Abstract
Objective
Mechanically ventilated patients with status asthmaticus who undergo prolonged paralysis are at risk for severe weakness due to myopathy. In the mid-1990s, we changed our usual method of achieving tolerance of ventilatory support in asthmatic patients from continuous paralysis to deep sedation. This study examines the impact of this change in practice on the development of clinically significant weakness in status asthmaticus.
Design and setting
Retrospective cohort study in university-affiliated county hospital.
Patients
Mechanically ventilated asthmatic patients seen before (n = 96) and after (n = 74) a clinical practice change in 1995 that markedly restricted use of paralytics.
Results
The duration of neuromuscular paralysis declined sharply after 1995 (23.7 ± 42.2 vs. 1.8 ± 4.0 h, P < 0.001), but this was not associated with a significant difference in the incidence of weakness (21 vs. 14%, P = 0.23). Within the post-1995 cohort, there was no significant difference in the duration of paralysis for weak and non-weak patients (3.5 ± 6.2 vs. 1.5 ± 3.5 h, P = 0.10). However, weak patients had a much longer duration of mechanical ventilation than did patients without weakness (11.9 ± 3.6 vs. 1.9 ± 1.8 days, P < 0.001).
Conclusion
Mechanically ventilated patients with status asthmaticus who are immobilized for prolonged periods of time by deep sedation remain at risk for clinically significant weakness.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute myopathy is a known complication of mechanical ventilation for status asthmaticus [1–12]. Several studies, including one by our group [10], have suggested that acute myopathy in status asthmaticus may result from concomitant use of corticosteroids and prolonged neuromuscular paralysis [4–12]. In the mid-1990s, we adopted a policy that encouraged use of deep sedation rather than continuous paralysis to achieve tolerance of ventilatory support, without other substantive changes in management. This study, the preliminary results of which have been presented previously [13], examines the impact of this change in practice on ICU-acquired weakness in status asthmaticus.
Methods
Patient enrollment
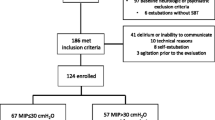
Patients who underwent mechanical ventilation for severe asthma in our medical ICU were divided into two cohorts: pre-1995 (May 1983–May 1995) (n = 96) [10] and post-1995 (May 1995–May 2004) (n = 74). None of the patients had severe sepsis, acute renal failure, multi-organ system failure, end-stage liver disease, or a primary diagnosis of chronic obstructive lung disease. Only the first hospitalization was included for patients who were intubated more than once for severe asthma. The study was approved by our institutional review board.
Clinical management (post-1995)
All patients received intravenous methylprednisolone and inhaled bronchodilators (albuterol, ipratroprium). Usual ventilator settings were a tidal volume of 8–9 ml/kg and a respiratory rate of 10–14 breaths/min [14]. Deep sedation (using combinations of propofol, fentanyl, and lorazepam) was used to suppress respiratory drive until there was evidence for a significant reduction in the severity of airflow obstruction, after which sedation was tapered or discontinued to allow a trial of spontaneous ventilation.
Although prolonged paralysis was discouraged, most patients received succinylcholine at the time of intubation and bolus doses of vecuronium or atracurium were sometimes given while sedation was being escalated. We estimated that each bolus of vecuronium or atracurium would provide approximately 30 min of complete paralysis. Continuous paralysis was reserved for those few patients with fulminant airflow obstruction in whom uninterrupted muscle relaxation was deemed necessary.
Outcome definition
Clinically significant weakness was defined as a generalized reduction in muscle strength involving all four limbs that markedly interfered with activities of daily living, and required inpatient physical rehabilitation and/or home physical therapy after hospital discharge [10]. An electromyogram (EMG) was not required for inclusion in the study.
Statistical analysis
Statistical analysis was performed using SPSS version 11.5 (SPSS, Chicago, IL). Descriptive data are presented as mean ± SD except where noted. The Mann–Whitney U test was used to compare continuous variables as these variables were typically not normally distributed. Categorical variables were compared using the Fisher exact test.
Results
Comparison of pre-1995 and post-1995 cohorts
The pre-1995 and post-1995 cohorts did not differ with respect to age (39 ± 17 vs. 38 ± 13 years), gender (56 vs. 45% male), or duration of mechanical ventilation (3.2 ± 4.0 vs. 3.2 ± 4.0 days) (P > 0.2 for all comparisons). The percentage of patients who received any neuromuscular blocking agent (NMBA) in the pre-1995 and post-1995 time periods was similar (67 vs. 77%, P = 0.17), but the duration of paralysis declined markedly after 1995 (23.7 ± 42.2 vs. 1.8 ± 4.0 h, P < 0.001). The incidence of clinically significant weakness in the pre-1995 and post-1995 cohorts was 20 of 96 (21%) and 10 of 74 (14%), respectively (P = 0.23).
