Abstract
Introduction and hypothesis
The differential diagnosis of urinary symptoms may allow health professionals to establish a therapeutic objective and to choose the appropriate treatment for the patient's complaint. The aim of this study was to cross-culturally adapt the Three Incontinence Questionnaire (3IQ) into Brazilian Portuguese (3IQ-Br) and to analyze test-retest reliability, construct, and criterion validity in women.
Methods
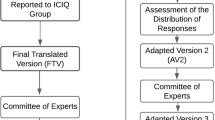
The cross-cultural adaptation of the 3IQ-Br included forward-translation, back-translation, and consensus among an expert committee. Participants with and without urinary incontinence (UI) completed the 3IQ-Br, King's Health Questionnaire (KHQ), and Questionnaire for Female Urinary Incontinence Diagnosis (QUID-Br). Only women with UI answered 3IQ-Br after 7–10 days. Test-retest reliability and construct validity were analyzed using the Cohen linear kappa (k). The 3IQ-Br accuracy was analyzed using the area under the curve (AUC) of the receiver-operating characteristic (ROC) curve, considering the sensitivity and specificity to correctly classify women with and without UI.
Results
The reliability of each question from the 3IQ-Br was considered substantial in the test-retest. The agreement among 3IQ-Br, QUID-Br, and KHQ was almost perfect for UI diagnosis (k > 0.8). The 3IQ-Br was considered to have good accuracy in distinguishing women with UI considering the KHQ (AUC 0.83, 95% confidence interval [CI] 0.78 to 0.87, p < 0.001), and fair to the QUID-Br (AUC 0.73, 95% CI 0.68 to 0.78; p < 0.001).
Conclusions
The results of this study showed that this version of the 3IQ-Br has acceptable measurement properties for identifying and differentiating UI symptoms in Brazilian women.


Similar content being viewed by others
References
McKellar K, Abraham N. Prevalence, risk factors, and treatment for women with stress urinary incontinence in a racially and ethnically diverse population.pdf. Neurourol Urodyn. 2019;38:934–40.
Abrams P, Cardozo L, Wagg A, et al. Evaluation and treatment of urinary incontinence, pelvic organ prolapse and faecal incontinence. Neurourol Urodyn. 2017;37:2271–2.
Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol Urodyn. 2010;29:4–20.
Pizzol D, Demurtas J, Celotto S, et al. Urinary incontinence and quality of life: a systematic review and meta-analysis. Aging Clin Exp Res. 2021;33:25–35.
Dedicação AC, Haddad M, Saldanha MES, et al. Comparação da qualidade de vida nos diferentes tipos de incontinência urinária feminina. Rev Bras Fisioter. 2009;13:116–22.
Norton JM, Dodson JL, Newman DK, et al. Nonbiologic factors that impact management in women with urinary incontinence: review of the literature and findings from a National Institute of Diabetes and Digestive and Kidney Diseases workshop. Int Urogynecol J. 2017;28:1295–307.
John G, Bardini C, Combescure C, et al. Urinary incontinence as a predictor of death: a systematic review and meta-analysis. PLoS ONE. 2016;11:1–19.
Brown JS, Bradley CS, Subak LL, et al. The sensitivity and specificity of a simple test to distinguish between urge and stress urinary incontinence. Ann Intern Med. 2006;144:715–24.
Nunes Tamanini JT, Levi D’Ancona CA, Botega NJ, et al. Validation of the Portuguese version of the King’s Health Questionnaire for urinary incontinent women. Rev Saude Publica. 2003;37:203–11.
Alem MER, Chaves TC, Figueiredo VB, Nascimento SL, Beleza ACS, Driusso P. Cross-cultural adaptation to Brazilian Portuguese and assessment of the measurement properties of the Questionnaire for Urinary Incontinence Diagnosis (QUID). Eur J Obstet Gynecol Reprod Biol. 2020;225:111–7.
