Abstract
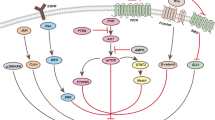
Searching for potential anticancer agents from natural sources is an effective strategy for developing novel chemotherapeutic agents. In this study, data supporting the in vitro and in vivo anticancer effects of delicaflavone, a rarely occurring biflavonoid from Selaginella doederleinii, were reported. Delicaflavone exhibited favorable anticancer properties, as shown by the MTT assay and xenograft model of human non-small cell lung cancer in male BALB/c nude mice without observable adverse effect. By transmission electron microscopy with acridine orange and Cyto-ID®Autophagy detection dyes, Western blot analysis, and RT-PCR assay, we confirmed that delicaflavone induces autophagic cell death by increasing the ratio of LC3-II to LC3-I, which are autophagy-related proteins, and promoting the generation of acidic vesicular organelles and autolysosomes in the cytoplasm of human lung cancer A549 and PC-9 cells in a time- and dose-dependent manner. Delicaflavone downregulated the expression of phospho-Akt, phospho-mTOR, and phospho-p70S6K in a time- and dose-dependent manner, suggesting that it induced autophagy by inhibiting the Akt/mTOR/p70S6K pathway in A549 and PC-9 cells. Delicaflavone is a potential anticancer agent that can induce autophagic cell death in human non-small cell lung cancer via the Akt/mTOR/p70S6K signaling pathway. Delicaflavone showed anti-lung cancer effects in vitro and in vivo. Delicaflavone induced autophagic cell death via Akt/mTOR/p70S6K signaling pathway. Delicaflavone did not show observable side effects in a xenograft mouse model. Delicaflavone may represent a potential therapeutic agent for lung cancer.
Key messages
-
Delicaflavone showed anti-lung cancer effects in vitro and in vivo.
-
Delicaflavone induced autophagic cell death via Akt/mTOR/p70S6K signaling pathway.
-
Delicaflavone did not show observable side effects in a xenograft mouse model.
-
Delicaflavone may represent a potential therapeutic agent for lung cancer.








Similar content being viewed by others
References
Jemal A, Center MM, De Santis C, Ward EM (2010) Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol Biomark Prev 19:1893–1907
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451:1069–1075
Rubinsztein DC (2006) The roles of intracellular protein degradation pathways in neurodegeneration. Nature 443:780–786
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873
Anding AL, Baehrecke EH (2015) Autophagy in cell life and cell death. Curr Top Dev Biol 114:67–91
Denton D, Xu TQ, Kumar S (2015) Autophagy as a pro-death pathway. Immunol Cell Biol 93:35–42
Nelson C, Baehrecke EH (2014) Eaten to death. FEBS J 281:5411–5417
Lum JJ, Bauer DE, Kong M, Harris MH, Li C, Lindsten T, Thompson CB (2005) Growth factor regulation of autophagy and cell survival in the absence of apoptosis. Cell 120:237–248
Morgan TM, Koreckij TD, Corey E (2009) Targeted therapy for advanced prostate cancer: inhibition of the PI3K/Akt/mTOR pathway. Curr Cancer Drug Targets 9:237–249
Downward J (2004) PI 3-kinase, Akt and cell survival. Semin Cell Dev Biol 15:177–182
Liu P, Cheng H, Roberts TM, Zhao JJ (2009) Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov 8:627–644
Xu G, Zhang W, Bertram P, Zheng XF, McLeod H (2004) Pharmacogenomic profiling of the PI3K/PTEN-AKT-mTOR pathway in common human tumors. Int J Oncol 24:893–900
Kim KW, Mutter RW, Cao C, Albert JM, Freeman M, Hallahan DE, Lu B (2006) Autophagy for cancer therapy through inhibition of pro-apoptotic proteins and mammalian target of rapamycin signaling. J Biol Chem 281:36883–36890
Sui YX, Li SG, Shi PY, Wu YJ, Li YX, Chen WY, Huang LY, Yao H, Lin X (2016) Ethyl acetate extract from Selaginella doederleinii Hieron inhibits the growth of human lung cancer cells A549 via caspase-dependent apoptosis pathway. J Ethnopharmacol 190:261–271
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Chan LL, Shen D, Wilkinson AR, Patton W, Lai N, Chan E, Kuksin D, Lin B, Qiu J (2012) A novel image-based cytometry method for autophagy detection in living cells. Autophagy 8:1371–1382
Oeste CL, Seco E, Patton WF, Boya P, Perez-Sala D (2013) Interactions between autophagic and endo-lysosomal markers in endothelial cells. Histochem Cell Biol 139:659–670
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36:2503–2518
Kabeya Y, Mizushima N, Yamamoto A, Oshitani-Okamoto S, Ohsumi Y, Yoshimori T (2004) LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci 117:2805–2812
Liang C, Feng P, Ku B, Dotan I, Canaani D, Oh BH (2006) Autophagic and tumour suppressor activity of a novel Beclin1-binding protein UVRAG. Nat Cell Biol 8:688–699