Comparison of weak and non-weak patients in post-1995 cohort
As expected, EMGs revealed myopathy as the cause of muscle weakness in the six patients who underwent electrophysiologic study [10, 15]. Weak and non-weak patients did not differ with regard to age (40 ± 11 vs. 37 ± 13, P = 0.34) or gender (30 vs. 53% male, P = 0.5). There was no significant difference in the amount of sedative used for the two groups on the first hospital day, but the cumulative dose of propofol, lorazepam, and fentanyl over the first 3 days was higher in those patients who became weak since many of the non-weak patients had been extubated by day 3 (Table 1). In addition, the dose of methylprednisolone over the first 3 days was higher in those who developed weakness (Table 1).
The duration of mechanical ventilation was markedly longer for weak patients than for those without weakness (11.9 ± 3.6 vs. 1.9 ± 1.8 days, P < 0.001) (Table 1). Weak patients were intubated for 7–17 days. The mean duration of paralysis was only slightly longer in those who developed weakness, and did not achieve statistical significance (Table 1).
Chart review was used to determine the reason for prolonged mechanical ventilation in the ten weak patients. One patient developed a nosocomial pneumonia (without sepsis syndrome) that contributed to delayed extubation. In the remaining nine cases, protracted status asthmaticus was the dominant reason for prolonged intubation, with 7–12 days having elapsed before the patients respiratory status was sufficiently improved to allow discontinuation of sedation and a spontaneous breathing trial. Delayed awakening after withdrawal of sedation accounted for an additional 1–3 days of intubation. Respiratory muscle weakness did not appear to contribute to delayed extubation, as evidenced by the fact that once underlying respiratory failure had resolved and the patient was sufficiently awake, spontaneous breathing trials were uniformly successful and extubation was accomplished without sequelae (other than a single episode of post-extubation stridor).
Discussion
The principal finding of this study is that mechanically ventilated asthmatic patients who were deeply sedated remained at risk for clinically significant muscle weakness despite a marked reduction in their exposure to neuromuscular paralysis. In the post-1995 cohort, duration of paralysis was similar in weak and non-weak patients. In contrast, the duration of mechanical ventilation was markedly longer for weak patients than for patients without weakness.
Previous studies found an increased risk of acute myopathy in patients with status asthmaticus who underwent prolonged neuromuscular blockade [9–12], but it has been uncertain whether the risk of prolonged paralysis resulted from loss of neuromuscular transmission per se or resultant muscle inactivity. Our findings suggest a role for prolonged muscle inactivity in the development of muscle weakness in status asthmaticus, regardless of whether it is achieved by interruption of neuromuscular transmission or by deep sedation.
The duration of mechanical ventilation was much longer for weak patients than for those without weakness (Table 1), as reported in earlier studies involving patients with asthma or COPD [7, 11, 16]. One possible explanation for this finding is that involvement of the diaphragm could lead to delayed weaning [17, 18]. Alternatively, immobility during prolonged mechanical ventilation could contribute to the development of myopathy. In our previous study, delayed weaning due to respiratory muscle impairment was seen in only 1 of 20 patients [10]. In the present study, prolonged need for mechanical ventilation invariably resulted from slow resolution of airflow obstruction and delayed awakening after use of high-dose benzodiazepine infusions [19]. While the retrospective study design limits our ability to draw firm conclusions regarding the relationship between muscle weakness and prolonged mechanical ventilation, our experience would suggest that in the setting of status asthmaticus, the latter is more likely due to an increased risk of myopathy from prolonged immobility than to delayed weaning from respiratory muscle impairment.
Corticosteroids are believed to be important in the pathogenesis of asthma-related acute myopathy [1–3, 8]. All patients in the current study received relatively high-doses of methylprednisolone, but the amount of corticosteroid given to weak patients was higher than in those without weakness (Table 1). While this likely reflects more severe and persistent airflow obstruction in patients who became weak, a causal role for corticosteroid dose in the development of weakness cannot be excluded. Severe sepsis and multi-organ failure, important risk factors for ICU-acquired weakness in the general critical care population [20], did not occur in the current study and are rare in the setting of status asthmaticus.
There are several important limitations to the present study. First, due to the retrospective and non-randomized study design, we are unable to prove causality between prolonged muscle inactivity and subsequent muscle weakness. Second, the present study dealt specifically with status asthmaticus and its findings may not be applicable to other conditions in which ICU-acquired weakness occurs. Finally, and most importantly, the results of the present study do not negate the risk of prolonged paralysis in the development of muscle weakness in status asthmaticus. This study was not sufficiently powered to compare the relative risk of prolonged paralysis and deep sedation with regard to muscle weakness, nor did we assess its severity. Indeed, restriction of paralysis was associated with a trend towards less weakness that could conceivably have reached statistical significance with a larger study. However, our data confirms that prolonged paralysis is not a prerequisite for development of severe muscle weakness in patients with status asthmaticus, and suggests that there may be a significant risk of weakness when patients are immobilized by deep sedation for a prolonged period of time.
