Abstract
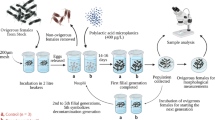
The aim of this study was to determine the median lethal concentration (LC50−96h), effective concentration (EC50−96h), risk assessment, and development of Nile tilapia Oreochromis niloticus larvae submitted to florfenicol (FF) exposure. Fish (n = 147; 8.6 ± 0.6 mg; 7 fish/aquarium) were randomly distributed in 21 aquaria (1 L) and exposed to five concentrations of FF 58.73; 131.31; 198.96; 241.88 and 381.81 mg L−1 plus one control and a control with solvent, totalizing seven treatments and three replicates. The estimated median LC50−96h of FF for Nile tilapia larvae was 349.94 mg L−1. The EC50−96h of FF was 500 mg L−1 for weight reduction and was 1040 mg L−1 for length reduction. After the exposure period, final weight and length differed (p < 0.05) among treatments, showing the lowest biometric values with the highest concentrations of FF. The pH and dissolved oxygen were altered (p < 0.05) during the experimental period. The FF high doses used to determine the LC 50 after 96 h negatively affected the development of the larvae. On the other hand, through risk assessment analysis this antibiotic can be classified as low toxicity to Nile tilapia larvae and show low environmental risk.



Similar content being viewed by others
References
Barreto FM, Silva MR, Braga PAC, Bragotto APA, Hisano H, Reyes FGR (2018) Evaluation of the leaching of florfenicol from coated medicated fish feed into water. Environ Pollut 242:1245–1252. https://doi.org/10.1016/j.envpol.2018.08.017
Becaro AA, Jonsson CM, Puti FC, Siqueira MC, Mattoso LHC, Correa DS, Ferreira MD (2015) Toxicity of PVA-stabilized silver nanoparticles to algae and microcrustaceans. Environ Nanotechnol Monit Manage 3:22–29. https://doi.org/10.1016/j.enmm.2014.11.002
Botelho BG, Christofoletti CA, Correia JE, Ansoar Y, Olinda RA, Tornisielo VL (2015) Genotoxic responses of juvenile tilapia (Oreochromis niloticus) exposed to florfenicol and oxytetracycline. Chemosphere 132:206–212. https://doi.org/10.1016/j.chemosphere.2015.02.053
Cabello FC, Godfrey HP, Buschmann AH, Dölz HJ (2016) Aquaculture as yet another environmental gateway to the development and globalization of antimicrobial resistance. Lancet Infect Dis 16:127–133. https://doi.org/10.1016/S1473-3099(16)00100-6
CEC (2003) Commission of the European Communities—Technical guidance document in support of Commission Directive 93/67/EEC on risk assessment for new notified substances and Commission Regulation (EC) No 1488/94 on risk assessment for existing substances. Office for official publication of the European Communities, Luxembourg, p. 337
Connor SR (1995) SCH 25298: Indirect photolysis screening test with synthetic humic water following EPA/TSCA 40 CFR Ch. 1, 795.70. Schering-Plough Report No. P-6051
Feng JB, Jia XP (2009) Single dose pharmacokinetic study of florfenicol in tilapia (Oreochromis niloticus × O. aureus) held in freshwater at 22 °C. Aquaculture 289:129–133. https://doi.org/10.1016/j.aquaculture.2008.12.023
Ferreira CSG, Nunes BA, José Henriques-Almeida MM, Guilhermino L (2007) Acute toxicity of oxytetracycline and florfenicol to the microalgae Tetraselmis chuii and to the crustacean Artemia parthenogenetica. Ecotoxicol Environ Saf 67:452–458. https://doi.org/10.1016/j.ecoenv.2006.10.006
Food and Agriculture Organization of the United Nations, FAO (2005) Responsible use of antibiotics in aquaculture—meeting the sustainable development goals. FAO, Rome
Gaikowski MP, Wolf JC, Schleis SM, Tuomari D, Endris RG (2012) Safety of florfenicol administered in feed to tilapia (Oreochromis sp.). Toxicol Pathol 41:639–652. https://doi.org/10.1177/0192623312463986
Gaunt PS, Endris R, McGinnis A, Baumgartner W, Camus A, Steadman J, Sweeney D, Sun F (2010) Determination of florfenicol dose rate in feed for control of mortality in Nile tilapia infected with Streptococcus iniae. J Aquat Anim Health 22:158–166. https://doi.org/10.1577/H09-044.1
Hernández-Serrano P (2005) Responsible use of antibiotics in aquaculture. FAO - Fisheries Technical Paper. No. 469. FAO, Rome
Hernando MD, Mezcua M, Fernández-Alba AR, Barceló D (2006) Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 69:334–342. https://doi.org/10.1016/j.talanta.2005.09.037
Horsberg TE, Hoff KE, Nordmo R (1996) Pharmacokinetics of florfenicol and its metabolite florfenicol amine in Atlantic salmon. J Aquat Anim Health 8:292–301. 10.1577/1548-8667(1996)008<0292:POFAIM>2.3.CO;2
