Abstract
Purpose
Stereotactic body radiotherapy (SBRT) is a treatment option for early-stage lung cancer. We aimed to examine the differences in failure patterns after SBRT according to the clinical T stage.
Methods
A total of 120 patients with early-stage lung cancer (T1-3N0M0) who underwent SBRT were analysed. The clinical stage in patients whose tumours were in contact with the chest wall was confirmed using four-dimensional computed tomography (4D-CT). Local failure, regional node metastasis, and distant metastasis were confirmed from clinical charts.
Results
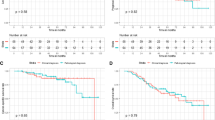
Median follow-up time was 27.5 months (range 7–122) after SBRT. Thirteen patients were restaged from clinical T2 with visceral pleural invasion to T3 with chest wall invasion using 4D-CT analysis. Thirty-seven patients developed recurrences. The median progression-free survival (PFS) and overall survival (OS) were 38.1 and 53.8 months, respectively. The 3‑year PFS and OS rates were 50.7% and 60.3%, respectively. A significant difference was observed in PFS according to the clinical T stage (p = 0.001). No significant differences were observed in OS according to the clinical T stage (p = 0.213). The proportion of locoregional failures relative to distant metastasis decreased with progression from T1 to T3. The pleural dissemination rate was significantly higher in T3 tumours than in T1 and T2 tumours (p = 0.010).
Conclusion
Clinical T stage is associated with PFS after SBRT for lung cancer. There were differences in the failure patterns according to T stage. 4D-CT might provide significant information for assessing chest wall invasion associated with unfavourable PFS.



Similar content being viewed by others
References
National Comprehensive Cancer Network (2022) NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Non-Small Cell Lung Cancer Version 3. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf. Accessed 28 Mar 2022
Nagata Y, Kimura T (2018) Stereotactic body radiotherapy (SBRT) for Stage I lung cancer. Jpn J Clin Oncol 48:405–409
Tandberg DJ, Tong BC, Ackerson BG, Kelsey CR (2018) Surgery versus stereotactic body radiation therapy for stage I non-small cell lung cancer: A comprehensive review. Cancer 124:667–678
Chi A, Fang W, Sun Y, Wen S (2019) Comparison of long-term survival of patients with early-stage non-small cell lung cancer after surgery vs stereotactic body radiotherapy. JAMA Netw Open 2:e1915724
Chang JY, Senan S, Paul MA et al (2015) Stereotactic ablative radiotherapy versus lobectomy for operable stage I non-small-cell lung cancer: a pooled analysis of two randomised trials [published correction appears in Lancet Oncol. 2015 Sep;16(9):e427]. Lancet Oncol 16:630–637
Chang JY, Mehran RJ, Feng L et al (2021) Stereotactic ablative radiotherapy for operable stage I non-small-cell lung cancer (revised STARS): long-term results of a single-arm, prospective trial with prespecified comparison to surgery. Lancet Oncol 22:1448–1457
Stokes WA, Bronsert MR, Meguid RA et al (2018) Post-treatment mortality after surgery and stereotactic body radiotherapy for early-stage non-small-cell lung cancer. J Clin Oncol 36:642–651
Slotman BJ, Lagerwaard FJ, Senan S (2006) 4D imaging for target definition in stereotactic radiotherapy for lung cancer. Acta Oncol 45:966–972
Hof H, Rhein B, Haering P et al (2009) 4D-CT-based target volume definition in stereotactic radiotherapy of lung tumours: comparison with a conventional technique using individual margins. Radiother Oncol 93:419–423
Qi Y, Li J, Zhang Y et al (2021) Effect of abdominal compression on target movement and extension of the external boundary of peripheral lung tumours treated with stereotactic radiotherapy based on four-dimensional computed tomography. Radiat Oncol 16:173
Choong CK, Pasricha SS, Li X et al (2015) Dynamic four-dimensional computed tomography for preoperative assessment of lung cancer invasion into adjacent structures. Eur J Cardiothorac Surg 47:239–243
Bai JH, Hsieh MS, Liao HC, Lin MW, Chen JS (2019) Prediction of pleural invasion using different imaging tools in non-small cell lung cancer. Ann Transl Med 7:33
Mizuno K, Muto M (2021) Preoperative evaluation of pleural adhesion in patients with lung tumors using four-dimensional computed tomography performed during natural breathing. Medicine 100:e27800
American Association of Physicists in Medicine (2016) Adult Routine Chest CT Protocols Version 2.1 5/4/2016. https://www.aapm.org/pubs/CTProtocols/documents/AdultRoutineChestCT.pdf. Accessed 24 May 2022
Hamamoto Y, Kataoka M, Yamashita M et al (2012) Lung-cancer related chest events detected by periodical follow-up CT after stereotactic body radiotherapy for stage I primary lung cancer: retrospective analysis of incidence of lung-cancer related chest events and outcomes of salvage treatment. Jpn J Radiol 30:671–675
Brooks ED, Sun B, Feng L et al (2018) Association of long-term outcomes and survival with multidisciplinary salvage treatment for local and regional recurrence after stereotactic ablative radiotherapy for early-stage lung cancer. JAMA Netw Open 1:e181390
Guckenberger M, Andratschke N, Alheit H et al (2014) Definition of stereotactic body radiotherapy: principles and practice for the treatment of stage I non-small cell lung cancer. Strahlenther Onkol 190:26–33
Brierley JD, Gospodarowicz MK, Wittekind CH (2017) TNM classification of malignant tumours, 8th edn. Wiley-Blackwell, Hoboken
Inagaki T, Doi H, Ishida N et al (2022) Escalated maximum dose in the planning target volume improves local control in stereotactic body radiation therapy for T1–2 lung cancer. Cancers 14:933
