofac Orthop 111:525–532
Stein A et al (2005) Low-level laser irradiation promotes proliferation and differentiation of human osteoblasts in vitro. Photomed Laser Surg 23:161–166
Silveira PCL, Streck EL, Pinho RA (2007) Evaluation of mitochondrial respiratory chain activity in wound healing by low-level laser therapy. J Photochem Photobiol B 86:279–282
Pereira AN et al (2002) Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg Med 31:263–267
Sato S et al (2001) Nanosecond high-intensity pulsed laser ablation of myocardium tissue at the ultraviolet, visible, and near-infrared wavelengths: in vitro study. Lasers Surg Med 29:464–473
Acknowledgments
We thank KLD™ Biosistemas Equipamentos Eletrônicos Ltda and Nelson Fuirini Junior, Brazil for supporting this work.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pires-Oliveira, D.A.A., Oliveira, R.F., Amadei, S.U. et al. Laser 904 nm action on bone repair in rats with osteoporosis. Osteoporos Int 21, 2109–2114 (2010). https://doi.org/10.1007/s00198-010-1183-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1183-8