LeLievre MK (1991) SCH 25298: acute toxicity to bluegill sunfish (Lepomis macrochirus) under static conditions. Schering-Plough Report No. A-25393
LeLievre MK (1991b) SCH 25298: Acute toxicity to rainbow trout (Oncorhynchus mykiss) under static conditions. Schering-Plough Report No. A-25394
Miranda CD, Rojas R, Abarca A, Hurtado L (2013) Effect of florfenicol and oxytetracycline treatments on the intensive larval culture of the Chilean scallop Argopecten purpuratus (Lamarck, 1819). Aquacult Res 45:16–30. https://doi.org/10.1111/j.1365-2109.2012.03200.x
Nordmo R, Varma KJ, Sutherland IH, Brokken ES (2006) Florfenicol in Atlantic salmon, Salmon salar L.: field evaluation of efficacy against furunculosis in Norway. J Fish Dis 17:239–244. https://doi.org/10.1111/j.1365-2761.1994.tb00219.x
Organization for Economic Co-operation and Development, OECD (1992) Guidelines for the testing of chemicals. Fish Acute Toxicity Test. Adopted test guideline 203. Environment Directorate. OECD, Paris
Organization for Economic Co-operation and Development, OECD (1995) Guidance document for aquatic effects assessment. OECD Environment Monographs 92. OECD, Paris
Paschoal RP, Campana EH, Corrêa LL, Montezzi LF, Barrueto LR, Silva IR, Picão RC (2017) Concentration and variety of carbapenemase producers in recreational coastal waters showing distinct levels of pollution. Antimicrob Agents Chemother 61:1–6. https://doi.org/10.1128/AAC.01963-17
Pavanelli GC, Eiras JC, Takemoto RM (2008) Doenças de peixes: profilaxia, manejo e tratamento. Eduem, 3ed. Maringá. ISBN: 9788576281177
Popma TJ, Lovshin LL (1995) Worldwide prospects for commercial production of Tilapia. Aquaculture production manual. Auburn University, Alabama. International Center for Aquaculture and Aquatic Environments. Accessed http://www.aces.edu/dept/fisheries/aquaculture/docs/worldtilapia.pdf
Pouliquen H, Delépée R, Larhantec-Verdier M, Morvan ML, Bris HL (2007) Comparative hydrolysis and photolysis of four antibacterial agents (oxytetracycline oxolinic acid, flumequine and florfenicol) in deionised water, freshwater and seawater under abiotic conditions. Aquaculture 262:23–28. https://doi.org/10.1016/j.aquaculture.2006.10.014
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Rigos G, Troisi GM (2005) Antibacterial agents in Mediterranean finfish farming: a synopsis of drug pharmacokinetics in important euryhaline fish species and possible environmental implications. Rev Fish Biol Fish 15:53–73. https://doi.org/10.1007/s11160-005-7850-8
Samuelsen OB, Torsvik V, Erik A (1992) Long-range changes in oxytetracycline concentration and bacterial resistance towards oxytetracycline in a fish farm sediment after a medication. Sci Total Environ 114:25–36. https://doi.org/10.1016/0048-9697(92)90411-K
Statpoint Technologies (2014) Statgraphics Centurion XVII (Version 17.1.04). StatPoint Technologies, Herndon
Throne JE, Weaver DK, Chew V, Baker JE (1995) Probit analysis of correlated data: multiple observations over time at one concentration. J Econ Entomol 88:1510–1512. https://doi.org/10.1093/jee/88.5.1510
Tomita RY, Beyruth Z (2002) Toxicologia de agrotóxicos em ambiente aquático. Biológico 64:135–142
World Health Organization, WHO (2006) Antimicrobial use in aquaculture and antimicrobial resistance. Report of a joint FAO/OIE/WHO expert consultation on antimicrobial use in aquaculture and antimicrobial resistance. WHO, Seoul, 13–16 June 2006
Zucker E (1985) Hazard evaluation division—standard evaluation procedure—acute toxicity test for freshwater fish. U.S. Environmental Protection Agency, Office of Pesticide Programs, Washington
Acknowledgements
This research was partially funded by Project BRS Aqua (BNDES, SAP-MAPA, CNPq, FEA and Embrapa). The authors would like to thank Marley Mendonça Tavares (Laboratory of Waste and Contaminants, Embrapa Environment) for water analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Rights and permissions
About this article
Cite this article
Mattioli, C.C., Chiste, B.M., Takeshita, N.A. et al. Acute Toxicity and Risk Assessment of Florfenicol for Nile Tilapia Larvae. Bull Environ Contam Toxicol 105, 721–727 (2020). https://doi.org/10.1007/s00128-020-03013-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-020-03013-6