Abstract
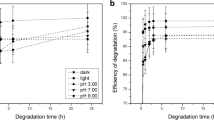
The large-scale use of pesticides is one of the main causes of the dramatic degradation of our environment. Pesticides such as imidacloprid (IMID) have been linked to declines in bee health and toxicity to other beneficial insects. They pose a threat to human health due to their persistence in the environment and accumulation in the food chain. Therefore, it is essential to test possible environmentally-friendly solutions for their elimination. The present study evaluates the efficiency of microalgae Nannochloropsis sp. for the removal of IMID from synthetic wastewater. The influence of aeration, light, and the presence of UV radiation on the degradation of IMID were factors considered in the study. A rapid RP-HPLC method was developed and validated for the analysis and quantification of IMID in the context of bioremediation with microalgae. Nannochloropsis sp. removed 4.39 µg mL−1 from an initial content of 9.59 µg mL−1 (reaching approximately 50%) of IMID in the first 20 h. This study demonstrated that the removal of IMID by the marine microalgae Nannochloropsis sp. is both effective and light-dependent.




Similar content being viewed by others
References
Abinandan S, Subashchandrabose SR, Venkateswarlu K, Megharaj M (2018) Nutrient removal and biomass production: advances in microalgal biotechnology for wastewater treatment. Crit Rev Biotechnol 38:1244
Abinandan S, Subashchandrabose SR, Pannerselvan L et al (2019) Potential of acid-tolerant microalgae, Desmodesmus sp. MAS1 and Heterochlorella sp. MAS3, in heavy metal removal and biodiesel production at acidic pH. Biores Technol. https://doi.org/10.1016/j.biortech.2019.01.053
Acero JL, Real FJ, Javier Benitez F, Matamoros E (2019) Degradation of neonicotinoids by UV irradiation: kinetics and effect of real water constituents. Sep Purif Technol. https://doi.org/10.1016/j.seppur.2018.09.076
Aguilar-Ruiz RJ, Del M, Martínez-Macias R et al (2020) Removal of copper improves the lipid content in Nannochloropsis oculata culture. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-10283-4
Amini M, Amini Khoei Z, Erfanifar E (2019) Nitrate (NO 3–) and phosphate (PO 43– ) removal from aqueous solutions by microalgae Dunaliella salina. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2019.101097
Baskaran S, Kookana RS, Naidu R (1997) Determination of the insecticide imidacloprid in water and soil using high-performance liquid chromatography. J Chromatogr A. https://doi.org/10.1016/S0021-9673(97)00652-3
Bonmatin JM, Giorio C, Girolami V et al (2015) Environmental fate and exposure; neonicotinoids and fipronil. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-014-3332-7
Campos-Mañas MC, Plaza-Bolaños P, Martínez-Piernas AB et al (2019) Determination of pesticide levels in wastewater from an agro-food industry: target, suspect and transformation product analysis. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.05.147
Chen S, Deng J, Deng Y, Gao N (2019) Influencing factors and kinetic studies of imidacloprid degradation by ozonation. Environ Technol (United Kingdom). https://doi.org/10.1080/09593330.2018.14391052018.1439105
Coscollà C, Hart E, Pastor A, Yusà V (2013) LC-MS characterization of contemporary pesticides in PM10 of Valencia Region, Spain. Atmos Environ 77:394–403. https://doi.org/10.1016/j.atmosenv.2013.05.022
Daraghmeh A, Shraim A, Abulhaj S et al (2007) Imidacloprid residues in fruits, vegetables and water samples from Palestine. Environ Geochem Health. https://doi.org/10.1007/s10653-006-9060-2
Doan TY, Obbard JP (2011) Improved Nile Red staining of Nannochloropsis sp. J Appl Phycol. https://doi.org/10.1007/s10811-010-9608-5
Doan TY, Obbard JP (2012) Enhanced intracellular lipid in Nannochloropsis sp. via random mutagenesis and flow cytometric cell sorting. Algal Res. https://doi.org/10.1016/j.algal.2012.03.001
Encarnação T, Arranja CTCT, Cova TFGGTFGG et al (2018) Monitoring oil production for biobased feedstock in the microalga Nannochloropsis sp.: a novel method combining the BODIPY BD-C12 fluorescent probe and simple image processing. J Appl Phycol. https://doi.org/10.1007/s10811-018-1437-y
Encarnação T, Palito C, Pais AACC et al (2020) Removal of pharmaceuticals from water by free and imobilised microalgae. Molecules (Basel, Switzerland). https://doi.org/10.3390/molecules25163639
Eroglu E, Agarwal V, Bradshaw M et al (2012) Nitrate removal from liquid effluents using microalgae immobilized on chitosan nanofiber mats. Green Chem. https://doi.org/10.1039/c2gc35970g
Escapa C, Coimbra RN, Paniagua S et al (2017) Paracetamol and salicylic acid removal from contaminated water by microalgae. J Environ Manage. https://doi.org/10.1016/j.jenvman.2016.06.051
Estrada FGA, Marques JMC, Valente AJM (2019) Molecular dynamics insights for screening the ability of polymers to remove pesticides from water. ChemistryOpen. https://doi.org/10.1002/open.201800293
Food and Drug Administration (FDA) (1994) Reviewer Guidance: Validation of chromatographic methods. Center for Drug Evaluation and Research (CDER)
Food and Drug Administration (FDA) (2001) Guidance for Industry: Bioanalytical method validation. U.S. Department of Health and Human Services, Food and Drug Administration Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM).