Kimura T, Nagata Y, Eba J et al (2017) A randomized Phase III trial of comparing two dose-fractionations stereotactic body radiotherapy (SBRT) for medically inoperable Stage IA non-small cell lung cancer or small lung lesions clinically diagnosed as primary lung cancer: Japan Clinical Oncology Group Study JCOG1408 (J-SBRT trial). Jpn J Clin Oncol 47:277–281
Palma DA, Olson R, Harrow S et al (2019) Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet 393:2051–2058
Nagata Y, Hiraoka M, Shibata T et al (2015) Prospective trial of stereotactic body radiation therapy for both operable and inoperable T1N0M0 non-small cell lung cancer: Japan Clinical Oncology Group Study JCOG0403. Int J Radiat Oncol Biol Phys 93:989–996
Hörner-Rieber J, Bernhardt D, Blanck O et al (2019) Long-term follow-up and patterns of recurrence of patients with oligometastatic NSCLC treated with pulmonary SBRT. Clin Lung Cancer 20:e667–e677
Robinson CG, DeWees TA, El Naqa IM et al (2013) Patterns of failure after stereotactic body radiation therapy or lobar resection for clinical stage I non-small-cell lung cancer [published correction appears in J Thorac Oncol. 2013 Oct;8(10):1343]. J Thorac Oncol 8:192–201
Timmerman RD, Hu C, Michalski JM et al (2018) Long-term results of stereotactic body radiation therapy in medically inoperable stage I non-small cell lung cancer. JAMA Oncol 4:1287–1288
Klement RJ, Abbasi-Senger N, Adebahr S et al (2019) The impact of local control on overall survival after stereotactic body radiotherapy for liver and lung metastases from colorectal cancer: a combined analysis of 388 patients with 500 metastases. BMC Cancer 19:173
Kato H, Ichinose Y, Ohta M et al (2004) A randomized trial of adjuvant chemotherapy with uracil-tegafur for adenocarcinoma of the lung. N Engl J Med 350:1713–1721
Huh SJ, Nishimura T, Park W, Onishi H, Ahn YC, Nakamura K (2020) Current status and comparison of national health insurance systems for advanced radiation technologies in Korea and Japan. Radiat Oncol J 38:170–175
Eriguchi T, Takeda A, Sanuki N et al (2016) Stereotactic body radiotherapy for T3 and T4N0M0 non-small cell lung cancer. J Radiat Res 57:265–272
Narita A, Takeda A, Eriguchi T et al (2019) Stereotactic body radiotherapy for primary non-small cell lung cancer patients with clinical T3-4N0M0 (UICC 8th edition): outcomes and patterns of failure. J Radiat Res 60:639–649
Takeda AH, Watanabe Y, Nagata T et al (2018) Detection of alternative subpleural lymph flow pathways using indocyanine green fluorescence. Surg Today 48:640–648
Drake JA, Sullivan JL, Weksler B (2018) Adjuvant chemotherapy improves survival in patients with completely resected T3N0 non-small cell lung cancer invading the chest wall. J Thorac Cardiovasc Surg 155:1794–1802
Ahmad U, Crabtree TD, Patel AP et al (2017) Adjuvant chemotherapy is associated with improved survival in locally invasive node negative non-small cell lung cancer. Ann Thorac Surg 104:303–307
Verma V, McMillan MT, Grover S, Simone CB 2nd (2017) Stereotactic body radiation therapy and the influence of chemotherapy on overall survival for large (≥5 centimeter) non-small cell lung cancer. Int J Radiat Oncol Biol Phys 97:146–154
Kawaguchi K, Mori S, Usami N et al (2009) Preoperative evaluation of the depth of chest wall invasion and the extent of combined resections in lung cancer patients. Lung Cancer 64:41–44
Wilke L, Andratschke N, Blanck O et al (2019) ICRU Report 91 on prescribing, recording, and reporting of stereotactic treatments with small photon beams: Statement from the DEGRO/DGMP working group stereotactic radiotherapy and radiosurgery. Strahlenther Onkol 195:193–198
Mauguen A, Pignon JP, Burdett S et al (2013) Surrogate endpoints for overall survival in chemotherapy and radiotherapy trials in operable and locally advanced lung cancer: a re-analysis of meta-analyses of individual patients’ data. Lancet Oncol 14:619–626
Acknowledgements
We thank Mr. Kenji Matsumoto from the Department of Central Radiology, Kindai University Hospital.
Funding
This work was supported by JSPS KAKENHI (grant number JP20K08093).
Author information
Authors and Affiliations
Contributions
TI and HD conceived this study, analysed the data and drafted the manuscript. TI, HD, MI, NI, AR, ST, YW, TU, KN, MH and YN performed radiotherapy. TI and MI collected the data. YN supervised the project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
T. Inagaki, H. Doi, M. Inada, N. Ishida, A. Ri, S. Tatsuno, Y. Wada, T. Uehara, K. Nakamatsu, M. Hosono and Y. Nishimura declare that they have no competing interests.
Ethical standards
This study was conducted in accordance with the guidelines of the Declaration of Helsinki. This study was approved by our institutional review board (approval number R03-116). Consent to participate: Written informed consent for radiotherapy was obtained from all individual participants prior to radiotherapy. Informed consent for this study was obtained in the form of opt-out. Consent to publish: Not applicable.
Additional information
Data availability
The data are available from the corresponding author upon reasonable request.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Inagaki, T., Doi, H., Inada, M. et al. Difference in failure patterns after stereotactic body radiotherapy for lung cancer according to clinical T stage based on 4D computed tomography. Strahlenther Onkol 199, 465–476 (2023). https://doi.org/10.1007/s00066-022-02030-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-022-02030-0