Abstract
Purpose
To compare the effectiveness and safety of intra-articular injections of autologous expanded mesenchymal stromal stem cells alone (MSCs), or in combination with platelet-rich plasma (MSCs + PRP), in patients with knee osteoarthritis.
Methods
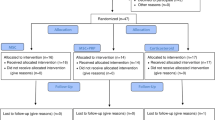
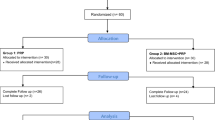
Eighteen patients (57.6 ± 9.6 years) with radiographic symptomatic knee osteoarthritis (Dejour grades II–IV) were randomized to receive intra-articular injections of MSCs (n = 9) or MSCs + PRP (n = 9). Injections were performed 2–3 weeks after bone marrow aspiration (± 80–100 ml) which was obtained from both posterior iliac crests.
Results
The Knee Injury and Osteoarthritis Outcome Score (KOOS) improved significantly throughout the 12 months for both groups (p < 0.05). No statistically significant differences between groups were found in KOOS subscales and global score improvements at 12-month end-point (n.s.). The MSCs group showed significant improvements in the pain, function and daily living activities, and sports and recreational activities subscales (p < 0.05). Similarly, the MSCs + PRP group showed significant improvements in the pain, function and daily living activities and quality of life subscales (p < 0.05). The average number of fibroblast colony forming units (CFU-F) was 56.8 + 21.9 for MSCs group and 50.7 ± 21.7 for MSCs + PRP group. Minimal adverse effects were seen in both groups (10 adverse events, in 5 patients).
Conclusions
Intra-articular injections of expanded MSCs alone or in combination with PRP are safe and have a beneficial effect on symptoms in patients with symptomatic knee osteoarthritis. Adding PRP to the MSCs injections did not provide additional benefit. These results are encouraging and support the recommendation of this minimally invasive procedure in patients with knee osteoarthritis, without requiring hospitalization. The CFU-F results may be used as reference for future research.
Level of evidence
Prospective cohort study, Level II.


Similar content being viewed by others
References
Amable PR, Carias RBV, Teixeira MVT, da Cruz Pacheco Í, do Amaral RJFC., Granjeiro JM et al (2013) Platelet-rich plasma preparation for regenerative medicine: optimization and quantification of cytokines and growth factors. Stem Cell Res Ther 4:67
Bastos Filho R, Magnussen RA, Duthon V, Demey G, Servien E, Granjeiro JM et al (2013) Total knee arthroplasty after high tibial osteotomy: a comparison of opening and closing wedge osteotomy. Int Orthop 37:427–431
Buckwalter JA, Saltzman C, Brown T (2004) The impact of osteoarthritis: implications for research. Clin Orthop Relat Res 427:S6-S15
Campbell KA, Saltzman BM, Mascarenhas R, Khair MM, Verma NN, Bach BR et al (2015) Does intra-articular platelet-rich plasma injection provide clinically superior outcomes compared with other therapies in the treatment of knee osteoarthritis? A systematic review of overlapping meta-analyses. Arthroscopy 31:2213–2221
Caplan AI (2007) Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol 213:341–347
Caplan AI (1991) Mesenchymal stem cells. J Orthop Res 9:641–650
Carstairs A, Genever P (2014) Stem cell treatment for musculoskeletal disease. Curr Opin Pharmacol 16:1–6
da Silva Meirelles L, Sand TT, Harman RJ, Lennon DP, Caplan AI (2008) MSC frequency correlates with blood vessel density in equine adipose tissue. Tissue Eng Part A 15:221–229
Dejour H, Carret J, Walch G (1991) Les Gonarthroses. 7émes Journées Lyonnaises de Chirurgie de Genou. 7émes Journées Lyonnaises de Chirurgie de Genou, Les Gonarthroses
