Abstract
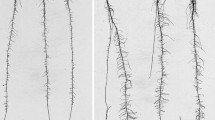
Maize genotypes may adapt to dry environments by avoiding desiccation by means of a deeper root system or by maintaining growth and water extraction at low water potentials. The aim of this study was to determine the quantitative genetic control of root growth and root morphology in a population of 236 recombinant inbred lines (RILs) from the cross between CML444 (high-yielding) × SC-Malawi (low-yielding), which segregates for the response to drought stress at flowering. The RILs and the parental lines were grown on blotting paper in growth pouches until the two-leaf stage under non-stressed conditions; the parents were additionally exposed to desiccation stress induced by polyethylene glycol with a molecular weight of 8000 Dalton (PEG-8000). The lengths of axile and lateral roots were measured non-destructively at 2, 5, 7 and 9 days after germination, by scanning with an A4 scanner followed by digital image analysis. CML444 had a lower rate constant of lateral root elongation (kLat) than SC-Malawi, but the two genotypes did not differ in their response to desiccation. QTLs affecting root vigor, as depicted by increments in kLat, the elongation rate of axile roots (ERAx) and the number of axile roots (NoAx) were identified in bins 2.04 and 2.05. QTLs for NoAx and ERAx collocated with QTLs for yield parameters in bins 1.03–1.04 and 7.03–04. The correspondence of QTLs for axile root traits in bins 1.02–1.03 and 1.08 and QTLs for lateral roots traits in bins 2.04–2.07 in several mapping populations suggests the presence of genes controlling root growth in a wide range of genetic backgrounds.



Similar content being viewed by others
References
Basten CJ, Weir BS, Zeng ZB (2002) QTL CARTOGRAPHER: a reference manual and tutorial for QTL mapping. Department of Statistics. North Carolina State University
Blum A (1985) Breeding crop varieties for stress environments. Crc Rev Plant Sci 2:199–238
Bolaños J, Edmeades GO, Martinez L (1993) Eight cycles of selection for drought tolerance in lowland tropical maize III. Responses in drought-adaptive physiological and morphological traits. Field Crops Res 31:269–286
Bruce WB, Edmeades GO, Baker TC (2002) Molecular and physiological approaches to maize improvement for drought tolerance. J Exp Bot 53:13–25
Butler D (2006) asreml: asreml() fits the linear mixed mode. R package version 2.00
Campos H, Cooper A, Habben JE, Edmeades GO, Schussler JR (2004) Improving drought tolerance in maize: a view from industry. Field Crops Res 90:19–34
Edmeades GO, Bolanos J, Chapman SC, Lafitte HR, Banziger M (1999) Selection improves drought tolerance in tropical maize populations: I. Gains in biomass, grain yield, and harvest index. Crop Sci 39:1306–1315
Enns LC, McCully ME, Canny MJ (2006) Branch roots of young maize seedlings, their production, growth, and phloem supply from the primary root. Funct Plant Biol 33:391–399
Fischer RA, Turner NC (1978) Plant productivity in arid and semi-arid zones. Annu Rev Plant Physiol Plant Mol Biol 29:277–317
Fuad-Hassan A, Tardieu F, Turc O (2008) Drought-induced changes in anthesis–silking interval are related to silk expansion: a spatio-temporal growth analysis in maize plants subjected to soil water deficit. Plant Cell Environ 31:1349–1360
Gallais A, Hirel B (2004) An approach to the genetics of nitrogen use efficiency in maize. J Exp Bot 55:295–306
Giuliani S, Sanguineti MC, Tuberosa R, Bellotti M, Salvi S, Landi P (2005) Root-ABA1, a major constitutive QTL, affects maize root architecture and leaf ABA concentration at different water regimes. J Exp Bot 56:2061–2070
Hetz W, Hochholdinger F, Schwall M, Feix G (1996) Isolation and characterisation of rtcs, a mutant deficient in the formation of nodal roots. Plant J 10:845–857
Hochholdinger F, Feix G (1998) Early post-embryonic root formation is specifically affected in the maize mutant lrtl. Plant J 16:247–255
Hochholdinger F, Tuberosa R (2009) Genetic and genomic dissection of maize root development and architecture. Curr Opin Plant Biol 12:172–177