Wu J, Dang Y, Su W, Liu C, Ma H, Shan Y, Pei Y, Wan B, Guo J, Yu L (2006) Molecular cloning and characterization of rat LC3A and LC3B-- two novel markers of autophagosome. Biochem Bioph Res Commun 339:437–442
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Potze L, Mullauer FB, Colak S, Kessler JH, Medema JP (2014) Betulinic acid-induced mitochondria-dependent cell death is counterbalanced by an autophagic salvage response. Cell Death Dis 5:e1169
Blommaart EF, Luiken JJ, Blommaart PJ, van Woerkom GM, Meijer AJ (1995) Phosphorylation of ribosomal protein S6 is inhibitory for autophagy in isolated rat hepatocytes. J Biol Chem 270:2320–2326
Arico S, Petiot A, Bauvy C, Dubbelhuis PF, Meijer AJ, Codogno P (2001) The tumor suppressor PTEN positively regulates macroautophagy by inhibiting the phosphatidylinositol 3-kinase/protein kinase B pathway. J Biol Chem 276:35243–35246
Shigemitsu K, Tsujishita Y, Hara K, Nanahoshi M, Avruch J, Yonezawa K (1999) Regulation of translational effectors by amino acid and mammalian target of rapamycin signaling pathways. Possible involvement of autophagy in cultured hepatoma cells. J Biol Chem 274:1058–1065
Bursch W, Ellinger A, Gerner C, Frohwein U, Schulte-Hermann R (2000) Programmed cell death (PCD). Apoptosis, autophagic PCD, or others? Ann N Y Acad Sci 926:1–12
Corcelle EA, Puustinen P, Jaattele M (2009) Apoptosis and autophagy: targeting autophagy signaling in cancer cells-‘trick or treat’? FEBS J 276:6084–6096
Chiu W, Chen C-H, Chang J-N, Chen C-H, Hsu Y-H (2016) Far-infrared promotes burn wound healing by suppressing NLRP3 inflammasome caused by enhanced autophagy. J Mol Med 94:809–819
Suzuki SW, Yamamoto H, Oikawa Y, Kondo-Kakuta C, Kimura Y, Hirano H, Ohsumi Y (2015) Atg13 HORMA domain recruits Atg9 vesicles during autophagosome formation. PNAS 112:3350–3355
Yang Y, Li Y, Chen X, Cheng X, Liao Y, Yu X (2016) Exosomal transfer of miR-30a between cardiomyocytes regulates autophagy after hypoxia. J Mol Med 94:711–724
Mochida K, Oikawa Y, Kimura Y, Kirisako H, Hirano H, Ohsumi Y, Nakatogawa H (2015) Receptor-mediated selective autophagy degrades the endoplasmic reticulum and the nucleus. Nature 522:359–362
Liu H, He Z, Simon H-U (2015) Protective role of autophagy and autophagy-related protein 5 in early tumorigenesis. J Mol Med 93:159–164
Yang Z, Goronzy JJ, Weyand CM (2015) Autophagy in autoimmune disease. J Mol Med 93:707–717
Degenhardt K, Mathew R, Beaudoin B, Bray K, Anderson D, Chen G, Mukherjee C, Shi Y, Gélinas C, Fan Y et al (2006) Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 10:51–64
Chitra P, Saiprasad G, Manikandan R, Sudhandiran G (2015) Berberine inhibits Smad and non-Smad signaling cascades and enhances autophagy against pulmonary fibrosis. J Mol Med 93:1015–1031
Yu T, Zuber J, Li J (2015) Targeting autophagy in skin diseases. J Mol Med 93:31–38
Wang Y, Gao J, Zhang D, Zhang J, Ma J, Jiang H (2010) New insights into the antifibrotic effects of sorafenib on hepatic stellate cells and liver fibrosis. J Hepatol 53:132–144
Martelli AM, Evangelisti C, Chiarini F, Grimaldi C, Cappellini A, Ognibene A, McCubrey JA (2010) The emerging role of the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin signaling network in normal myelopoiesis and leukemogenesis. Biochim Biophys Acta 1803:991–1002
Acknowledgements
This work was supported by the National Nature Science Foundation of China (No. 21275028, 81303298, and 81202987), the Fujian Provincial Natural Science Foundation (No. 2016J01371), Program for New Century Excellent Talents in Fujian Province University (No.JA14128), Training project of young talents in health system of Fujian Province (No. 2016-ZQN-63), and the Fujian Agriculture and Forestry University Foundation for excellent youth teachers (No. xjq201414).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The animals were handled according to the guidelines of the China Animal Welfare Legislation, as provided by the Committee on Ethics in the Care and Use of Laboratory Animals of Fujian Medical University.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Yuxia Sui and Hong Yao are contributed equally to this work.
Electronic supplementary material
ESM 1
(PDF 545 kb)
Rights and permissions
About this article
Cite this article
Sui, Y., Yao, H., Li, S. et al. Delicaflavone induces autophagic cell death in lung cancer via Akt/mTOR/p70S6K signaling pathway. J Mol Med 95, 311–322 (2017). https://doi.org/10.1007/s00109-016-1487-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-016-1487-z