Fuentes E, Cid C, Báez ME (2015) Determination of imidacloprid in water samples via photochemically induced fluorescence and second-order multivariate calibration. Talanta. https://doi.org/10.1016/j.talanta.2014.11.0172014.11.017
García-Esquivel Z, Montes-Magallón S, González-Gómez MA (2007) Effect of temperature and photoperiod on the growth, feed consumption, and biochemical content of juvenile green abalone, Haliotis fulgens, fed on a balanced diet. Aquaculture. https://doi.org/10.1016/j.aquaculture.2006.09.0222006.09.022
Genç N, Doğan EC, Narcı AO, Bican E (2017) Multi-response optimization of process parameters for imidacloprid removal by reverse osmosis using taguchi design. Water Environ Res. https://doi.org/10.2175/106143017x14839994523460
Greatti M, Barbattini R, Stravisi A et al (2006) Presence of the a.i. imidacloprid on vegetation near corn fields sown. Bull Insectol 59:99
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate animals. Springer, Boston
Guo L, Dai Z, Guo J et al (2020) Oligotrophic bacterium Hymenobacter latericoloratus CGMCC 16346 degrades the neonicotinoid imidacloprid in surface water. AMB Express. https://doi.org/10.1186/s13568-019-0942-y
Han W, Tian Y, Shen X (2018) Human exposure to neonicotinoid insecticides and the evaluation of their potential toxicity: an overview. Chemosphere 192:59
Hayat W, Zhang Y, Hussain I et al (2019) Efficient degradation of imidacloprid in water through iron activated sodium persulfate. Chem Eng J. https://doi.org/10.1016/j.cej.2019.03.261
Hrybova NYu, Khyzhan OI, Maksin VI et al (2019) Determination of xenobiotic imidacloprid content in surface waters. J Water Chem Technol. https://doi.org/10.3103/s1063455x19050072
Hultberg M, Bodin H, Ardal E, Asp H (2016) Effect of microalgal treatments on pesticides in water. Environ Technol 37:893–898. https://doi.org/10.1080/09593330.2015.1089944
Kandil MM, Trigo C, Koskinen WC, Sadowsky MJ (2015) Isolation and Characterization of a Novel Imidacloprid-Degrading Mycobacterium sp. Strain MK6 from an Egyptian Soil. J Agric Food Chem. https://doi.org/10.1021/acs.jafc.5b00754
Kim HJ, Shelver WL, Li QX (2004) Monoclonal antibody-based enzyme-linked immunosorbent assay for the insecticide imidacloprid. Anal Chim Acta 509:111–118. https://doi.org/10.1016/j.aca.2003.12.006
Liu Z, Tian Y, Zhou X, Liu X, Huang L (2020) Comparison of two different nickel oxide films for electrochemical reduction of imidacloprid. RSC Adv. https://doi.org/10.1039/C9RA09505E
Magnusson B (2014) The fitness for purpose of analytical methods. Eurachem, Teddington
Mallick N, Bagchi SK, Koley S, Singh AK (2016) Progress and challenges in microalgal biodiesel production. Front Microbiol. https://doi.org/10.3389/fmicb.2016.01019
Mohseni A, Kube M, Fan L, Roddick FA (2020) Potential of Chlorella vulgaris and Nannochloropsis salina for nutrient and organic matter removal from municipal wastewater reverse osmosis concentrate. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-09103-6
Morrissey CA, Mineau P, Devries JH et al (2015) Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: a review. Environ Int 74:291
Negi G, Pankaj AS, Sharma A (2014) In situ biodegradation of endosulfan, imidacloprid, and carbendazim using indigenous bacterial cultures of agriculture fields of Uttarakhand, India. Int J Bioeng Life Sci 8:935
Nie J, Sun Y, Zhou Y et al (2020a) Bioremediation of water containing pesticides by microalgae: mechanisms, methods, and prospects for future research. Sci Total Environ 707:136080. https://doi.org/10.1016/j.scitotenv.2019.136080
Nie J, Sun Y, Zhou Y et al (2020b) Bioremediation of water containing pesticides by microalgae: mechanisms, methods, and prospects for future research. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2019.136080