do Amaral RJFC., da Silva NP, Haddad NF, Lopes LS, Ferreira FD, Cappelletti PA et al (2016) Platelet-rich plasma obtained with different anticoagulants and their effect on platelet numbers and mesenchymal stromal cells behavior in vitro. Stem Cells Int. https://doi.org/10.1155/2016/7414036
Foster TE, Puskas BL, Mandelbaum BR, Gerhardt MB, Rodeo SA (2009) Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med 37:2259–2272
Freitag J, Bates D, Boyd R, Shah K, Barnard A, Huguenin L et al (2016) Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety and efficacy—a review. BMC Musculoskelet Disord 17:230
Gomoll A, Filardo G, De Girolamo L, Esprequeira-Mendes J, Marcacci M, Rodkey W et al (2012) Surgical treatment for early osteoarthritis. Part I: cartilage repair procedures. Knee Surg Sports Traumatol Arthrosc 20:450–466
Goncalves R, Cabri J, Pinheiro J, Ferreira P, Gil J (2010) Reliability, validity and responsiveness of the Portuguese version of the Knee injury and Osteoarthritis Outcome Score—Physical Function Short-form (KOOS-PS). Osteoarthritis Cartilage 18:372–376
Goyal D, Keyhani S, Lee EH, Hui JHP (2013) Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy 29:1579–1588
Haas DA, Kaplan RS (2017) Variation in the cost of care for primary total knee arthroplasties. Arthroplast Today 3:33–37
Heijink A, Gomoll AH, Madry H, Drobnič M, Filardo G, Espregueira-Mendes J et al (2012) Biomechanical considerations in the pathogenesis of osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc 20:423–435
Ishiguro N, Kojima T, Poole AR (2002) Mechanism of cartilage destruction in osteoarthritis. Nagoya J Med Sci 65:73–84
Jo CH, Lee YG, Shin WH, Kim H, Chai JW, Jeong EC et al (2014) Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem cells 32:1254–1266
Khoshbin A, Leroux T, Wasserstein D, Marks P, Theodoropoulos J, Ogilvie-Harris D et al (2013) The efficacy of platelet-rich plasma in the treatment of symptomatic knee osteoarthritis: a systematic review with quantitative synthesis. Arthroscopy 29:2037–2048
Koh Y-G, Choi Y-J (2012) Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee 19:902–907
Koh Y-G, Jo S-B, Kwon O-R, Suh D-S, Lee S-W, Park S-H et al (2013) Mesenchymal stem cell injections improve symptoms of knee osteoarthritis. Arthroscopy 29:748–755
Koh Y-G, Kwon O-R, Kim Y-S, Choi Y-J (2014) Comparative outcomes of open-wedge high tibial osteotomy with platelet-rich plasma alone or in combination with mesenchymal stem cell treatment: a prospective study. Arthroscopy 30:1453–1460
Lawrence RC, Felson DT, Helmick CG, Arnold LM, Choi H, Deyo RA et al (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States: Part II. Arthritis Rheum 58:26–35
Lohmander LS, Roos EM (2007) Clinical update: treating osteoarthritis. Lancet 370:2082–2084
Mazor M, Lespessailles E, Coursier R, Daniellou R, Best T, Toumi H (2014) Mesenchymal stem-cell potential in cartilage repair: an update. J Cell Mol Med 18:2340–2350
Meheux CJ, McCulloch PC, Lintner DM, Varner KE, Harris JD (2016) Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy 32:495–505
Mifune Y, Matsumoto T, Takayama K, Ota S, Li H, Meszaros LB et al (2013) The effect of platelet-rich plasma on the regenerative therapy of muscle derived stem cells for articular cartilage repair. Osteoarthritis Cartilage 21:175–185
Mishra A, Tummala P, King A, Lee B, Kraus M, Tse V et al (2009) Buffered platelet-rich plasma enhances mesenchymal stem cell proliferation and chondrogenic differentiation. Tissue Eng Part C Methods 15:431–435
Osborne H, Anderson L, Burt P, Young M, Gerrard D (2015) Australasian College of Sports Physicians—position statement: the place of mesenchymal stem/stromal cell therapies in sport and exercise medicine. Br J Sports Med 50:1237–1244