Hund A, Frachboud Y, Soldati A, Frascaroli E, Salvi S, Stamp P (2004) QTL controlling root and shoot traits of maize seedlings under cold stress. Theor Appl Genet 109:618–629
Hund A, Richner W, Soldati A, Fracheboud Y, Stamp P (2007) Root morphology and photosynthetic performance of maize inbred lines at low temperature. Eur J Agron 27:52–61
Hund A, Ruta N, Liedgens M (2009a) Rooting depth and water use efficiency of tropical maize inbred lines, differing in drought tolerance. Plant Soil 318:311–325
Hund A, Trachsel S, Stamp P (2009b) Growth of axile and lateral roots of maize: I development of a phenotyping platform. Plant Soil. doi:10.1007/s11104-009-9984-2
Kato Y, Abe J, Kamoshita A, Yamagishi J (2006) Genotypic variation in root growth angle in rice (Oryza sativa L.) and its association with deep root development in upland fields with different water regimes. Plant Soil 287:117–129
Lagerwererff J, Eagle HE, Ogata G (1961) Control of osmotic pressure of culture solutions with polyethylene glycol. Science 133:1486–1487
Landi P, Sanguineti MC, Liu C, Li Y, Wang TY, Giuliani S, Bellotti M, Salvi S, Tuberosa R (2007) Root-ABA1 QTL affects root lodging, grain yield, and other agronomic traits in maize grown under well-watered and water-stressed conditions. J Exp Bot 58:319–326
Lawlor DW (1970) Absorption of polyethylene glycols by plants and their effects on plant growth. New Phytol 69:501–513
Lebreton C, Lazic Jancic V, Steed A, Pekic S, Quarrie SA (1995) Identification of QTL for drought responses in maize and their use in testing causal relationships between traits. J Exp Bot 46:853–865
Liu JC, Li JS, Chen FJ, Zhang FS, Ren TH, Zhuang ZJ, Mi GH (2008) Mapping QTLs for root traits under different nitrate levels at the seedling stage in maize (Zea mays L.). Plant Soil 305:253–265
Lorens GF, Bennett JM, Loggale LB (1987) Differences in drought resistance between 2 corn hybrids.1. Water relations and root length density. Agr J 79:802–807
Ludlow MM, Muchow RC (1990) A critical-evaluation of traits for improving crop yields in water-limited environments. Adv Agr 43:107–153
McCully ME (1999) Roots in soil: unearthing the complexities of roots and their rhizospheres. Annu Rev Plant Physiol Plant Mol Biol 50:695–718
Messmer R (2006) The genetic dissection of key factors involved in the drought tolerance of tropical maize (Zea mays L.). Diss. ETH No. 16695, Zurich, Switzerland. http://e-collection.ethbib.ethz.ch/show?type=diss&nr=16695
Messmer R, Fracheboud Y, Banziger M, Vargas M, Stamp P, Ribaut JM (2009) Drought stress and tropical maize: QTL-by-environment interactions and stability of QTLs across environments for yield components and secondary traits. Theor Appl Genet 119:913–930
Money NP (1989) Osmotic-pressure of aqueous polyethylene glycols—relationship between molecular-weight and vapor-pressure deficit. Plant Physiol 91:766–769
Nayyar H, Gupta D (2006) Differential sensitivity of C-3 and C-4 plants to water deficit stress: association with oxidative stress and antioxidants. Env Exp Bot 58:106–113
Oertli JJ (1985) The response of plant-cells to different forms of moisture stress. J Plant Physiol 121:295–300
Paponov IA, Sambo P, Erley GSA, Presterl T, Geiger HH, Engels C (2005) Grain yield and kernel weight of two maize genotypes differing in nitrogen use efficiency at various levels of nitrogen and carbohydrate availability during flowering and grain filling. Plant Soil 272:111–123
Price AH, Steele KA, Moore BJ, Jones RGW (2002) Upland rice grown in soil-filled chambers and exposed to contrasting water-deficit regimes II. Mapping quantitative trait loci for root morphology and distribution. Field Crops Res 76:25–43
Pritchard SG, Rogers HH (2000) Spatial and temporal deployment of crop roots in CO2-enriched environments. New Phytol 147:55–71
Rajcan I, Tollenaar M (1999) Source: sink ratio and leaf senescence in maize: II. Nitrogen metabolism during grain filling. Field Crops Res 60:255–265
R Development Core Team (2004) R: A language and environment for statistical computing. http://www.R-project.org. R Foundation for Statistical Computing, Vienna, Austria