Ochando-Pulido JM, Martinez-Ferez A (2015) On the recent use of membrane technology for olive mill wastewater purification. Membranes 5:513
Patil AL, Patil PN, Gogate PR (2014) Degradation of imidacloprid containing wastewaters using ultrasound based treatment strategies. Ultrason Sonochem. https://doi.org/10.1016/j.ultsonch.2014.02.029
Quinn JC, Yates T, Douglas N et al (2012) Nannochloropsis production metrics in a scalable outdoor photobioreactor for commercial applications. Biores Technol. https://doi.org/10.1016/j.biortech.2012.04.073
Raina-Fulton R (2015) Determination of neonicotinoid insecticides and strobilurin fungicides in particle phase atmospheric samples by liquid chromatography-tandem mass spectrometry. J Agric Food Chem 63:5152–5162. https://doi.org/10.1021/acs.jafc.5b01347
Ramasubramanian T, Paramasivam M, Nirmala R (2016) Development, validation and application of a sensitive analytical method for residue determination and dissipation of imidacloprid in sugarcane under tropical field condition. Environ Monit Assess. https://doi.org/10.1007/s10661-016-5374-4
Sadaria AM, Supowit SD, Halden RU (2016) Mass balance assessment for six neonicotinoid insecticides during conventional wastewater and wetland treatment: nationwide reconnaissance in United States wastewater. Environ Sci Technol. https://doi.org/10.1021/acs.est.6b01032
Sánchez-Bayo F (2014) The trouble with neonicotinoids. Science 346:806
Sharma I, Bhardwaj R, Pati PK (2013) Stress modulation response of 24-epibrassinolide against imidacloprid in an elite indica rice variety Pusa Basmati-1. Pestic Biochem Physiol. https://doi.org/10.1016/j.pestbp.2013.01.0042013.01.004
Taverniers I, De Loose M, Van Bockstaele E (2004) Trends in quality in the analytical laboratory. II. Analytical method validation and quality assurance. Trac-Trends Anal Chem 23:535–552. https://doi.org/10.1016/j.trac.2004.04.001
Ummalyma SB, Pandey A, Sukumaran RK, Sahoo D (2018) Bioremediation by microalgae: current and emerging trends for effluents treatments for value addition of waste streams. Springer, Singapore, pp 355–375
van der Sluijs JP, Vaage NS (2016) Pollinators and Global food security: the need for Holistic Global Stewardship. Food Ethics. https://doi.org/10.1007/s41055-016-0003-z
Wamhoff H, Schneider V (1999) Photodegradation of imidacloprid. J Agric Food Chem. https://doi.org/10.1021/jf980820j
Yari K, Rahmani A, Asgari G et al (2017) Degradation of imidacloprid pesticide in aqueous solution using an eco-friendly electrochemical process. Desalin Water Treat. https://doi.org/10.5004/dwt.2017.21330
Zhou X, Tian Y, Liu X et al (2018) Reduction of imidacloprid by sponge iron and identification of its degradation products. Water Environ Res. https://doi.org/10.2175/106143017x15131012188187
Acknowledgements
The authors acknowledge the Fundação para a Ciência e a Tecnologia (FCT), Portuguese Agency for Scientific Research, for the PhD research grants (SFRH/BD/ 81385/2011) to TE.The authors are also grateful for support from “The Coimbra Chemistry Centre” which is funded by the FCT, through the projects FCT UID/QUI/UI0313/2019, WaterJPI/0006/2016 and COMPETE Programme (Operational Programme for Competitiveness). The authors wish to thank Dr. Abílio Sobral for providing HPLC conditions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Encarnação, T., Santos, D., Ferreira, S. et al. Removal of Imidacloprid from Water by Microalgae Nannochloropsis sp. and Its Determination by a Validated RP-HPLC Method. Bull Environ Contam Toxicol 107, 131–139 (2021). https://doi.org/10.1007/s00128-021-03228-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-021-03228-1