Abstract.
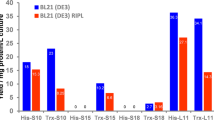
An efficient Escherichia coli expression system for the production of a perchloric acid-soluble protein (PSP) has been constructed. Complementary DNA encoding PSP was inserted into an inducible bacterial expression vector pGEX-4T-1. After the plasmid introduced into E. coli was expressed by isopropyl 1-thio-β-D-galactopyranoside (IPTG), the recombinant product was purified by glutathione-Sepharose 4B affinity chromatography. The purified product showed the expected NH2-terminal sequence, but the translation inhibitory activity of this product was 10 times lower compared with that of authentic PSP isolated from rat liver.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 8 October 1998; received after revision 6 November 1998; accepted 6 November 1998
Rights and permissions
About this article
Cite this article
Oka, T., Nishimoto, Y., Sasagawa, T. et al. Production of functional rat liver PSP protein in Escherichia coli. CMLS, Cell. Mol. Life Sci. 55, 131–134 (1999). https://doi.org/10.1007/s000180050277
Published:
Issue Date:
DOI: https://doi.org/10.1007/s000180050277