Abstract
Objective
To evaluate the regulatory effects of honokiol on high-glucose (HG)-induced inflammatory responses of human renal mesangial cells (HRMCs).
Materials and methods
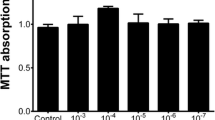
We performed MTS assays to determine the non-cytotoxic concentration of honokiol for HRMCs. Enzyme-linked immunosorbent assays were performed to analyze the expressions of the proteins interleukin (IL)-1β, IL-18, tumor necrosis factor (TNF)-α, transforming growth factor (TGF)-β1, monocyte chemoattractant protein (MCP)-1, macrophage inflammatory protein (MIP)-1α, RANTES, and prostaglandin (PG) E2. The total nitric oxide (NO) concentration was determined using the Griess reaction.
Results
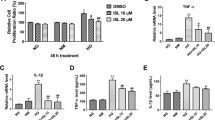
Treatment with 50 mmol/L glucose markedly increased the level of IL-1β, IL-18, TNF-α, PGE2, NO, TGF-β1, MCP-1, MIP-1α, and RANTES. Honokiol (~20 μmol/L) treatment inhibited the HG-induced expression of inflammatory cytokines such as IL-1β, IL-18, TNF-α, PGE2, NO, and TGF-β1 in a dose-dependent manner. Moreover, it markedly inhibited the expression of chemokines such as MCP-1, MIP-1α, and RANTES, which are upregulated under HG conditions.
Conclusion
Honokiol inhibits the HG-induced expression of inflammatory factors in HRMCs. Honokiol may be considered a promising drug with potent anti-inflammatory activities in addition to its strong anti-cancer, anti-angiogenesis, and anti-neurodegenerative effects.


Similar content being viewed by others
References
Navarro JF, Mora C. Role of inflammation in diabetic complications. Nephrol Dial Transplant. 2005;20(12):2601–4.
Brown Z, Robson RL, Westwick J. Regulation and expression of chemokines: potential role in glomerulonephritis. J Leukoc Biol. 1996;59(1):75–80.
Lee HY, Noh HJ, Gang JG, Xu ZG, Jeong HJ, Kang SW, Choi KH, Han DS. Inducible nitric oxide synthase (iNOS) expression is increased in lipopolysaccharide (LPS)-stimulated diabetic rat glomeruli: effect of ACE inhibitor and angiotensin II receptor blocker. Yonsei Med J. 2002;43(2):183–92.
Kanwar YS, Wada J, Sun L, Xie P, Wallner EI, Chen S, Chugh S, Danesh FR. Diabetic nephropathy: mechanisms of renal disease progression. Exp Biol Med. 2008;233(1):4–11.
Teramoto K, Negoro N, Kitamoto K, Iwai T, Iwao H, Okamura M, Miura K. Microarray analysis of glomerular gene expression in murine lupus nephritis. J Pharmacol Sci. 2008;106(1):56–67.
Campbell S, Burkly LC, Gao HX, Berman JW, Su L, Browning B, Zheng T, Schiffer L, Michaelson JS, Putterman C. Proinflammatory effects of TWEAK/Fn14 interactions in glomerular mesangial cells. J Immunol. 2006;176(3):1889–98.
Mezzano S, Aros C, Droguett A, Burgos ME, Ardiles L, Flores C, Schneider H, Ruiz-Ortega M, Egido J. NF-κB activation and overexpression of regulated genes in human diabetic nephropthy. Nephrol Dial Transplant. 2004;19(10):2505–12.
Utimura R, Fujihara CK, Mattar AL, Malheiros DM, Noronha IL, Zatz R. Mycophenolate mofetil prevents the development of glomerular injury in experimental diabetes. Kidney Int. 2003;63(1):209–16.
Kikuchi Y, Imakiire T, Yamada M, et al. Mizoribine reduces renal injury and macrophage infiltration in non-insulin-dependent diabetic rats. Nephrol Dial Transplant. 2005;20(8):1573–81.
Lee SY, Cho JY. Inhibitory effects of honokiol on LPS and PMA-induced cellular responses of macrophages and monocytes. BMB Rep. 2009;42(9):574–9.
Sheu ML, Chiang CK, Tsai KS, Ho FM, Weng TI, Wu HY, Liu SH. Inhibition of NADPH oxidase-related oxidative stress-triggered signaling by honokiol suppresses high glucose-induced human endothelial cell apoptosis. Free Radic Biol Med. 2008;44(12):2043–50.
