Abstract
Aims/hypothesis. Glucagon-like peptide-1 ameliorates the symptoms of diabetes through stimulation of insulin secretion and enhancement of beta-cell mass. We have therefore investigated the effects of glucagon-like peptide-1 on the development of diabetes, using db/db mice as a model of Type II diabetes.
Methods. The potent glucagon-like peptide-1 analogue Exendin-4 or vehicle (control) was administered (i.p.; 1 nmol/kg) to obese 6-week old db/db mice daily for 14 days (n=10).
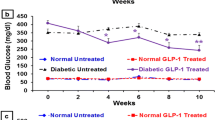
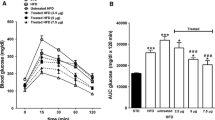
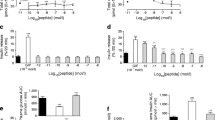
Results. By 8 weeks of age, control db/db mice developed hyperglycaemia (fasting: 10.4±0.5 mmol/l), hyperinsulinaemia and impaired glucose tolerance. However, Exendin-4 treatment prevented hyperglycaemia (fasting: 6.1±1.0 mmol/l, p<0.01), with reduced plasma insulin concentrations (p<0.001) and improved glucose tolerance (p<0.05). Peripheral insulin sensitivity was not affected. However, insulin release in vivo and in vitro from the perfused pancreas was improved by Exendin-4, as were pancreatic insulin concentrations (0.54±0.02 vs 0.32±0.01 µg/mg protein, p<0.05). These changes occurred in conjunction with increased beta-cell mass (3.01±0.31 vs 2.22±0.22 mg, p<0.05) and proliferation (BrdU+ beta-cells: 1.08±0.20 vs 0.47±0.11%, p<0.05), as well as decreased apoptosis (Tunel+ beta-cells: 0.37±0.06 vs 1.20±0.21%). Western blot demonstrated increased expression of Akt1 (by fivefold, p<0.01) and p44 MAP kinase (by sixfold, p<0.01), and decreased activation of caspase-3 (by 30%, p<0.05).
Conclusion/interpretation. Our results suggest that Ex4 treatment delays the onset of diabetes in 6–8 week old db/db mice, through a mechanism involving Akt1 and expansion of the functional beta-cell mass.
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Wang, Q., Brubaker, P. Glucagon-like peptide-1 treatment delays the onset of diabetes in 8 week-old db/db mice. Diabetologia 45, 1263–1273 (2002). https://doi.org/10.1007/s00125-002-0828-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-002-0828-3