Khan MJ, Omar MA, Laniado M. Diagnostic agreement of the 3 Incontinence Questionnaire to video-urodynamics findings in women with urinary incontinence. Cent Eur J Urol. 2018;71:84–91.
Kelleher CJ, Cardozo LD, Khullar V, et al. A new questionnaire to assess the quality of life of urinary incontinent women. BJOG An Int J Obstet Gynaecol. 1997;104:1374–9.
Fonseca MCM, Sartori MGF, de Castello Girão MJB, et al. Validação do questionário de qualidade de vida (King’s Health Questionnaire) em mulheres brasileiras com incontinência urinária. Rev Bras Ginecol e Obs. 2006;27:235–42.
Bradley CS, Rovner ES, Morgan MA, et al. A new questionnaire for urinary incontinence diagnosis in women: development and testing. Am J Obstet Gynecol. 2005;192:66–73.
Bradley CS, Rahn DD, Nygaard IE, et al. The Questionnaire for Urinary Incontinence Diagnosis (QUID): validity and responsiveness to change in women undergoing non-surgical therapies for treatment of stress predominant urinary incontinence. Neurourol Urodyn. 2010;29:727–34.
Wild D, Grove A, Martin M, et al. Principles of good practice for the translation and cultural adaptation process for patient-reported outcomes ( PRO ) measures: report of the ISPOR Task Force for Translation and Cultural Adaptation. Value Heal. 2005;8:94–104.
Beaton DE, Bombardier C, Guillemin F, et al. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine (Phila Pa 1976). 2000;25:3186–91.
Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J Clin Epidemiol. 2010;63:737–45.
Terwee CB, Bot SDM, de Boer MR, et al. Quality criteria were proposed for measurement properties of health status questionnaires. J Clin Epidemiol. 2007;60:34–42.
Deyo RA, Centor RM. Assessing the responsiveness of functional scales to clinical change: an analogy to diagnostic test performance. J Chronic Dis. 1986;39:897–906.
Akobeng AK. Understanding diagnostic tests 3: receiver operating characteristic curves. Acta Paediatr Int J Paediatr. 2007;96:644–7.
Hanley JA, McNeil BJ. The meaning an use of the area under a recevier operating characteristic (ROC) curve. Radiology. 1982;143:29–36.
Mokkink LB, de Vet HCW, Prinsen CAC, et al. COSMIN Risk of Bias checklist for systematic reviews of Patient-Reported Outcome Measures. Qual Life Res. 2018;27:1171–9.
Raine R, Fitzpatrick R, Barratt H, et al. Challenges, solutions and future directions in the evaluation of service innovations in health care and public health. Heal Serv Deliv Res. 2016;4:1–136.
Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: an international Delphi study. Qual Life Res. 2010;19:539–49.
Mokkink LB, Prinsen CA, Patrick DL, et al. COSMIN manual for systematic reviews of PROMs, user manual. 2018;1–78.
Sánchez BS, Torres Lacomba M, Navarro Brazález B, et al. Responsiveness of the Spanish pelvic floor distress inventory and pelvic floor impact questionnaires short forms (PFDI-20 and PFIQ-7) in women with pelvic floor disorders. Eur J Obstet Gynecol Reprod Biol. 2015;190:20–5.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and 2018/26718-9, São Paulo. Research Foundation (FAPESP).
Author information
Authors and Affiliations
Contributions
MER Alem: project development, data collection and analysis, manuscript writing.
JB Silva: data collection, support for writing the manuscript.
ACS Beleza: drafting and revising the article critically.
TC Chaves: data analysis.
P Driusso: project development, drafting and revising the article critically, and final approval of the version to be published.
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alem, M.E.R., da Silva, J.B., Beleza, A.C.S. et al. Cross-cultural adaptation and measurement property analysis of the Brazilian Portuguese version of the Three Incontinence Questionnaire. Int Urogynecol J 33, 3053–3060 (2022). https://doi.org/10.1007/s00192-021-05036-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-021-05036-x