Kim BH, Cho JY. Anti-inflammatory effect of honokiol is mediated by PI3K/Akt pathway suppression. Acta Pharmacol Sin. 2008;29(1):113–22.
Thorvaldson L, Stalhammar S, Sandler S. Effects of a diabetes-like environment in vitro on cytokine production by mouse splenocytes. Cytokine. 2008;43(1):93–7.
Hasegawa G, Nakano K, Sawada M, et al. Possible role of tumor necrosis factor and interleukin-1 in the development of diabetic nephropathy. Kidney Int. 1991;40(6):1007–12.
Leehey DJ, Isreb MA, Marcic S, Singh AK, Singh R. Effect of high glucose on superoxide in human mesangial cells: role of angiotensin II. Nephron Exp Nephrol. 2005;100(1):46–53.
Iglesias-de la Cruz MC, Ziyadeh FN, Isono M, Kouahou M, Han DC, Kalluri R, Mundel P, Chen S. Effects of high glucose and TGF-β1 on the expression of collagen IV and vascular endothelial growth factor in mouse podocytes. Kidney Int. 2002;62(3):901–13.
Clarkson MR, Murphy M, Gupta S, Lambe T, Mackenzie HS, Godson C, Martin F, Brady HR. High glucose-altered gene expression in mesangial cells. Actin-regulatory protein gene expression is triggered by oxidative stress and cytoskeletal disassembly. J Biol Chem. 2002;277(12):9707–12.
Stentz FB, Umpierrez GE, Cuervo R, Kitabchi AE. Proinflammatory cytokines, markers of cardiovascular risks, oxidative stress, and lipid peroxidation in patients with hyperglycemic crises. Diabetes. 2004;53(8):2079–86.
Tesch GH. MCP-1/CCL2: a new diagnostic marker and therapeutic target for progressive renal injury in diabetic nephropathy. Am J physiol, Renal physiol. 2008;294(4):F697–701.
Wu F, Yao H, Bader A, Dong F, Zhu F, Wu N, Wang B, Li H, Brockmeyer NH, Altmeyer P. Decorin gene transfer inhibited the expression of TGFβ1 and ECM in rat mesangial cells. Eur J Med Res. 2007;12(8):360–8.
Sánchez-López E, Rodriguez-Vita J, Cartier C, Rupérez M, Esteban V, Carvajal G, Rodrígues-Díez R, Plaza JJ, Egido J, Ruiz-Ortega M. Inhibitory effect of interleukin-1β on angiotensin II-induced connective tissue growth factor and type IV collagen production in cultured mesangial cells. Am J physiol, Renal physiol. 2008;294(1):F149–60.
Shui HA, Ka SM, Wu WM, Lin YF, Hou YC, Su LC, Chen A. LPS-evoked IL-18 expression in mesangial cells plays a role in accelerating lupus nephritis. Rheumatology (Oxford). 2007;46(8):1277–84.
Viberti GC, Bognetti E, Wiseman MJ, Benigni A, Remuzzi G. Glomerular hyperfiltration and urinary prostaglandins in type 1 diabetes mellitus. Diabet Med. 1989;6(3):219–23.
Yu Z, Zhang W, Kone BC. Signal transducers and activators of transcription 3 (STAT3) inhibits transcription of the inducible nitric oxide synthase gene by interacting with nuclear factor κB. Biochem J. 2002;367(Pt 1):97–105.
Schwarz M, Radeke HH, Resch K, Uciechowski P. Lymphocyte-derived cytokines induce sequential expression of monocyte- and T cell-specific chemokines in human mesangial cells. Kidney Int. 1997;52(6):1521–31.
Huang J, Siragy HM. Glucose promotes the production of interleukine-1β and cyclooxygenase-2 in mesangial cells via enhanced (Pro)renin receptor expression. Endocrinology, 2009;150(12):5557–5565.
Acknowledgments
This work was supported by grants from Science and Technology Foundation (2009C33150, 20090833B24) and Education Foundation (20070114) of Zhejiang Provincial, China. This manuscript is proofread by a native English professional with science background at Elixigen Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Ian Ahnfelt-Rønne.
J.-P. Wu and W. Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wu, JP., Zhang, W., Wu, F. et al. Honokiol: an effective inhibitor of high-glucose-induced upregulation of inflammatory cytokine production in human renal mesangial cells. Inflamm. Res. 59, 1073–1079 (2010). https://doi.org/10.1007/s00011-010-0227-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-010-0227-z