Conclusion
In conclusion, we found that among patients with severe asthma who underwent mechanical ventilation, clinically significant muscle weakness was not eliminated by changing the method of achieving tolerance of ventilatory support from continuous paralysis to prolonged deep sedation. The most important risk factor for weakness was the duration of mechanical ventilation. This suggests that corticosteroid-treated patients with status asthmaticus who are pharmacologically immobilized for prolonged periods of time are at risk for subsequent weakness, regardless of whether their muscles are rendered inactive by chemical denervation or deep sedation.
References
MacFarlane IA, Rosenthal FD (1977) Severe myopathy after status asthmaticus. Lancet 8038:615
Williams TJ, O’Hehir RE, Czarny D, Horne M, Bowes G (1988) Acute myopathy in severe acute asthma treated with intravenously administered corticosteroids. Am Rev Resp Dis 137:460–463
Shee CD (1990) Risk factors for hydrocortisone myopathy in acute severe asthma. Respir Med 84:229–233
Waclawik AJ, Sufit RL, Beinlich BR, Schutta HS (1992) Acute myopathy with selective degeneration of myosin filaments following status asthmaticus treated with methylprednisolone and vecuronium. Neuromuscul Disord 2:19–26
Danon M, Carpenter S (1991) Myopathy with thick filament (myosin) loss following prolonged paralysis with vecuronium during steroid treatment. Muscle Nerve 14:1131–1139
Griffin D, Fairman N, Coursin D, Rawsthorne L, Grossman JE (1992) Acute myopathy during treatment of status asthmaticus with corticosteroids and steroidal muscle relaxants. Chest 102:510–514
Douglass JA, Tuxen DV, Horne M, Scheinkestel CD, Weinmann M, Czarny D, Bowes G (1992) Myopathy in severe asthma. Am Rev Respir Dis 146:517–519
Nates JL, Cooper DJ, Day B, Tuxen DV (1997) Acute weakness syndromes in critically ill patients—a reappraisal. Anaesth Intensive Care 25:502–513
Shapiro JM (1993) Myopathy in status asthmaticus: relation to neuromuscular blockade and corticosteroid administration. J Intensive Care Med 8:144–152
Leatherman J, Fleugel WL, David WS, Davies SF, Iber C (1996) Muscle weakness in mechanically ventilated patients with severe asthma. Am J Respir Crit Care Med 153:1686–1690
Behbehani NA, Al-Mane F, D’yachkova Y, Pare P, Fitzgerald JM (1999) Myopathy following mechanical ventilation for acute severe asthma: The role of muscle relaxants and corticosteroids. Chest 115:1627–1631
Adnet F, Dhissi G, Borron SW, Galinski M, Rayeh F, Cupa M, Pourriat JL, Lapostolle F (2001) Complication profiles of adult asthmatics requiring paralysis during mechanical ventilation. Intensive Care Med 27:1729–1736
Kesler SM, Sprenkle MD, David WS, Leatherman JW (2006) ICU-acquired weakness complicating severe asthma despite limited use of neuromuscular paralysis. Am J Resp Crit Care Med 173:A138
Leatherman JW, McArthur C, Shapiro RS (2004) Effect of prolongation of expiratory time on dynamic hyperinflation in mechanically ventilated patients with severe asthma. Crit Care Med 32:1542–1545
David WS, Roehr CL, Leatherman JW (1998) EMG findings in acute myopathy with status asthmaticus, steroids and paralytics. Clinical and electrophysiologic correlation. Electromyogr Clin Neurophysiol 38:371–376
Amaya-Villar R, Garnacho-Montero J, Garcia-Garmendia JL, Madrazo-Osuna J, Garnacho-Montero MC, Luque R, Ortiz-Leyba C (2005) Steroid-inducted myopathy in patients intubated due to exacerbation of chronic obstructive pulmonary disease. Intensive Care Med 31:157–161
De Jonghe B, Sharshar T, Hopkinson N, Outin H (2004) Does ICU-acquired paresis lengthen weaning from mechanical ventilation? Intensive Care Med 30:1117–1121
DeJonghe B, Bastuji-Garin S, Durand M-C, Mailissin I, Rodriques P, Cerf C, Outin H, Sharshar T, For the Groupe de Reflexion et d’Etude des Neuromyopathies En Reanimation (2007) Respiratory weakness is associated with limb weakness and delayed weaning in critical illness. Crit Care Med 35:2007–2015
Schweickert WD, Gehlbach BK, Pohlman AS, Hall JB, Kress JP (2004) Daily interruption of sedative infusions and complications of critical illness in mechanically ventilated patients. Crit Care Med 32:1272–1276
Stevens RD, Dowdy DD, Michaels RK, Mendez-Tellez PA, Provonost P, Needham D (2007) Neuromuscular dysfunction acquired in critical illness: a systemic review. Intensive Care Med 33:1876–1891
Conflict of interest statement
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kesler, S.M., Sprenkle, M.D., David, W.S. et al. Severe weakness complicating status asthmaticus despite minimal duration of neuromuscular paralysis. Intensive Care Med 35, 157–160 (2009). https://doi.org/10.1007/s00134-008-1267-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1267-5