Ribaut JM, Ragot M (2007) Marker-assisted selection to improve drought adaptation in maize: the backcross approach, perspectives, limitations, and alternatives. J Exp Bot 58:351–360
Ribaut JM, Betràn FJ, Monneveux P, Setter T (2008) Drought tolerance in maize. In: Hake SC, Bennetzen JL (eds) Hand book of maize: its biology. Springer, Netherlands, pp 311–344
Sanguineti MC, Duvick DN, Smith S, Landi P, Tuberosa R (2006) Effects of long-term selection on seedling traits and ABA accumulation in commercial maize hybrids. Maydica 51:329–338
Scholander PF, Bradstreet ED, Hemmingsen EA (1965) Sap pressure in vascular plants. Science 148:339–346
Sharp RE, Davies WJ (1985) Root-growth and water-uptake by maize plants in drying soil. J Exp Bot 36:1441–1456
Sharp RE, Poroyko V, Hejlek LG, Spollen WG, Springer GK, Bohnert HJ, Nguyen HT (2004) Root growth maintenance during water deficits: physiology to functional genomics. J Exp Bot 55:2343–2351
Shen L, Courtois B, McNally KL, Robin S, Li Z (2001) Evaluation of near-isogenic lines of rice introgressed with QTLs for root depth through marker-aided selection. Theor Appl Genet 103:75–83
Steele KA, Price AH, Shashidhar HE, Witcombe JR (2006) Marker-assisted selection to introgress rice QTLs controlling root traits into an Indian upland rice variety. Theor Appl Genet 112:208–221
Steudle E (2000) Water uptake by roots: effects of water deficit. J Exp Bot 51:1531–1542
Taiz L, Zeiger E, Taiz L, Zeiger E (2002) Plant physiology, 3rd edn, vol I-xxvi. pp 1–690
Taromina G, Sauer M, Stauffer JL, Multani D, Niu X, Sakai H, Hochholdinger F (2007) The maize (Zea mays L.) RTCS gene encodes a LOB domain protein that is a key regulator of embryonic seminal and post-embryonic shoot-borne root initiation. Plant J 50:649–659
Tuberosa R, Sanguineti MC, Landi P, Salvi S, Casarini E, Conti S (1998) RFLP mapping of quantitative trait loci controlling abscisic acid concentration in leaves of drought stressed maize (Zea mays L.)
Tuberosa R, Salvi S, Sanguineti MC, Landi P, Maccaferri M, Conti S (2002) Mapping QTLs regulating morpho-physiological traits and yield: case studies, shortcomings and perspectives in drought-stressed maize. Ann Bot 89:941–963
van der Weele CM, Spollen WG, Sharp RE, Baskin TI (2000) Growth of Arabidopsis thaliana seedlings under water deficit studied by control of water potential in nutrient-agar media. J Exp Bot 51:1555–1562
Verslues PA, Agarwal M, Katiyar-Agarwal S, Zhu J, Zhu JK (2006) Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status. Plant J 45:523–539
Wan CG, Xu WW, Sosebee RE, Machado S, Archer T (2000) Hydraulic lift in drought-tolerant and -susceptible maize hybrids. Plant Soil 219:117–126
Wang GL, Kang MS, Moreno O (1999) Genetic analyses of grain-filling rate and duration in maize. Field Crops Res 61:211–222
Zhu JM, Kaeppler SM, Lynch JP (2005a) Mapping of QTL controlling root hair length in maize (Zea mays L.) under phosphorus deficiency. Plant Soil 270:299–310
Zhu JM, Kaeppler SM, Lynch JP (2005b) Mapping of QTLs for lateral root branching and length in maize (Zea mays L.) under differential phosphorus supply. Theor Appl Genet 111:688–695
Acknowledgments
The authors would like to thank Dr. Yunbi Xu for supplying the mapping population, Susanne Hochmann and David Brändli for their technical assistance, Jann Röder for programming the scanning software and Cairsty Grassie for linguistic corrections on the manuscript. We would also like to thank two anonymous reviewers for their valuable comments for the improvement of the manuscript. This study was supported by the Generation Challenge Programme (Project 15).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Y. Xu.
Rights and permissions
About this article
Cite this article
Trachsel, S., Messmer, R., Stamp, P. et al. Mapping of QTLs for lateral and axile root growth of tropical maize. Theor Appl Genet 119, 1413–1424 (2009). https://doi.org/10.1007/s00122-009-1144-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-009-1144-9