Owen M, Friedenstein A (1988) Stromal stem cells: marrow-derived osteogenic precursors. Ciba Found Symp 136:42–60
Pas HI, Winters M, Haisma HJ, Koenis MJ, Tol JL, Moen MH (2017) Stem cell injections in knee osteoarthritis: a systematic review of the literature. Br J Sports Med 51:1125–1133
Peeters C, Leijs MJ, Reijman M, van Osch G, Bos P (2013) Safety of intra-articular cell-therapy with culture-expanded stem cells in humans: a systematic literature review. Osteoarthritis Cartilage 21:1465–1473
Pers Y-M, Ruiz M, Noël D, Jorgensen C (2015) Mesenchymal stem cells for the management of inflammation in osteoarthritis: state of the art and perspectives. Osteoarthritis Cartilage 23:2027–2035
Rubio-Azpeitia E, Andia I (2014) Partnership between platelet-rich plasma and mesenchymal stem cells: in vitro experience. Musc Ligaments Tendons J 4:52–62
Saw K-Y, Anz A, Jee CS-Y, Merican S, Ng RC-S, Roohi SA et al (2013) Articular cartilage regeneration with autologous peripheral blood stem cells versus hyaluronic acid: a randomized controlled trial. Arthroscopy 29:684–694
Tan Y, Jiang M, Yu H, Li J, Qing Z (2013) Therapeutic effect of arthroscopy combined with autologous bone marrow stem cell grafting on knee osteoarthritis. J Tradit Chin Orthop Traumatol 10:35–38
Vega A, Martín-Ferrero MA, Del Canto F, Alberca M, García V, Munar A et al (2015) Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation 99:1681–1690
Vinatier C, Mrugala D, Jorgensen C, Guicheux J, Noël D (2009) Cartilage engineering: a crucial combination of cells, biomaterials and biofactors. Trends Biotechnol 27:307–314
Wolfstadt JI, Cole BJ, Ogilvie-Harris DJ, Viswanathan S, Chahal J (2015) Current concepts: the role of mesenchymal stem cells in the management of knee osteoarthritis. Sports Health 7:38–44
Wong KL, Lee KBL, Tai BC, Law P, Lee EH, Hui JH (2013) Injectable cultured bone marrow—derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years’ follow-up. Arthroscopy 29:2020–2028
Xia P, Wang X, Lin Q, Li X (2015) Efficacy of mesenchymal stem cells injection for the management of knee osteoarthritis: a systematic review and meta-analysis. Int Orthop 39:2363–2372
Xie X, Wang Y, Zhao C, Guo S, Liu S, Jia W et al (2012) Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration. Biomaterials 33:7008–7018
Zhang W, Moskowitz R, Nuki G, Abramson S, Altman R, Arden N et al (2008) OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16:137–162
Funding
This study was funded by ESHO Empresa de Serviços Hospitalares S.A. (Grant number: not applicable).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that this study received funding from ESHO Empresa de Serviços Hospitalares S.A. Ricardo Bastos received grants from ESHO Empresa de Serviços Hospitalares S.A. The others authors declare no conflict of interest.
Ethical approval
The research was approved by the Comissão Nacional de Ética em Pesquisa (Brazilian National Ethics in Research Committee)—CONEP, under number 14878813.4.0000.5533 and was conducted in accordance with the principles of the Declaration of Helsinki.
Informed consent
All the participants signed the written consent form before inclusion in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bastos, R., Mathias, M., Andrade, R. et al. Intra-articular injections of expanded mesenchymal stem cells with and without addition of platelet-rich plasma are safe and effective for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc 26, 3342–3350 (2018). https://doi.org/10.1007/s00167-018-4883-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-018-4883-9